Neurodegenerative diseases comprise a class of disorders characterized by a progressive loss of neurological function, either by cell death or dysfunction. The most common disorders of this class are Alzheimer’s disease (AD), Parkinson’s disease (PD), Huntington’s disease (HD) and amyotrophic lateral sclerosis (ALS) also known as Lou Gehrig’s disease. While these diseases appear to have variable causes at the molecular, cellular and systems levels, they all manifest as a continued loss of brain function characterized by behavioral and cognitive deficits.
While neurodegenerative diseases appear to be a uniquely human phenomenon, a plethora of approaches for modeling the disorders in rodents has been developed. These models fall into two major categories: pharmacologically induced degenerations (i.e. lesion models) that effectively reproduce the consequences of the disease, or genetic modifications intended to recapitulate both the causes and consequences of the diseases.
Behavioral tests for evaluating the consequences of neurodegenerative diseases mostly concern memory (spatial, working and episodic), attention and the broad domain of executive function, which is comprised of reasoning, decision-making, and cognitive flexibility. These behaviors are all impacted by the various neurodegenerative diseases, thus there is a great deal of overlap between the tests used across the various major diseases listed above. However, few models recapitulate the entire spectrum of behavioral aberrations seen in the human disease and thus choosing an appropriate model to test a given aspect of each disease is paramount.[1]
Here, we will discuss the most widely studied and modeled neurodegenerative diseases, their common rodent models and means of assay, as well as the successes and pitfalls of the animal model approach to their study. First, however, we must consider the variables determining the selection of an appropriate animal model of neurodegenerative disease.
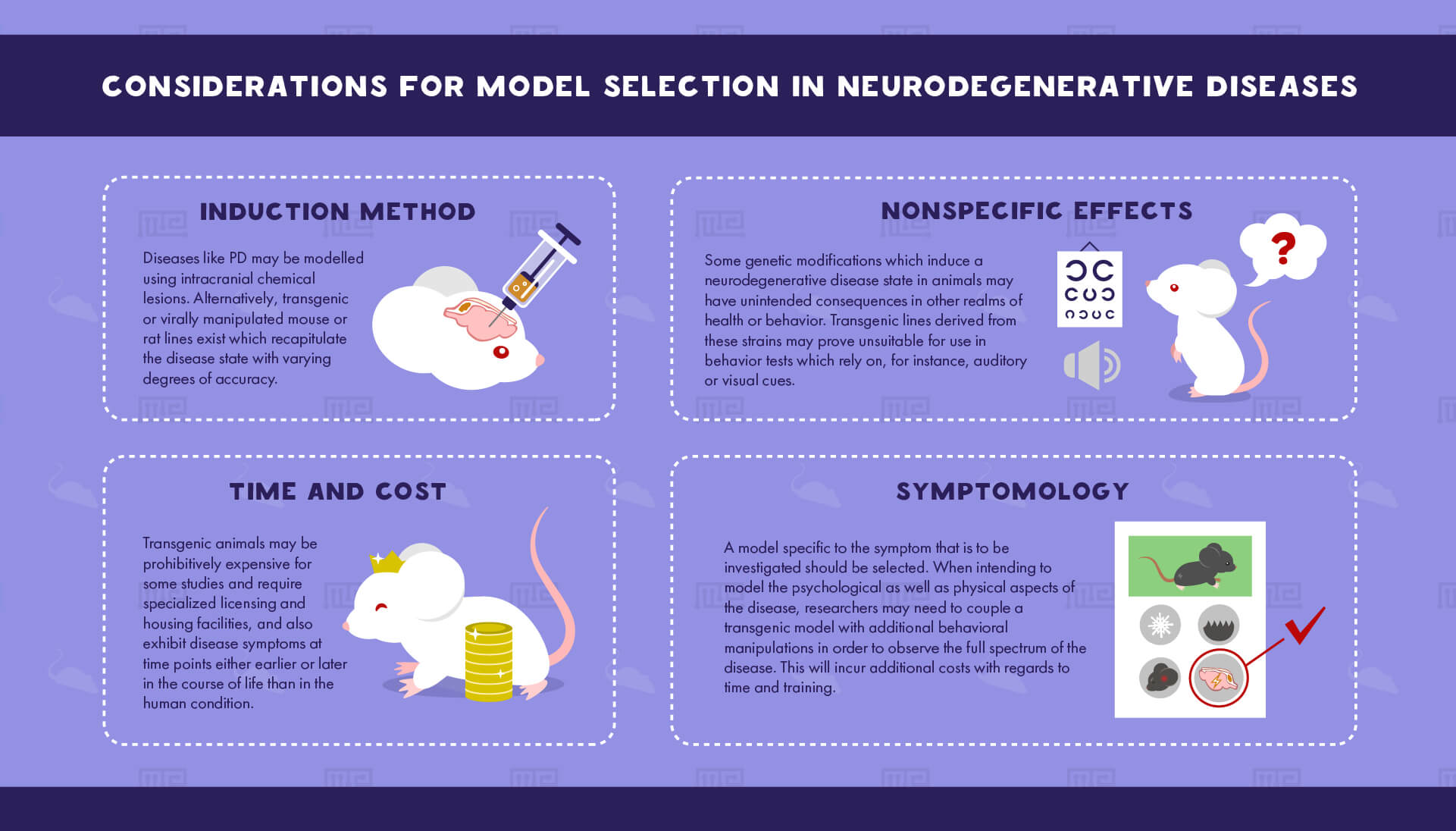
Considerations for Model Selection
Selecting an appropriate model for the investigation of a neurodegenerative disease requires significant consideration. For any given disease, there exist numerous (sometimes hundreds) of potential animal models, each with its own particularities.
Induction method
The first consideration when deciding on an appropriate model is the disease induction method. Many diseases, such as PD, may be modeled using intracranial chemical lesions. Alternatively, transgenic or virally manipulated mouse or rat lines exist which recapitulate the disease state with varying degrees of accuracy. The advantage of the chemical lesion method is that this approach is usually significantly less expensive as compared to the acquisition of a transgenic rodent line. However, given that neurodegenerative diseases are progressive, the effects of a neurochemical lesion will not model the disease with the same accuracy as many transgenic lines. Models induced with neurotoxic lesions exhibit symptoms of the condition very quickly (within several days) following the introduction of a toxin, as opposed to progressively over the course of life as in the case of a transgenic line. This may be advantageous for studies which intend to look at a short period of effects, or for laboratories which lack sufficient housing to keep animals for many months but do not provide an accurate picture of the time course of the disease state in humans. For example, mice of the transgenic PDAPP model of AD, which effectively mirrors the molecular symptoms of the disease, show no difference in the crucial test of novel object recognition at 3-5 months of age but show significant deficits when tested between 6 and 11 months of age.[2] This lengthy developmental period of the disease may pose logistical challenges for some researchers.
A final note in considering chemical neurotoxin versus genetically induced disease models is the global nature of most neurodegenerative diseases. Unless multiple intracranial injection sites are used, the effects of a chemically induced model will be limited to one brain structure or region, which is not an accurate reflection of the more global state of these diseases in humans. Systems-level compensations by interconnected brain regions may play an important role in the human disease condition, and thus selective chemical ablation of individual brain regions will not offer insight into the disease in this regard.
Time and cost
Despite the advantages of using a transgenic rodent line, which include progressive development of the disease and more global representation of neuronal consequences, there are disadvantages to their employment. First, transgenic animals may be prohibitively expensive for some studies and require specialized licensing and housing facilities. Additionally, many transgenic rodent models exhibit disease symptoms at time points either earlier or later in the course of life than in the human condition. Thus, a chemical induction model may be used to more appropriately select an age of maturity concurrent with the disease state in humans.
Symptomology
In addition to the considerations of induction methods, as well as the time and cost of individual models, researchers must select a model specific to the symptom that they would like to investigate. While the disease states in humans are comprised of both physical and mental declines, animal models often recapitulate only a fraction of these symptoms. For example, a significant percentage of HD-related mortality is the result of suicide following severe anxiety and depression. However, the rodent models of HD focus primarily on the loss of motor coordination which is characteristic of the disease. Therefore, researchers intending to model the psychological as well as physical aspects of the disease may need to couple a transgenic model with additional behavioral manipulations in order to observe the full spectrum of the disease. This will incur additional costs with regards to time and training. Similarly, in models which do exhibit both the psychological and physical symptoms of a disease, these aspects do not always present in the same order of time as in the human.
Nonspecific effects
Finally, some genetic modifications which induce a neurodegenerative disease state in animals may have unintended consequences in other realms of health or behavior. For instance, the R6/2 mouse model of HD (discussed in more detail below) exhibits a high likelihood of developing diabetes and cardiovascular abnormalities which are not present in human HD patients. Of a more global consideration for the use of inbred mouse strains for the genetic modelling of neurodegenerative diseases is the concern of accelerated aging in sensory perception. Many modified strains of C57BL mice, often used for the creation of transgenic lines, exhibit high rates of hearing loss earlier in life as compared to outbred strains, for instance. Others, such as SWR/J and CBA/J mice which are also frequently employed for transgenic modeling, exhibit comparatively rapid visual degeneration. Thus, transgenic lines derived from these strains may prove unsuitable for use in behavior tests which rely on auditory or visual cues, respectively.
Amyotrophic Lateral Sclerosis
ALS is a fatal neurodegenerative disease characterized by loss of both upper and lower motor neurons (UMN and LMN, respectively). In humans, the disease progresses rapidly, leading to death from respiratory failure within 3-5 years. The majority of ALS cases are classified as “sporadic” ALS (sALS) and are believed to be caused by environmental factors such as toxin exposure, while the remaining 10% are genetically inherited, termed “familial” ALS (fALS) .[3] Thus, in modeling the disease in rodents, approaches exist for both inducing UMN and LMN death via toxin exposure as well as through genetic manipulation.
BSSG Mouse Model (sALS)
The most common rodent model for sALS is exposure to β-sitosterol β-d-glucoside (BSSG), a toxin most often obtained from cycad seed flour. Mice fed BSSG over the course of 10-15 weeks exhibit significant losses of both UMN and LMN which progress at a rate not dissimilar to the disease time-course in humans, continuing even after the toxin-feeding has ceased. Along with the UMN and LMN loss, the mice develop behavioral abnormalities which clearly mimic those seen in the disease state in humans.[4]
Specifically, BSSG-exposed mice have shown deficits in basic motor functions as well as in cognitive tasks examining reference memory. Using the rotarod test, BSSG-exposed mice have been found to exhibit altered posture, concurrent with limb extension problems commonly seen in ALS patients. Interestingly, despite no differences in swimming speed, BSSG-exposed mice tested in the Morris water maze exhibit increased latency to platform finding, indicating deficits in the reference memory. Similarly, these mice show significantly increased errors in the radial arm maze, an additional test of reference memory.
All of these behavioral symptoms, paired with the notable loss of UMN and LMN induced by dietary BSSG, indicate that cycad seed feeding is a valuable model for sALS in mice. Of note, however, exposure to this toxic dietary additive produces slightly different neurodegenerative symptomology in rats than in mice, leading to more Parkinsonian-like outcomes in the former.[5]
TDP-43 Mouse Model (fALS)
Many cases of fALS are caused by a mutation in the gene encoding for TDP-43, a nuclear protein found in cytoplasmic protein aggregates of advanced fALS patients. While more than 20 transgenic mouse models targeting TDP-43 have been developed, the model with the most consistent measures of translational validity is the TDP43-Q331K model in which the human form of TDP-43 is moderately overexpressed.[6] Of note, however, despite displaying a wide spectrum of ALS-like phenotypic symptoms, TDP43-Q331K mice show primarily LMN loss with no UMN degeneration. They also do not exhibit death by respiratory failure, as the disease progression halts at 20 months. Nonetheless, the behavioral phenotype is still amongst the most valid of fALS models.
TDP43-Q331K transgenic mice exhibit progressive degeneration of motor skills akin to those seen in fALS patients. Using a grip-strength meter, the mice were found to show increasingly weaker hindlimb grip strength up to 10 months of age. When tested in the rotarod apparatus, the animals show increasingly shorter latencies to falling, as tested at 3, 6 and 10 months of age.[7] Both of these findings are consistent with continued LMN loss over the course of life in a similar fashion to human fALS patients. Thus, despite the aforementioned caveats, TDP43-Q331K transgenic mice provide a largely valid model for the 10% of ALS cases caused by genetic abnormalities.
Alzheimer’s Disease
A very significant body of evidence suggests that the primary underlying cause of AD is the progressive accumulation of amyloid ß-peptide (Aß) and neurofibrillary tangles (NFTs). These accumulations lead to significant inflammation and subsequently, neuronal dysfunction. While more than 100 genetic mouse models of AD exist, the most commonly used models are designed to increase the rate of amyloid precursor protein cleavage (leading to Aß accumulation) or to overexpress mutated tau proteins (which then results in NFT production).[8]
Mouse lines which increase the rate of amyloid precursor protein cleavage and lead to Aß accumulation have variable behavioral outcomes. Unfortunately, the memory deficits in humans are often in the verbal domain, which is not something that can be easily tested in mice. However, the verbal memory decline in humans is indicative of a problem with working memory (temporary information holding and processing), which has a great deal of neurological overlap with spatial working memory. Thus, the tasks most often used to model AD in mice are those concerned with spatial learning and memory such as the Morris water maze, though simple tests such as the T-maze and Y-maze are also often employed.[9]
PDAPP Mouse Model
One of the earliest and most widely used of the AD models is the PDAPP transgenic mouse line, first described in 1995.[10] By 4 months of age, PDAPP mice exhibit significant deficits in the Morris water maze (thereby indicating a reduction in spatial memory function) which persist throughout their lives.[11] By 6 months of age, these same animals show reduced novel object recognition, a test of working memory and attention.[12] Notable, however, is that these animals do not exhibit deficits in recognition memory until after 12 months of age.[13] Thus, researchers seeking to model multiple aspects of AD will be required to employ this model for one full year or more if they intend to investigate the full spectrum of memory dysfunction.
TgCRND8 Mouse Model
A more recent model of AD, the TgCRND8 transgenic line first developed in 2006, models the NFTs associated with the disease.[14] While these mice also exhibit deficits in the Morris water maze, they additionally develop problems with associative learning, as shown by a failure to acquire fear conditioning. In a similar fashion to the disease progression in humans, the mice exhibit significant deficits in executive function as they age. Such aberrations are shown by problems in performing the decision making tasks employed in the T-maze and Y-maze alternation paradigms.[15]
Thus, while certain transgenic mouse lines created to model AD show selective deficits associated with the disease, others exhibit a more comprehensive spectrum of cognitive problems. Testing with the Morris water maze, novel object recognition, Y-maze or T-maze, and through fear conditioning can assess problems in the domains of spatial memory, attention and working memory, executive function, and associative learning, respectively.
Parkinson’s Disease
Parkinson’s disease (PD) is characterized by a loss of dopamine neurons in subregions of the striatum, particularly in the substantia nigra. It is a complex progressive disorder which has been intensively studied and has been described in significant detail here.
Traditional models for the disease began with intracranial lesions of this region using the neurotoxic drugs MPTP or 6-OHDA. While these models do still exist, they are extremely limited in modeling the disease as they are non-progressive (i.e. the effects appear immediately, rather than slowly as in the real disease state) and the behavioral consequences only recapitulate the movement disorders of the disease.[16]
Conversely, following the discovery of genes linked to the development of PD, numerous transgenic models have been developed to better mimic the spectrum and time-course of effects in human patients. Interestingly, while dopaminergic cell loss normally reaches >50% in the substantia nigra of human brains before they develop significant symptoms of PD, mouse models tend to exhibit behavioral symptoms prior to this point. On the contrary, certain genetic modifications to rats, rather than mice, result in a more accurate rate of cell loss prior to behavioral displays of the disease.[17] Thus, the current models of PD considered to most accurately reflect the human condition are the rat models.
PINK1 Rat Model
PINK1-KO rats are engineered to lack the mitochondrial kinase PTEN-induced kinase 1 (PINK1) which is normally responsible for activating Parkin, which in turn sequesters defective mitochondria (a process which is inhibited in PD patients). In this model, PINK1-KO rats exhibit age-dependent loss of dopaminergic neurons beginning at 6-8 months of age (which would correlate to 30-40 years of age in humans, the time for early-onset PD). By this time, the rats exhibit deficits in locomotor activity including reduced speed and distance traveled in an open field apparatus, and deficits in balance beam procedures.[18]
α-Synuclein Mouse Model
The second recently developed rat model of PD is the α-Synuclein transgenic model. In human PD, the accumulation of protein tangles known as Lewy bodies is one of the primary markers of the disease, and they are largely comprised of α-Synuclein. While many mouse models transgenically targeting α-Synuclein surprisingly fail to exhibit dopamine cell loss and PD-like behavioral symptoms, the rat models succeed in this regard. Specifically, the BAC transgenic rats that overexpress human α-Synuclein exhibit reduced striatal dopamine cells by 12 months, which progresses significantly by 18 months. At this earlier time point, these rats show PD-like locomotor deficits, including a reduction in travel distance and rearing behavior in open field environments.[19]
Outside of the motor symptoms, BAC transgenic rats overexpressing human α-Synuclein show many other symptoms of PD seen in humans including reduced odor discrimination in a two-chamber olfaction test and decreased novelty-seeking behavior. Adding to the translational validity of this model, reductions in odor discrimination appear before motor dysfunction – an important distinction which has lead to olfaction testing as an early identification test for PD patients.
Furthermore, like human PD patients, these rats show elevated levels of anxiety, as shown by reduced exploration and time spent in open areas of open field environments as well as decreased crossing likelihood in the elevated beam test. Together with the aforementioned motor abnormalities which develop along with Lewy body like protein tangles in these rats, these behavioral symptoms characterize a well-rounded model of PD.[19]
Huntington’s Disease
Huntington’s disease (HD) is a neurodegenerative disorder with extremely wide-reaching symptoms. The disease characteristics include motor impairment and involuntary movements as well as cognitive deficits in the domains of executive function (planning, cognitive flexibility, abstract thinking..etc.) and memory, along with neuropsychiatric symptomology (depression, anxiety, and irritability). Caused by a heritable mutation of the huntingtin gene (htt), the disease currently has no cure and most commonly begins at mid-life, progressing towards death within 15 to 20 years of onset.
Currently, a number of rodent models of HD exist, though none fully recapitulate the time-course and full spectrum of disease symptomology. Transgenic HD models have been developed in both mice and rats, with the latter being advantageous due to rats having more complex behaviors than mice, as well as their larger brain mass leading to an increased range of potential neurological assays such as PET imaging which are substantially more difficult in mice. However, due to availability and efforts towards research continuity, mouse models of the disease remain more widely used.[20]
R6/2 mouse model
The transgenic mouse model of HD most often employed is the R6/2 model which expresses a truncated form of the mutated human htt, specifically an exon 1 fragment with approximately 150 allele repeats. The R6/2 mouse develops cognitive signs of the disease as early as 5 weeks of life. The symptoms progress continually throughout their lives, during which the animals exhibit disordered learning and memory as indicated by deficits in the Morris water maze and T-maze among others.[21] In the Morris water maze, changing the location of the platform after learning acquisition may be used as a measure of cognitive flexibility (commonly known as “reversal” training). In this domain as well, R6/2 mice show diminished capacity, in line with the inhibited executive functions seen in HD patients.
The R6/2 HD model also displays significant motor deficits in line with the human condition. Using elevated beams, forced swim tests, and the rotarod test, these motor abnormalities have been observed as early as 5 weeks, though they progress to a more profound level at 8 weeks of age.[22] Finally, using a startle chamber, R6/2 mice have been evaluated for function in the prepulse inhibition assay (also known as the acoustic startle test), in which they develop deficits at approximately 12 weeks of age. As this symptom appears long after the onset of motor abnormalities, it is likely indicative of cognitive decline similar to what is seen in HD patients.
Despite these many similarities in both the motor and cognitive domains of HD between R6/2 mice and human HD patients, there are a number of caveats to the translational validity of the model. Specifically, the early onset of the symptoms is more akin to the relatively rare juvenile-onset form of the disease found in humans than to the more common adult-onset form. Additionally, R6/2 mice are more likely to co-present with epilepsy and cardiac dysfunction, as well as diabetes, none of which are common symptoms of adult-onset HD. Thus, while R6/2 mice may provide a valuable model for certain HD aspects, they do not provide a full and accurate picture of the disease.
HD Rat Model
Unlike the previously discussed HD mouse model, the HD rat model expresses a chimeric form of the rat and human htt gene (rather than only the human htt). In an additional contrast, rather than an early onset of the disease, the rat HD model exhibits a time-course more in line with adult-onset HD. At approximately 10 months of age, HD rats begin to show signs of cognitive decline,[23] indicated by poor performance in the choice reaction time task. At 15 months, the rats exhibit involuntary movements characteristic of HD as observed in the open field setting.
As noted earlier, the larger size of the rat brain leads to the potential for PET imaging. Indeed, researchers have found enlarged ventricles and striatal lesions in this model, concurrent with neurological symptomology in HD patients. However, the development of this model is still in the early stages as compared to the more commonly used mouse models, and thus caution is advised in its use. Nonetheless, the current findings and the vast potential for this rat model of HD indicates potentially immense translational value in researching the disease.
Translational Value of Neurodegenerative Disease Models
The modeling of neurodegenerative diseases in rodents presents several problems with regards to translational value. First, many neurodegenerative diseases do not yet have a clearly defined cause. While certain diseases are associated with genetic mutations, these are more often risk factors than direct causes. Rather than a single inducing factor, genetic variants frequently predispose an individual to a disease. This predisposition may lead to the disease when coupled with other factors such as environmental or chemical insults including toxin exposure, immune challenges or other neurological stressors.
In addition to these challenges, models of neurodegenerative diseases often succeed in recapitulating only part of the spectrum of a disorder. While many models of ALS successfully achieve motor dysfunctions for instance, these same models often do not exhibit the psychological aspects of the disease such as depression. Since the brain exists as a complex system of interacting parts, no one symptom presents a realistic picture of the overall impact on brain function.
Finally, neurodegenerative diseases are all progressive in nature, with many aspects of the disease arising or developing at different rates and at different points in the life cycle. These aspects often fail to be reliably reproduced in animal models of the diseases and therefore do not represent an accurate template on which to assay potential treatments.
Overall, the reductionist approach used in modeling neurodegenerative diseases has offered many valuable insights into the nature of the disorders and has lead to the discovery of early biomarkers and behavioral indicators of the disease which allow for early interventions. However, effective treatments or cures have rarely emerged from animal models of neurodegenerative diseases.
Successful translation
In one of the more successful examples of translation from an animal model to human therapeutic intervention, the development of PDAPP transgenic mice modeling AD led to a successful treatment in a case study of AD human patients in less than a decade. Using the PDAPP mouse model, researchers found in 1999 that immunization with the Aß peptide prevented the development of both the neurological and behavioral aspects of the disease.[24] By 2003, researchers published a case-report of application of this treatment in human AD patients with a high rate of success.[25]
Unsuccessful translation
Conversely, early evidence showed that the acetylcholinesterase inhibitor Rivastigmine significantly improved the behavioral symptoms of AD in multiple mouse models of the disease, leading to early excitement that an effective pharmacotherapeutic intervention had been found.[26] The drug was found to improve performance in the Morris water maze in addition to other tests. Nonetheless, when eventually tested in human AD patients, the drug was found to be ineffective in treating any of the symptoms for which it was useful in the mouse models.[27] Issues of metabolic differences and incompleteness of the mouse models were cited as possible explanations, highlighting the often incompatible translational prospects of treatments between rodents and humans.
Conclusions
The study of neurodegenerative diseases comprises an important and complex field of research. While many rodent models of the key diseases discussed herein have been developed, none recapitulate every aspect of the disease symptomology and time-course. Choosing an animal model to best present the symptoms of interest is paramount to successful research. Important considerations include the type of symptom (motor or cognitive), the importance of time of onset and rate of progression, and the additional abnormalities which any given model may present that do not occur in the human condition. Selection of an appropriate testing apparatus is equally crucial in experimental design, and as outlined above, neurodegenerative diseases may lead to behavioral abnormalities in a very large range of potential tests. Therefore, the foundations of experimental design in the domain of neurodegenerative disease research are crucially concerned with both animal model selection and behavioral assay determination.
For our apparatuses used in assessing neurodegenerative models see our the Catalepsy test, the Grip Strength test, the Balance Beam, the Parallel Bars, the Geotaxis Test, the Static Rods Test, the Gait Test, the Parallel Rod test, and the Pole test. See our activity range here.
References
- Jucker, M. (2010). The benefits and limitations of animal models for translational research in neurodegenerative diseases. Nature Medicine, 16, 1210.
- Cramer, P. E., Cirrito, J. R., Wesson, D. W., Lee, C. Y. D., Karlo, J. C., Zinn, A. E., … Landreth, G. E. (2012). ApoE-Directed Therapeutics Rapidly Clear β-Amyloid and Reverse Deficits in AD Mouse Models. Science, 335(6075), 1503.
- Taylor, J. P., Brown, R. H., & Cleveland, D. W. (2016). Decoding ALS: from genes to mechanism. Nature, 539(7628), 197-206.
- Morrice, J. R., Gregory-Evans, C. Y., & Shaw, C. A. (2018). Animal models of amyotrophic lateral sclerosis: A comparison of model validity. Neural regeneration research, 13(12), 2050-2054.
- Van Kampen, J. M., Baranowski, D. C., Robertson, H. A., Shaw, C. A., & Kay, D. G. (2015). The Progressive BSSG Rat Model of Parkinson’s: Recapitulating Multiple Key Features of the Human Disease. PloS one, 10(10), e0139694.
- Lutz, C. (2018). Mouse models of ALS: Past, present and future. RNA Metabolism in Neurological Disease 2018, 1693, 1–10.
- Arnold, E. S., Ling, S. C., Huelga, S. C., Lagier-Tourenne, C., Polymenidou, M., Ditsworth, D., Kordasiewicz, H. B., McAlonis-Downes, M., Platoshyn, O., Parone, P. A., Da Cruz, S., Clutario, K. M., Swing, D., Tessarollo, L., Marsala, M., Shaw, C. E., Yeo, G. W., … Cleveland, D. W. (2013). ALS-linked TDP-43 mutations produce aberrant RNA splicing and adult-onset motor neuron disease without aggregation or loss of nuclear TDP-43. Proceedings of the National Academy of Sciences of the United States of America, 110(8), E736-45.
- Jankowsky, J. L., & Zheng, H. (2017). Practical considerations for choosing a mouse model of Alzheimer’s disease. Molecular neurodegeneration, 12(1), 89.
- Webster, S. J., Bachstetter, A. D., Nelson, P. T., Schmitt, F. A., & Van Eldik, L. J. (2014). Using mice to model Alzheimer’s dementia: an overview of the clinical disease and the preclinical behavioral changes in 10 mouse models. Frontiers in genetics, 5, 88.
- Games, D., Adams, D., Alessandrini, R., Barbour, R., Borthelette, P., Blackwell, C., … Zhao, J. (1995). Alzheimer-type neuropathology in transgenic mice overexpressing V717F β-amyloid precursor protein. Nature, 373, 523.
- Daumas, S., Sandin, J., Chen, K., Kobayashi, D., Tulloch, J., Martin, S., … Morris, R. (2008). Faster forgetting contributes to impaired spatial memory in the PDAPP mouse: Deficit in memory retrieval associated with increased sensitivity to interference? (Vol. 15).
- Dodart, J.-C., Meziane, H., Mathis, C., Bales, K. R., Paul, S. M., & Ungerer, A. (1999). Behavioral disturbances in transgenic mice overexpressing the V717F Β-amyloid precursor protein. Behavioral Neuroscience, 113(5), 982-990.
- Yassine, N., Lazaris, A., Dorner-Ciossek, C., Després, O., Meyer, L., Maitre, M., … Mathis, C. (2013). Detecting spatial memory deficits beyond blindness in tg2576 Alzheimer mice. Neurobiology of Aging, 34(3), 716–730.
- Oakley, H., Cole, S. L., Logan, S., Maus, E., Shao, P., Craft, J., … Vassar, R. (2006). Intraneuronal β-Amyloid Aggregates, Neurodegeneration, and Neuron Loss in Transgenic Mice with Five Familial Alzheimer’s Disease Mutations: Potential Factors in Amyloid Plaque Formation. The Journal of Neuroscience, 26(40), 10129.
- Shukla, V., Zheng, Y. L., Mishra, S. K., Amin, N. D., Steiner, J., Grant, P., Kesavapany, S., … Pant, H. C. (2013). A truncated peptide from p35, a Cdk5 activator, prevents Alzheimer’s disease phenotypes in model mice. FASEB journal : official publication of the Federation of American Societies for Experimental Biology, 27(1), 174-86.
- Creed, R. B. and Goldberg, M. S. (2018), New Developments in Genetic rat models of Parkinson’s Disease. Mov Disord., 33: 717-729.
- Dave, K. D., De Silva, S., Sheth, N. P., Ramboz, S., Beck, M. J., Quang, C., … Frasier, M. A. (2014). Phenotypic characterization of recessive gene knockout rat models of Parkinson’s disease. Neurobiology of Disease, 70, 190–203.
- Grant, L. M., Kelm-Nelson, C. A., Hilby, B. L., Blue, K. V., Paul Rajamanickam, E. S., Pultorak, J. D., Fleming, S. M., … Ciucci, M. R. (2015). Evidence for early and progressive ultrasonic vocalization and oromotor deficits in a PINK1 gene knockout rat model of Parkinson’s disease. Journal of neuroscience research, 93(11), 1713-27.
- Nuber, S., Harmuth, F., Kohl, Z., Adame, A., Trejo, M., Schönig, K., Zimmermann, F., Bauer, C., Casadei, N., Giel, C., Calaminus, C., Pichler, B. J., Jensen, P. H., Müller, C. P., Amato, D., Kornhuber, J., Teismann, P., Yamakado, H., Takahashi, R., Winkler, J., Masliah, E., … Riess, O. (2013). A progressive dopaminergic phenotype associated with neurotoxic conversion of α-synuclein in BAC-transgenic rats. Brain : a journal of neurology, 136(Pt 2), 412-32.
- Heng, M. Y., Detloff, P. J., & Albin, R. L. (2008). Rodent genetic models of Huntington disease. Neurobiology of Disease, 32(1), 1–9.
- Lione, L. A., Carter, R. J., Hunt, M. J., Bates, G. P., Morton, A. J., & Dunnett, S. B. (1999). Selective Discrimination Learning Impairments in Mice Expressing the Human Huntington’s Disease Mutation. The Journal of Neuroscience, 19(23), 10428.
- Carter, R. J., Lione, L. A., Humby, T., Mangiarini, L., Mahal, A., Bates, G. P., … Morton, A. J. (1999). Characterization of Progressive Motor Deficits in Mice Transgenic for the Human Huntington’s Disease Mutation. The Journal of Neuroscience, 19(8), 3248.
- Cao, C., Temel, Y., Blokland, A., Ozen, H., Steinbusch, H. W. M., Vlamings, R., … Visser-Vandewalle, V. (2006). Progressive deterioration of reaction time performance and choreiform symptoms in a new Huntington’s disease transgenic ratmodel. Behavioural Brain Research, 170(2), 257–261.
- Schenk, D., Barbour, R., Dunn, W., Gordon, G., Grajeda, H., Guido, T., … Seubert, P. (1999). Immunization with amyloid-β attenuates Alzheimer-disease-like pathology in the PDAPP mouse. Nature, 400, 173.
- Nicoll, J. A. R., Wilkinson, D., Holmes, C., Steart, P., Markham, H., & Weller, R. O. (2003). Neuropathology of human Alzheimer disease after immunization with amyloid-β peptide: a case report. Nature Medicine, 9(4), 448–452.
- Van Dam, D., Abramowski, D., Staufenbiel, M. et al. (2005). Symptomatic effect of donepezil, rivastigmine, galantamine and memantine on cognitive deficits in the APP23 model. Psychopharmacology, 180(1), 180-177.
- Feldman, H. H., Ferris, S., Winblad, B., Sfikas, N., Mancione, L., He, Y., … Lane, R. (2007). Effect of rivastigmine on delay to diagnosis of Alzheimer’s disease from mild cognitive impairment: the InDDEx study. The Lancet Neurology, 6(6), 501–512.