Description
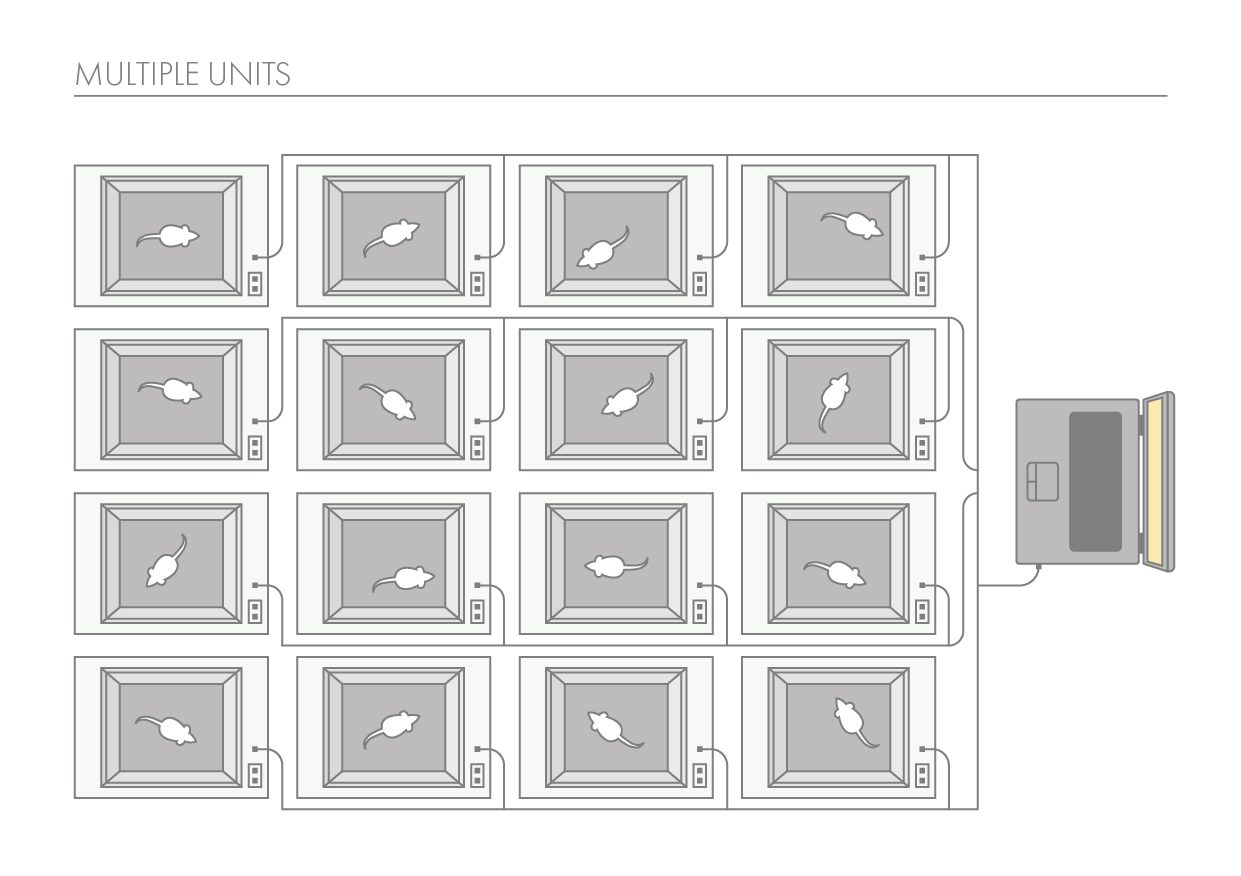
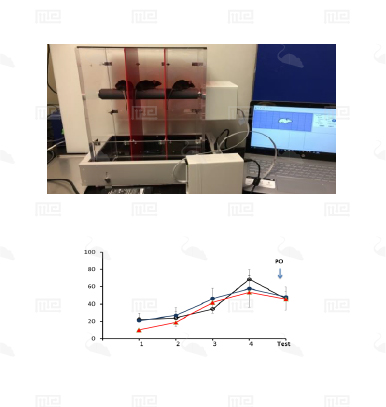
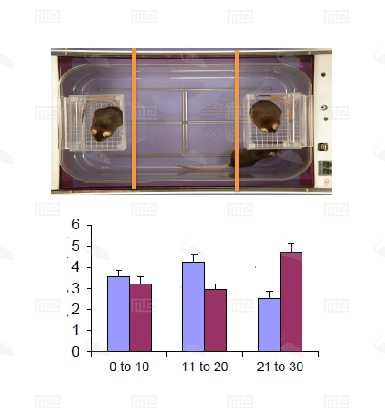
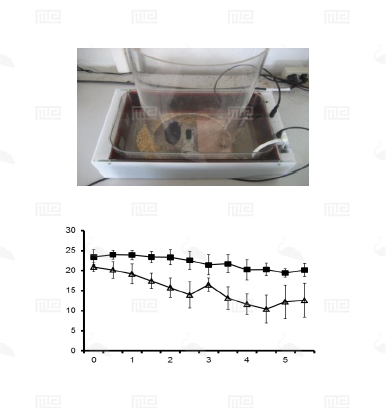
The SmartCageTM system is an automated non-invasive rodent behavioral monitoring system. The SmartCageTM system is a versatile, flexible, and user-friendly system that enables the biomedical researchers to conduct a variety of neurobehavioral assays for phenotypic analysis, in vivo drug screening, and assessment of neurobehavioral toxic compounds before testing in individual disease models through consistent and accurate monitoring of rodent home cage activity.
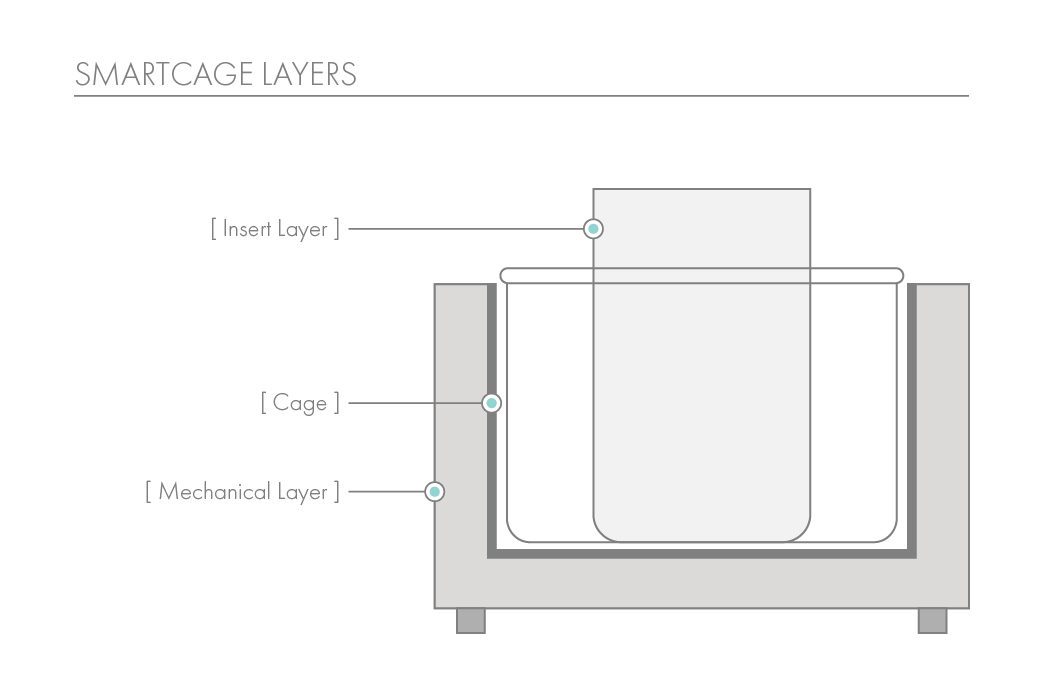
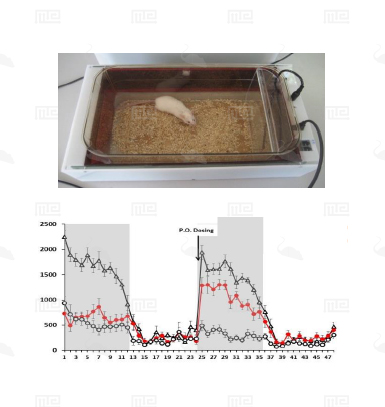
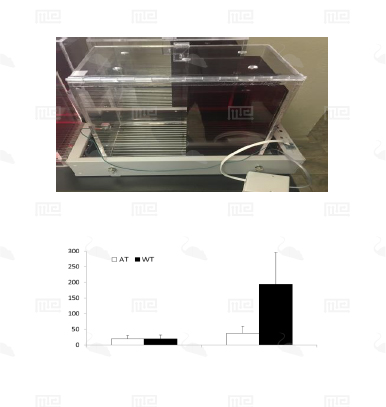
The SmartCage™ system is widely used for quantitative characterization of basic behavioral elements and their patterns in freely moving rodents. Each SmartCage™ system consists of a floor-vibration sensor, a motor control, an instrument amplifier, microcontroller units, an infrared (IR) matrix, and flexible modular devices. The system is non-invasive as it allows the animals to be tested and monitored in their home cages having bedding, food, and water, making the system valuable to conduct experimental manipulations and behavioral assessments for extended periods. The system automatically measures wake/active and sleep/inactive states. Locomotion (travel distance, travel time), rearing up counts, and animal movement patterns, for example, rotations (cycling) are the home cage activity variables that are recorded to develop the behavioral analysis.