Description
Five rods are included in the order. 35 mm, 28 mm, 22 mm, 15 mm and 9 mm width rods are included in your order for five total, each 60 cm in length. Each rod is fixed by a G-clamp to a laboratory shelf to assess motor function. The end of the rod near the bench has a mark 10 cm from the end, to denote the finishing line. The height of the rods above the floor is 60 cm.
OPTIONAL: If your laboratory does not have a 60cm bench, an optional wall with soft landing compartment can be ordered. Inquire for more details.
Other apparatuses used in assessing rodent motor function include the Grip Strength test, the Balance Beam, the Parallel Bars, the Geotaxis Test, the Gait Test, the Parallel Rod test, the Catalepsy Bar test, and the Pole test. See our activity range here.
Price and Size
Mouse & Rat
$ 990
+SH- Rod 1: 35mm, 60 cm length
- Rod 1: 28mm, 60 cm length
- Rod 1: 22mm, 60 cm length
- Rod 1: 15mm, 60 cm length
- Rod 1: 9mm, 60 cm length
Documentation
Introduction
Motor coordination is an important part of any behavioral assay to achieve accurate results. The tasks such as the Morris Water Maze and the Forced Swim Test relies on the ability of the subject to swim in order to determine investigation parameters, thus requiring subjects with good motor skills. The performance of the subject in a motor coordination task is a good indicator for choosing subjects that serve as suitable candidates for the chosen behavioral assay. The Static Rods is one such task that helps determine the motor capabilities of the subject.
The Static Rods task is made of simple dowels and clamps. Dowels of varying thicknesses are used to increase the difficulty of the task and refine the assessment further. One end of the dowel is attached to the edge of a fixture such that they horizontally protrude out in the space at a certain height from the ground.
The task measures the motor coordination capabilities of the subject that can be affected by different pharmacological compounds, lesions, or diseases. Selecting the right candidate for research or investigation is a vital first step in achieving accurate results.
Apparatus and Equipment
The apparatus consists of five acrylic/wooden dowels or rods of diameter 35, 28, 22, 15 and 9 mm that are 60 cm long. G-clamp is used to fix the dowel horizontally to the edge of a laboratory shelf or desk. Only one end of the rod is fixed to a platform, while the other end protrudes into space.
The clamped end of the dowel is marked 10 cm from the bench to denote the finishing line. The dowels are placed at the height of 60 cm above the floor.
Training Protocol
The apparatus is thoroughly cleaned and sterilized before and after a session with different subjects. The rod is clamped into place using a G-clamp, 60 cm above the floor surface and a soft cushion is placed on the floor to avoid injury to the subject in case it falls.
The task does not require any pre-training and testing can be performed immediately once the apparatus is set-up. The subjects are brought into the room 5 to 20 minutes before testing to habituate and are allowed resting periods between each trial to allow muscle strength recovery.
To assist with the manual scoring, video and tracking software such as the Noldus Ethovision XT can also be used.
Evaluation of motor coordination
Begin testing with the widest rod (35 mm diameter) clamped to the platform. Place the subject with its back facing the clamped end, at the far end of the rod (free end), preferably one head’s length from the end. Begin the timer immediately.
Two measures are recorded during the task: Orientation time and Transit time. Orientation is dependent on the subject staying upright. Allocate a maximum score of 120 sec (arbitrarily set test time) in case the subject turns upside down and clings to the rod. Further, allocate maximum score for transit time too, since a subject that transits upright is more coordinated than that clings the rod upside down. In the situation wherein the subject orients upside down, it is not tested further on the narrower rods.
In case, after orienting, the subject falls or reaches the maximum test time, it is not tested on the narrower rods and is assigned the maximum score for that particular rod and the subsequent rods. Stop testing if the subject falls off after more than 5 seconds on the rod. In case, the subject falls within the initial 5 seconds of the testing session, replace it once again on the rod and allow a maximum of three trials, of which select the best results.
Once the subject completes the task or falls off the rod, remove it and place it in its home cage and allow it to recover before testing again. Repeat the trials with decreasing thicknesses of the rods in the same manner.
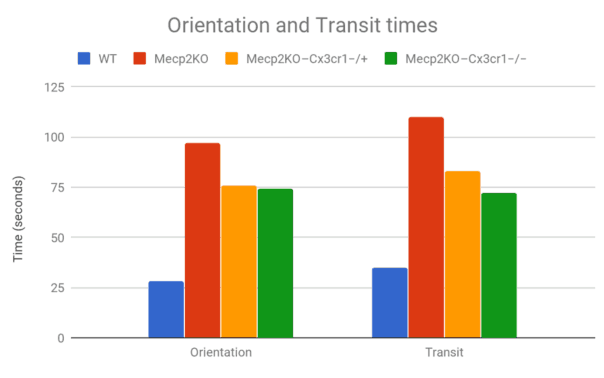
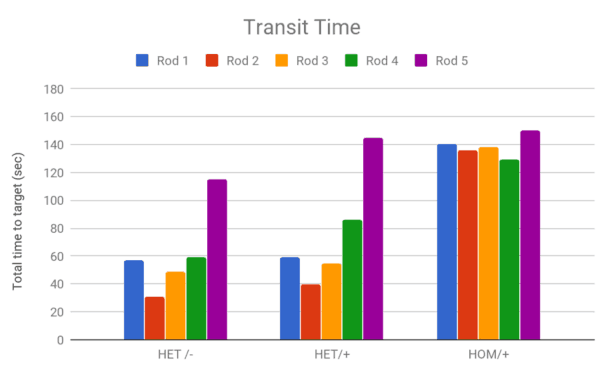
Sample Data
Two measures are recorded for the Static Rods task.
- Orientation time: The time taken to orientate 180° from the starting position towards the shelf.
- Transit time: The time taken to travel to the shelf end (nose beyond the 10 cm mark from the shelf end of the rod).
Other behaviors such as orienting upside-down are also recorded. A successful transit is recorded when the subject travels the rod in an upright position as it indicates good motor coordination.
Scoring is done based on the test time that is set. For example, if the test time is set to 120 seconds then falling or upside down orientation is allocated the maximum score of 120 seconds. The orientation time is allocated as the time taken by the subject to orient itself towards the clamped end and transit time as the time taken to cross the 10 cm finish line mark.
The data can be visualized as graphs to compare results between different strains of rodents or disease/intervention groups.
Strengths & Limitations
Strengths
Unlike the RotaRod, the Static Rods do not have a rotating element to it, thus making the Static Rods a simple and straightforward task that is easy to use and setup. The task serves as a simple measure of motor coordination and has been used in monitoring progress of diseases in mutant mice under different conditions (van Dellen et al., 2000).
Limitations
When positioning the subject, the experimenter may not correctly position the subject on the bar leading to the subject failing to grasp the bar and falling. Further, some subjects may fail to grip the bar assuming they are still supported.
Summary and Key Points
- The Static Rods is a static apparatus, unlike the RotaRod.
- The Static Rods measures motor coordination.
- Rods of varying diameter are used to refine the test.
- If the subject orients upside-down on the rod then it is allocated the maximum score for both orientation time and transit time.
- A subject that traverses the rod in an upright position has better motor coordination than the subject that orients upside-down on the rod.
- When the subject’s nose tip crosses the 10 cm finish mark, it is said to have completed the task successfully.
- Poor performance on the Static Rods can be seen in certain animal disease models, transgenic models, and subjects with certain lesions.
References
Deacon RM. Measuring motor coordination in mice. J Vis Exp. 2013 May 29;(75):e2609. doi: 10.3791/2609.
Horiuchi M, Smith L, Maezawa I, Jin LW. CX3CR1 ablation ameliorates motor and respiratory dysfunctions and improves survival of a Rett syndrome mouse model. Brain Behav Immun. 2017 Feb;60:106-116. doi: 10.1016/j.bbi.2016.02.014. Epub 2016 Feb 13.
Breuss MW, Hansen AH, Landler L, Keays DA. Brain-specific knockin of the pathogenic Tubb5 E401K allele causes defects in motor coordination and prepulse inhibition. Behav Brain Res. 2017 Apr 14;323:47-55. doi: 10.1016/j.bbr.2017.01.029. Epub 2017 Jan 25.
van Dellen A, Blakemore C, Deacon R, York D, Hannan AJ. Delaying the onset of Huntington’s in mice. Nature. 2000 Apr 13;404(6779):721-2.