Although neurons have traditionally received most of the attention when it comes to understanding disease pathophysiology, a paradigm shift is occurring that also brings microglia into the light.
A recent surge in findings has uncovered the link between diseases, microglia, and behavior. Such findings point to a new direction of behavioral research, one that also considers how microglia are involved in diseases.
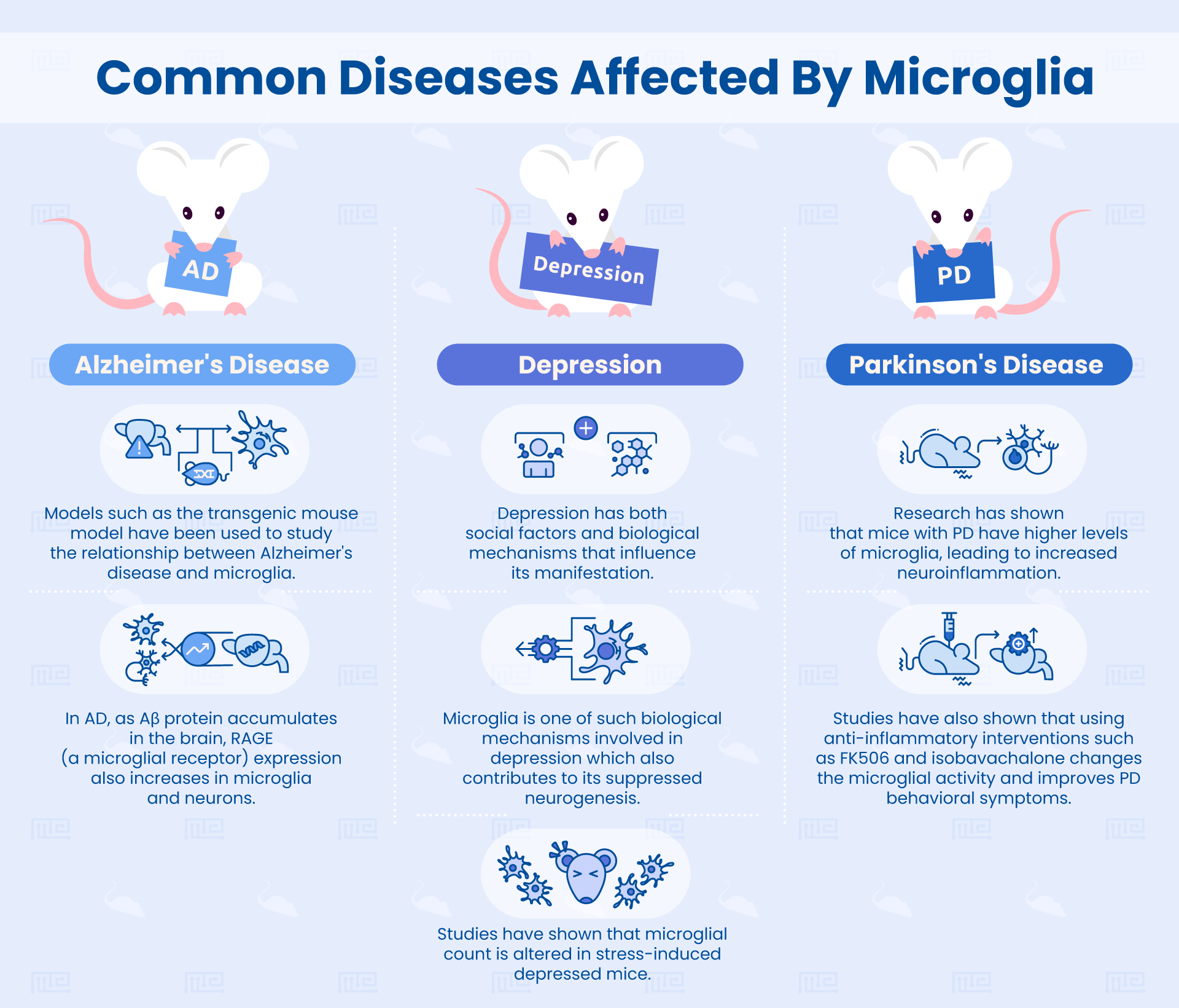
In this section, we will review some key research findings which link microglia to behavior in diseased states like depression, Parkinson’s, and Alzheimer’s disease.
But, first, to strengthen our background understanding of microglia, check out this introductory article Guide to Behavior and Microglia where we discuss what microglia are, including their function and various phenotypes.
Why Are Microglia of Interest to Behavioral Researchers Studying Diseases?
Ever since microglia have been implicated in affecting behavior, research has boomed, trying to figure out exactly how microglia contribute to disease pathophysiology.
Since microglia are a part of the central nervous system (CNS), current efforts have primarily focused on how microglia contribute to CNS diseases, while studying the subsequent outcomes on behavior and cognition.
Thus, researchers are interested in microglia since they could be an important piece of a puzzle, ultimately leading to a cure or at least more efficient treatment.
Microglia and Depression
Depression is a serious psychological condition with a rapidly increasing incidence in the USA, affecting youth, adults, and the elderly. While depression does have social factors influencing its manifestation, there are also biological mechanisms at play.[1]
A study by Kreisel et al. showed that changes in microglia underly depressive-like behavior and contribute to suppressed neurogenesis.[2] To induce depression, the researchers used a stress-induction method by exposing C57BL mice to a series of random stressors, including cage shaking, cold temperature exposure, and a wet cage.
The researchers found that the stress-induced depressed mice had:
- Increased microglial count after acute stress: There was an increase in microglia activity after 1-4 days of stress. A peak in microglial activity is a typical stress response.
- Decreased microglial count after chronic stress: The depressed mice had decreased microglial levels in the hippocampal dentate gyrus after 5 weeks of prolonged chronic stress. Also, the depressed mice had decreased levels of microglia in the medial prefrontal cortex but to a lesser extent. This shows how stress has various effects on microglia across time.
- Altered microglia physiology after chronic stress: Furthermore, the dentate gyrus microglia of the depressed mice had distorted morphology as indicated by the reduced length of the soma area and in microglial processes. Thus, chronic stress was able to affect microglial integrity. For more information about this, check out our article on how microglial physiology affects behavior.
- Decreased sucrose preference: In the Sucrose Preference Test, depressed mice had a significantly lower preference for the sweetened solution. Only 70% of depressed mice preferred the sweetened solution compared to 85% of control non-depressed mice. Normal, healthy mice are expected to prefer a sucrose solution over water. Lack of this preference is indicative of depression, anhedonia, and unresponsiveness to reward.
- Decreased social exploration: When challenged with the Social Interaction Test, depressed mice were placed in an observation cage and allowed to acclimate before introducing another new mouse. Social exploration was measured by how much time in contact the two mice spent with each other during a two-minute observation period. The depressed mice showed a significant reduction in social exploration when compared with control mice. Depressed mice displayed social exploration for about 5 seconds, while the control, non-stressed mice spent about 40 seconds socially exploring within the two-minute period.
To determine if the microglial activity can be returned to normal and the depressive-like behaviors eliminated, the researchers administered imipramine, an antidepressant drug. Imipramine was able to:
- Block acute stress-induced microglial activation: The increased microglial activity, the initial acute response to stress, was blocked in the stressed mice. This indicates that antidepressants can affect microglial activity.
- Block chronic stress-induced microglial decline: As a result of imipramine administration, the microglial decline associated with long-term stress exposure was blocked. Thus, the antidepressant can influence microglial activity whether exposure to stress is acute or chronic.
- Inhibit depressive-like behaviors:
- In the Sucrose Preference Test, depressed imipramine-treated mice had the same preference for the sweetened solution as non-depressed untreated controls. Both groups showed approximately 80% sucrose preference, indicating that they were motivated and responsive to the sweet solution’s rewarding taste.
- In the Social Interaction Test, the depressed imipramine-treated mice performed at the level of unstressed controls and averaged about 20 seconds of social exploration, a significant improvement from the baseline of 5 seconds of social exploration they averaged when untreated.
This study showed how microglial activity could be either active or suppressed based on the amount of time that the mouse experiences stress, demonstrating the dynamic nature of microglia. Furthermore, the positive effects of antidepressants on stress-induced depression and microglial activity were profiled.
Cognition and Microglia Signaling in Alzheimer’s Disease
Microglia haven’t just been implicated in depression, they have also been linked to neurodegenerative diseases like Alzheimer’s disease. Since Alzheimer’s disease does not have a definitive cure yet, any new insights on its pathophysiology could lead to a revolutionary breakthrough. That’s why researchers are taking the time to consider the role of microglia in the disease’s etiology.
In a study by Fang et al., the relationship between microglia, Alzheimer’s disease, and cognition was studied by using a transgenic (Tg) mouse model, a model that correlates with the levels of inflammation and mutant amyloid precursor protein (mAPP) as seen in human Alzheimer’s patients.[3] The researchers focused on a particular receptor for advanced glycation end products (RAGE)-dependent signaling in microglia and their role in exacerbating Alzheimer’s related damage and inflammatory responses.
RAGE is a receptor that belongs to the immunoglobulin superfamily that can bind to advanced glycation endproducts and Aβ protein (which are relevant to Alzheimer’s pathogenesis). RAGE expression is naturally low in the central nervous system and most tissues, but increases when the organism is in a diseased state. In Alzheimer’s diseases, as Aβ protein accumulates in the brain, RAGE expression also increases in microglia and neurons.
The researchers crossed overexpressing RAGE C57BL/6 mice with mAPP transgenic Alzheimer’s mice, creating mAPP/RAGE mice. Also, dominant-negative (DN-RAGE) mice were crossed with transgenic Alzheimer’s mice to serve as an additional control group. DN-RAGE mice have blocked RAGE-dependent signaling. Since the signaling is blocked, using this mouse strains enables researchers to establish the effects of RAGE signaling on pathophysiology.
Both RAGE and DN-RAGE mice have normal development, life span, and reproductive fitness. These two mice were compared with non-transgenic controls. The researchers found that:
- mAPP/RAGE mice have increased inflammatory markers: RAGE signaling is increased in diseased states. In mAPP/RAGE mice, higher RAGE activity was linked to higher levels of inflammatory markers when compared with mAPP mice and mAPP/DN-RAGE mice. IL-1β and TNF-α levels significantly increased in the mAPP and mAPP/RAGE groups with the latter having significantly higher levels. By comparison, normal control mice and mAPP/DN-RAGE mice were not significantly different from each other.
- mAPP/RAGE mice have higher levels of Aβ proteins in the hippocampus and cortex: In addition to increased inflammation markers, mAPP/RAGE mice’s levels of Aβ40 and Aβ42 proteins were two times higher in the hippocampus and cortex when compared with mAPP and mAPP/DN-RAGE mice. This means that RAGE signaling is correlated with Aβ protein load.
- mAPP/RAGE mice have impaired spatial and reference memory: When tested in the Water Radial Arm Maze, the mAPP/RAGE performed significantly worse than mAPP mice which, in turn, had more errors than the controls (nonTg and RAGE mice) did. By contrast, mAPP/DN-RAGE mice did not perform differently than their respective controls (NonTg and DN-RAGE mice) indicating that their cognition was at the level of normal mice. However, mAPP mice had significantly more errors than the controls. The number of errors indicates that the mAPP and mAPP/RAGE mice were not able to learn the task and find the escape platform as quickly as the controls did, indicating that they had a problem with their cognition.
This study shows the important role that expressed microglial receptors play in Alzheimer’s pathophysiology. By genetically manipulating mice to study overexpressed microglial RAGE receptors, the researchers established that RAGE signaling is associated with higher levels of disease severity and Aβ protein abundance.
FK506 Reduces Parkinson’s Neuroinflammation
Parkinson’s disease is characterized by dopaminergic neurodegeneration and abnormal accumulation of the α-synuclein protein in the brain.[4]
A study by Van der Perren et al. examined the effects of FK506 on an α-synuclein-based rat model of Parkinsons’ disease. FK506, also known as tacrolimus, has been previously shown to have neuroregenerative and neuroprotective effects in models of Parkinson’s disease. As a drug, FK506 is an immunosuppressive drug that’s given to patients receiving organ transplantation in order to reduce their chances of organ rejection. FK506 acts on the immune system in many different ways and can reduce microglial activation.
The researchers found that:
- Parkinson’s disease mice had higher levels of microglia: The α-synuclein model of Parkinson’s disease is one of the best models to use for studying neuroinflammation and microglial activation in Parkinson’s disease. Microglia levels are elevated in α-synuclein Parkinson’s rats.
- FK506 lowers microglial count in Parkinson’s disease mice: As a result of FK506 administration, the microglial count was significantly lower in FK506-treated Parkinson’s disease rats. This suggests that this anti-inflammatory drug can significantly alter microglial activity in a model of Parkinson’s disease.
- FK506 increased survival of dopaminergic neurons: Furthermore, there was a statistically higher rate of survival of dopaminergic neurons in Parkinson’s disease mice as a result of FK506 treatment. Thus, a correlation exists between the survival of dopaminergic neurons and a decrease in the microglial count, prompted by FK506 treatment.
- Forelimb use may improve with FK506: To study the behavioral and physical effects of FK506 treatment, a Cylinder Test was used. The Cylinder Test is designed to quantify forelimb use by measuring how long it takes for rodents to make 20 steps with their paws. If mice need a long period to complete this task by doing 20 steps, it indicates that they have poor forelimb use. The difference between the controls and the experimentally treated rats on forelimb use was verging on significant. The researchers suggested that future studies should cover a longer observation period.
This study demonstrated how a drug for Parkinson’s disease that has anti-inflammatory properties can ultimately decrease dopaminergic neurodegeneration and show promise for behavioral improvement.
Isobavachalcone Inhibits Microglia and Is Neuroprotective for Parkinson’s
Another study, using a different model of Parkinson’s disease, also shed light that using an anti-inflammatory intervention changes the microglial activity and improves behavioral symptoms.
A study by Jing et al. focused on the effects of isobavachalcone on a 1-methyl-4-phenyl-1,2,3,6- tetrahydropyridine (MPTP)-induced model of Parkinson’s disease. [5]
Isobavachalcone, the major component of Psoralea corylifolia, has been shown to have many functions, including anti-oxidation, immunoregulation, and the ability to regulate Aβ42 proteins,[6] possibly promising for modulating the Aβ42 deposits observed in Alzheimer’s patients. However, the therapeutic effects of isobavachalcone on Parkinson’s disease are still unclear.
The researchers established that:
- MPTP Parkinson’s mice have over-active microglia: Overexpressed microglia are found in the MPTP model, as indicated by the increased microglia-specific Iba-1 markers. Thus, the MPTP model of Parkinson’s disease can also be used to study elevated microglial levels.
- MPTP Parkinson’s mice have poor motor skills: The MPTP-induced Parkinson’s mice had significantly compromised motor skills, as demonstrated by their reduced balance and coordination abilities in the Rotarod. The Parkinson’s mice were able to stay on the apparatus for about 40 seconds while healthy controls were able to stay on the Rotarod for roughly 52 seconds. Thus, the Parkinson’s disease mice had poor motor skills combined with higher microglial levels.
- Isobavachalcone is neuroprotective: As a result of isobavachalcone treatment, neuroinflammation decreased. Iba-1 microglia-specific markers were lowered significantly in the experimental group. The reduction of microglial activity shows that isobavachalcone has anti-inflammatory properties that are beneficial for rodents with Parkinson’s disease.
- Isobavachalcone improves motor skills of Parkinson’s disease mice: As mentioned previously, Parkinson’s disease mice had poorer behavioral scores on the Rotarod when compared to healthy controls. When the diseased mice were given 50 mg/kg of isobavachalcone, their Rotarod performance was significantly better than the non-treated MPTP-diseased Parkinson’s mice. Thus, as a result of the treatment, the Parkinson’s mice were able to stay longer on the Rotarod, indicating that some of their motor disabilities were salvaged due to isobavachalcone administration.
This experiment demonstrated that microglial activation is associated with poorer outcomes in a Parkinson’s disease mouse model since results showed that neuroinflammation was correlated with disease-related behavioral outcomes. The Parkinson’s disease mice had improved behavioral scores on their motor skills assessment as a result of isobavachalcone treatment which also lowered microglial count.
For more information about this area of research, how microglia and inflammation affect behavior, check out our related article: Microglia, Inflammation, and Behavior.
Conclusion
Microglial involvement in disease manifestation is a growing, new area of research that focuses on uncovering the role that microglia play in diseased states such as Alzheimer’s and Parkinson’s diseases.
Future directions of research will continue to expand on what is known about the role microglia play in disease, but will also uncover how specific circumstances influence microglial activity and behavior. For example, recent findings have uncovered that gut microbiota can influence the maturation and function of CNS microglia.[7]
Special interest is being given to the environmental effects on microglia and behavior , as well as the relationship between microglial physiology and behavior. By understanding the range of microglial functions, a full picture can be gathered on how the environment and microglial physiology also contribute to disease manifestation and progress.[8]
Precision medicine is rising in practice since doctors are taking a special interest in subscribing treatment and prevention tailored to an individual’s environment, lifestyle, and genetics. As precision medicine rises in prevalence, microglia will also be taken into account.
References
- Weinberger, A. H., et al. “Trends in depression prevalence in the USA from 2005 to 2015: widening disparities in vulnerable groups.” Psychological medicine 48.8 (2018): 1308-1315.
- Kreisel, T., et al. “Dynamic microglial alterations underlie stress-induced depressive-like behavior and suppressed neurogenesis.” Molecular psychiatry 19.6 (2014): 699.
- Fang, Fang, et al. “RAGE-dependent signaling in microglia contributes to neuroinflammation, Aβ accumulation, and impaired learning/memory in a mouse model of Alzheimer’s disease.” The FASEB Journal 24.4 (2010): 1043-1055.
- Van der Perren, Anke, et al. “FK506 reduces neuroinflammation and dopaminergic neurodegeneration in an α-synuclein-based rat model for Parkinson’s disease.” Neurobiology of aging 36.3 (2015): 1559-1568.
- Jing, Haoran, et al. “Isobavachalcone Attenuates MPTP-Induced Parkinson’s Disease in Mice by Inhibition of Microglial Activation through NF-κB Pathway.” PLoS One 12.1 (2017): e0169560.
- Chen, Xiumin, Yanfang Yang, and Yingtao Zhang. “Isobavachalcone and bavachinin from Psoraleae Fructus modulate Aβ42 aggregation process through different mechanisms in vitro.” FEBS letters 587.18 (2013): 2930-2935.
- Erny, Daniel, et al. “Host microbiota constantly control maturation and function of microglia in the CNS.” Nature neuroscience 18.7 (2015): 965.
- Shemer, Anat, et al. “Microglia plasticity during health and disease: an immunological perspective.” Trends in immunology 36.10 (2015): 614-624.