In this article we will discuss the various autism spectrum disorder (ASD) models used in behavioral neuroscience. We will also take a special look at the behavioral correlates of these models. But, first, let’s begin with an introduction to ASD.
Introduction to Autism
Autism spectrum disorder (ASD) is a heterogenous neurodevelopmental disorder for behavioral deficits that affect approximately 1% of the population.[1] Different ASD patients can be affected in drastically different ways. For example, a subset of high-functioning patients presents enhanced sensory, visual, or cognitive abilities. Considering that autism risk shares some genetic correlations with measures of mental ability, some argue that autism is a disorder of enhanced, but imbalanced components of intelligence.[2]
To be diagnosed with ASD, an individual must show persistent symptoms falling within two behavioral domains:[3]
- Social interaction and communication impairments
- Repetitive and restricted behaviors, interests and activities
Furthermore, ASD patients commonly present a range of comorbidities, including:
- Seizures
- Anxiety
- Aggressive behaviors
- Hyperactivity
- Sensory processing defects
- Impaired motor coordination
In many other human syndromes, autistic-like behavior is used as a clinical symptom. For example, Rett syndrome and Fragile X syndrome are well-defined genetic diseases that present autistic-like traits.[1]
Current treatment for ASD mostly targets specific symptoms such as alleviating hyperactivity with antipsychotic drugs. Behavioral and educational therapies are also the main strategies for improving the communication abilities of ASD patients.[1]
Therefore, there is a pressing need to understand the biological mechanisms underlying autistic behaviors and develop more effective therapeutics. To this end, rodent models of ASD are critical research tools.
ASD Pathophysiology
However, modeling ASD is not so simple. This is because ASD is a multifactorial disorder involving many genetic and environmental factors.
There is very high inter-individual variability in the behavioral deficits that ASD patients exhibit, which is likely a result of different pathophysiology underlying different ASD cases.
For example, it is common to differentiate ASD patients with or without intellectual disability using the terms: Low-functioning autism and High-functioning autism. The high-functioning individuals may still communicate using speech whilst the low-functioning individuals may utilize communication through picture boards.
Genetics
ASD has a strong genetic basis as concordance between monozygotic twins is 90%, compared to 10% for dizygotic twins and siblings.[4]
Large-scale linkage analyses and whole genome association studies have identified an array of candidate genes, copy number variants, and epigenetic factors. The candidate genes are often functionally related, such as:
- Synaptic and dendritic morphology
- Development of neural connections
- Neurotransmission
Among them, the gene ENGRAILED 2 is a notable one that contributes to 40% of the cases of ASD susceptibility.[5]
Environmental Factors
Prenatal, perinatal and postnatal environmental factors have all been implicated in the pathogenesis of ASD.[6] The table below lists environmental factors that have been shown to contribute to ASD based on conception stages:
| Exposure time window | Environmental factors contributing to ASD |
| Prenatal | Exposure to teratogens
Viral infections Maternal anticonvulsants Maternal diabetes Advanced parental age Prenatal stress |
| Perinatal | Low birth weight
Short gestation length Birth asphyxia Perinatal stress |
| Postnatal | Autoimmune disease
Viral infection Hypoxia Mercury toxicity Zinc deficiency |
Table 1: Environmental factors contributing to ASD during different developmental time windows.
In recent years, a growing number of genetic and environmental factors have been added to the pathogenesis of ASD. In addition, researchers are increasingly aware of the complex interactions between genetic and environmental factors.
These interactions may be a main driver of the heterogeneous nature of ASD, as well as a key factor that complicates ASD research.
Rodent models are fundamental preclinical tools for controlling some of the variability and confounding factors. Compared to humans, mice and rats display behaviors that can be easily monitored and clearly interpreted using standardardised behavioral assays. Having said that, it is critical to choose the right research model and arsenal of tools for conducting a behavioral experiment. Furthermore, it is important to be familiar with the factors that can influence the results acquired from behavioral testing. Check out our General Guide to Behavioral Testing in Mice for the major factors that affect testing and plan ahead.
Modelling and Assessing Autistic Behaviors in Rodents
Although studying human diseases in rodent animals is a common scientific practice, it is natural to ask the question: How are human behaviors replicated and assessed in a species that looks so different to us?
In fact, for scientists to reap translational value from animal research, we first need to establish appropriate behavioral correlates and assays in rodents.
In this section, we will take a look at a few behavioral correlates and assays used in ASD behavioral research.
- Assays for Social Interaction
- Assays for Communication
- Assays for Repetitive Behaviour
These three categories of behavioral assays were developed in parallel with the traditional diagnostic criteria for ASD which required the presence of abnormal social interactions, communication deficits and repetitive behaviors.[4]
Assays for Social Interaction
Like humans, rodents are social animals that display a number of social behaviors such as reciprocal interactions, parenting, play and aggressive behaviors. Thus, many rodent behavioral assays have been designed to maximize relevance to the social deficits in ASD patients.
Reciprocal Social Interactions
When mice are placed together in a confined space, they engage in reciprocal social interactions between pairs or among groups. These interactions can be evaluated in the forms of:[4]
- Nose-to-nose sniffing, nose-to-anogenital sniffing
- Crawling over and under each other
- Pushing over each other
- Chasing
- Front approach and following
- Wrestling
To assess whether mice show autistic-like social interaction deficits, automated video tracking systems are often used to score the number and duration of interactions.
Social Approach
Sociability is commonly tested with a 3-chambered social approach (or Sociability Chamber) in mice.[4] The apparatus consists of a central space and two chambers. One of the chambers includes an unfamiliar mouse inside a wire cup whilst the other chamber contains a novel object.
Sociability is defined as the time that the subject mouse spends in the chamber containing another mouse than in the chamber containing the novel object. A healthy subject mouse should prefer to spend time with the other mouse rather than the non-social object.
Partition Test
The partition task is another simple assay for assessing sociability in mice.[4] It uses a standard cage divided by a perforated partition to separate a subject mouse from a cage mate. The subject mouse can receive visual, auditory, and olfactory stimuli from the cage mate, but physical interaction is blocked. The time spent near the partition is a measure of social interest, which is often reduced in ASD patients.
Social Preference Tests
Since rodents are social and novelty-seeking creatures, a subject mouse should display social recognition and spend greater time with the more novel (less familiar) rodent than a familiar rodent when presented with two partner choices.[4] Equipment used for social preference tasks include the three-chambered apparatus, the partition test apparatus and Y-maze. The rodent’s degree of sociability is scored according to the amount of time it spends with the more novel mouse.
Social Transmission of Food Preference
Interaction with a cage mate that has eaten a novel food can make the subject mouse familiar with the scent. As a result, the subject mouse should eat more of this food compared to a less familiar type of food. Since familiarity is gained through and close physical contact, it reflects the subject mouse’s social interaction abilities.[7] Thus, social transmission of food preference is another way of assessing social impairments in mouse models.
Assays for Communication
Communication deficits are another hallmark of autism. Modeling communication is more problematic because humans primarily communicate through speech, and rodents communicate through olfactory pheromones. Rodents also emit vocalizations in the ultrasonic range, which are thought to assist communication. Thus, communication paradigm tests generally consist of scoring the olfactory or auditory cues emitted by rodents as well as how they respond to receiving such cues.
Urinary Pheromones
Mice show high levels of interest in urinary scents from other mice.[7] They also leave urinary pheromones as territory marks. Counting the numbers of scent marks, sniffing bouts and duration in proximity to olfactory cues are ways to score olfactory communication.
Ultrasonic Vocalizations
Complex ultrasonic vocalizations are emitted by mice in different social situations, which can be picked up by sensitive ultrasonic microphones, headphones and advanced software. Mouse pups have been found to use ultrasonic vocalization to find their mothers. This type of communication probably plays a more important role in juvenile and maternal behaviors of rodents. Significant disturbances of ultrasonic vocalization are observed in ASD models such as Shank1 knockout mice and BTBR inbred males.[8]
Assays for Repetitive Behaviours
The second criteria for ASD diagnosis includes presence of motor stereotypies and repetitive behaviors. In human patients, these are usually presented as hand flapping and toe walking. in mice are usually in the forms of repetitive circling, jumping and grooming, as well as restricted exploration of an open field.[4] Scoring of stereotypies is mostly done by an investigator observing videotaped mouse behaviors and recording durations of repetitive behaviors.
Repetitive self-grooming and marble burying
Repetitive self-grooming and marble burying in mice are specific examples of stereotypies that can model particular ASD human behaviors like assembling the same puzzle or playing one game repeatedly in ASD patients. These repetitive behaviours have been observed in a wide range of ASD models, demonstrating the face validity (parallel phenotypes) of modeling this characteristic behavior of ASD in mice.[7]
Cognitive Rigidity
Another symptom of ASD that often accompanies repetitive behaviors is cognitive rigidity. It is defined as the inability to change behaviors when they are ineffective for achieving a goal. Normally, people or rodents are cognitively flexible when they are in a new situation that requires changes in their approach to a problem.
In mice, cognitive functions can be assessed using classic tests like the T-Maze and Morris Water Maze. But in studying ASD, the animal is tested on its ability of reverse learning in mazes as an indication for cognitive rigidity. Examples are:
- Finding a food reward in the opposite arm of the T-Maze from where it first learned to find the reward
- Relocating the hidden escape platform in a Morris Water Maze
An ASD mouse model is typically capable of initial learning but fails on reversal due to high cognitive rigidity.
Restricted Interests
Despite obsessive persistence in certain behaviors, most ASD patients show restricted interests in general. One method to measure restricted interests in rodents is the Open Field exploration test.[7]
For example, novel objects could be placed throughout a novel Open Field and mice are expected to sniff around and nose poke into holes at various places. Restricted interests could be reflected in perseverative exploration of very few objects or holes.
Summary of Behavioral ASD Correlates
With a rich set of classical and ASD-specific behavioral assays, researchers have the means to validate mouse models and study ASD in a comprehensive way.
- Behavioral phenotyping in animal models uses the three main criteria for diagnosing ASD: social interaction, communication, and repetitive behaviors.
- Although ASD-related behavioral correlates take different forms in humans and rodents, we can assess their patterns and durations to detect similar abnormalities.
- The more abnormalities of behavioral ASD correlates shown in an animal model, the more face validity it has for modeling human ASD.
Additional Behavioral ASD Deviations
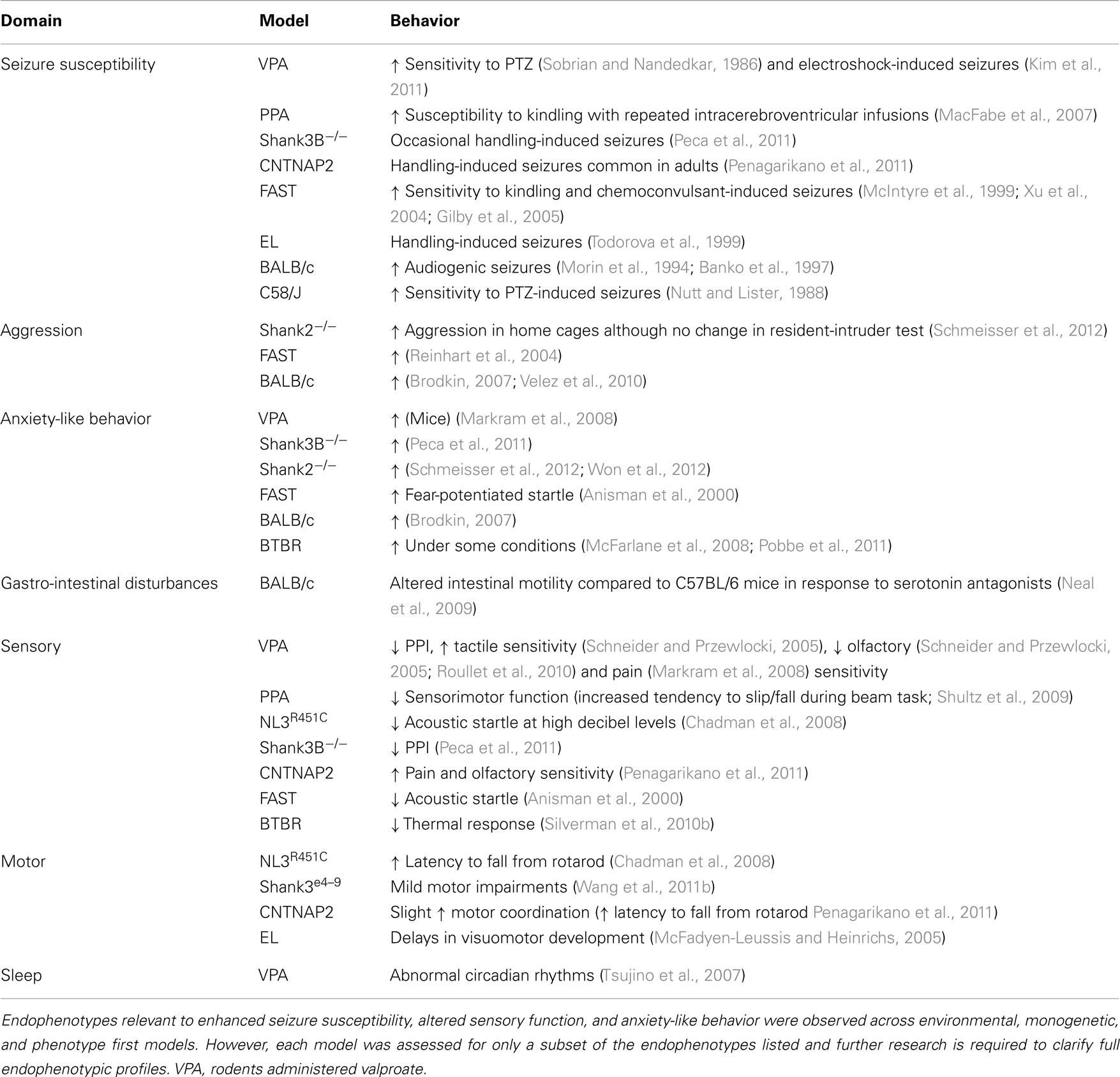
Apart from the main diagnostic behaviors of ASD described above, common ASD models also demonstrate deviations in other behavioral correlates of ASD (Figure 1).[9]
When many animal models can demonstrate core ASD symptoms, their ability to also present lesser known ASD-like symptoms become important to researchers in the process of choosing a model.
Mouse Models of Autism
A wide repertoire of mouse models of ASD have been developed and demonstrated face validity by showing the mouse counterparts of autistic-like behaviors.
In this section, we will take a look at the most commonly used ASD genetic and environment models.
Genetic Models
BTBR Inbred Model
The BTBR strain is the most extensively characterized inbred strain for replicating autistic-like behaviors.[7] It displays many relevant behavioral deficits analogous to autistic symptoms including:
- Reduced sociability
- Repetitive behaviors
- High levels of obsessive behaviors like self-grooming and marble burying
- Altered vocalizations
- Impaired social transmission of food preference
A common problem with using mouse models to study behaviors is the failure to replicate behavioral changes across laboratories. But the BTBR inbred strain has consistently shown the same social deficits. This consistency reinforces the confidence in using BTBR mice as a reliable ASD model.[10]
Despite having a promising face validity, researchers have also raised concerns about oddities about the BTBR model. For example, BTBR mice lack the corpus callosum,[11] an important brain structure that forms the main connection between the two brain hemispheres. It is unclear whether alterations in the corpus callosum contribute to the strain’s behavioral defects.
Nevertheless, a recent study re-evaluated additional non-diagnostic (lesser known) autistic symptoms and concluded that they are also similarly impaired (Table 2).[12]
| Behavioral test | Description | Results |
| Balance Beam Task | Mice need to walk across a narrow ‘walking bridge’, which reflects sensorineural balance and coordination. | Poor sensorimotor integration |
| Open Field task | The task exploits an innate fear to explore brightly lit open spaces to assess exploratory behavior and anxiety | Hyperactivity and impulsiveness |
| Morris Water Maze | This classic task tests spatial learning and memory based on the an animal’s innate desire to escape a stressful situation | Impaired spatial learning |
Table 2: Re-evaluation of non-diagnostic autistic symptoms in the BTBR mouse model using behavioral tests.
Therefore, these results further validated the translational value of BTBR mice in ASD research.
Transgenic Models with Single Gene Mutations
It is estimated that 25% of human ASD cases are caused by genetic alterations.[13] To date, a vast and heterogenous list of genes have been associated with ASD. Consequently, a battery of genetic knockout models has been generated. Some well-known gene targets are:[13]
- Fmr1: The gene underlying fragile X syndrome and the most common inheritable cause of ASD.
- Mecp2: The disease-causing gene for Rett syndrome, which also presents autistic-like symptoms.
- Tsc: A leading genetic cause of syndromic autism, in which 40-50% of people with Tsc mutations has ASD
- Cntnap2: A strong risk factor for ASD and other neurodevelopmental disorders based on neurobiological, genetic and imaging data.
- Nlgn: Mutations in neuroligins Nlgn3 and Ngln4 affect synaptogenesis and may predispose to ASD.
- Shank: Shank mutations disrupt proteins that act as molecular scaffolds in the postsynaptic density and are associated with ASD.
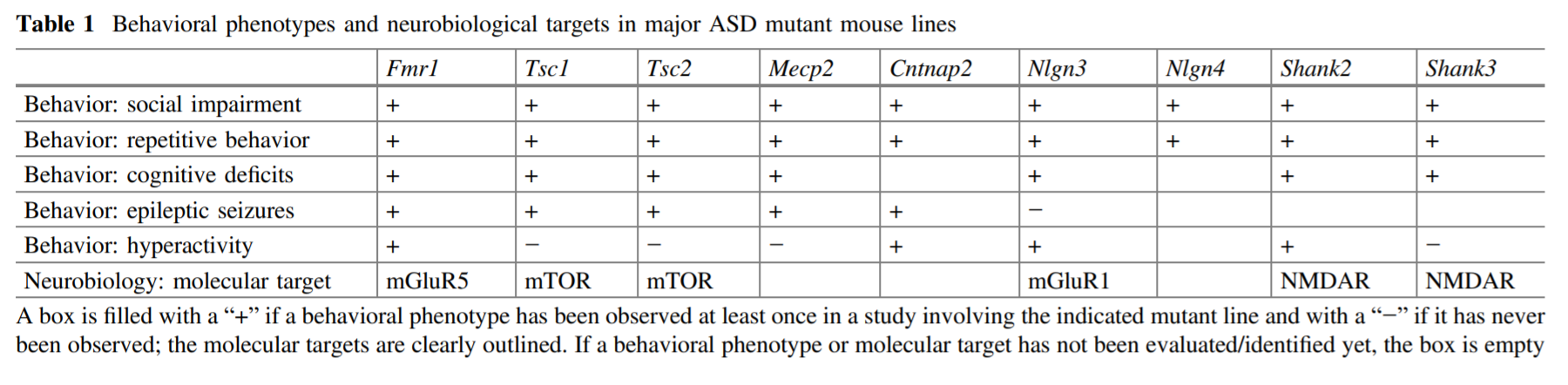
Table 2 summarizes the types of behavioral defects shown in these genetic knockout models[13]
Table 2: Genetic knockout models based on six ASD-associated genes demonstrate autistic-like symptoms.
According to the guideline for ASD diagnosis, the two core symptoms are impaired social interaction and repetitive behavior.[2] As can be seen from the table, all nine of the transgenic models can replicate these core symptoms, along with various other associated symptoms. These results establish a strong face validity for genetic mutant models.
Targeting candidate ASD genes in transgenic mice also establish their construct validity as they potentially replicate, at least in part, the same biological origin of ASD. These models are particularly helpful in addressing questions such as:
- What are the neural circuits responsible for the phenotypes in each mutant model?
- What are the common neurobiological pathways and molecular mechanisms among different mutant models?
- Are there vulnerable time windows when treatment is promising?
Lastly, it is worth noting the general difficulty of estimating the predictive validity of ASD models due to lack of effective treatment options for patients.
Most of the transgenic models are yet to be tested with pharmacological substances which are currently in clinical trials. But, the Cntnap2 model has shown predictive validity since its behavioral deficits are reversed with the prescribed ASD antipsychotic risperidone.[9]
Environmental Models
Apart from inbred and transgenic mouse models, environmentally induced ASD models are also valuable tools as there are many “idiopathic” cases that cannot be linked to any genetic causes. These are likely attributed to adverse environmental factors during prenatal, perinatal or postnatal development.
Valproic Acid (VPA)-Induced Model
VPA is the most studied environmental agent linked to autism.[14] It is an anticonvulsant and mood-stabilizing medication used to treat epilepsy and depression. In pregnant women who used the drug, their offspring had a significantly increased risk for ASD. Thus, VPA-induced ASD models replicate the causes of at least some ASD patients, establishing their construct validity.
In mice, exposure to VPA during gestation can be introduced via intraperitoneal injections or orally with food.[15] The exposure leads to repetitive behaviors and deficits in social interaction that are characteristic of ASD, demonstrating promising face validity.
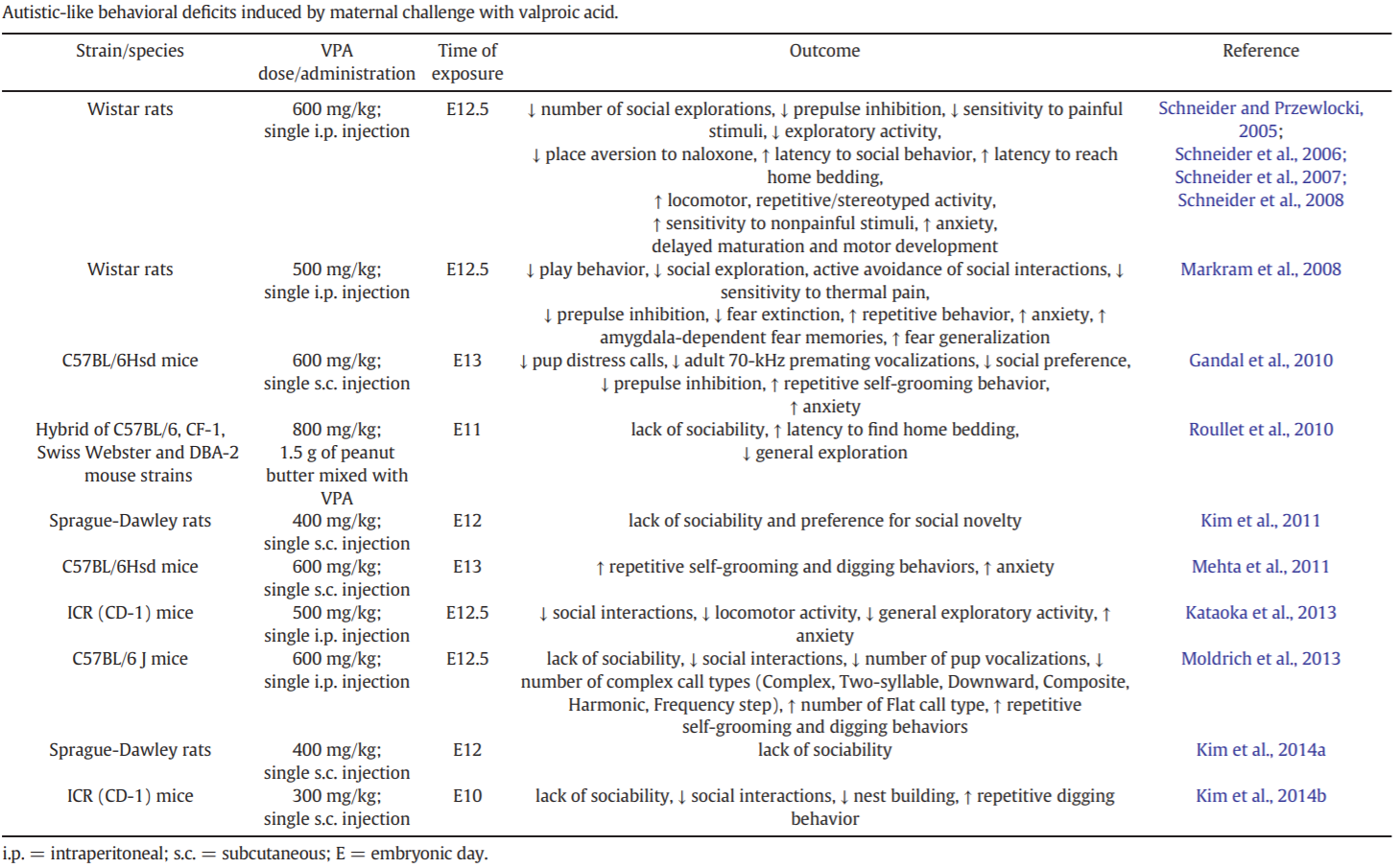
Table 3 lists the different rodent strains that have been treated with VPA, as well as their resemblance to the core and comorbid symptoms of autism.[15]
Table 3: Autistic-like behavioral deficits induced by maternal challenge with VPA.
The biggest difference between environmental models and genetic models is that the underlying etiology of environmental models is often elusive. The neurobiological processes that happen between the exposure and developing symptoms are much less straightforward compared to single-gene transgenic models. However, since ASD is not a single gene disorder in reality, genetic models are not necessarily the best for studying ASD.
Apart from construct and face validity, the VPA model has also shown some predictive validity. Remarkably, environmental enrichment on postnatal behavior resembles the success of early behavioral intervention and sensorimotor enrichment in autistic children.[15] Pharmacological treatments such as a mGluR5 inhibitor have also been shown to improve symptoms.[15]
Overall, the VPA model seems to be robust for modelling ASD and may better represent idiopathic cases that arise from environmental or epigenetic origins. Thus, it has been the most widely used environmental ASD model. Nevertheless, a few other environmental agents have been used.
Propionic acid
Propionic acid (PPA) is metabolite in the gut found to be elevated in ASD.[16] Thus, intracerebroventricular injections of PPA in rats were used as a model for ASD. Studies using this model found various behavioral and neuropathological changes associated with ASD, suggesting a possible link between diet, metabolism and ASD.
Maternal viral infection
Viral infection during pregnancy increases the risk of offspring developing ASD. This observation is modeled in rodents by infecting pregnant mice with a virus such as influenza or injecting synthetic double-stranded RNA.[17] These models led to the discovery that maternal immune activation elevates cytokines in the fetal environment, which induce abnormal ASD-like phenotypes.[17][18]
Summary of ASD Models
With a variety of ASD models to choose from, researchers have the means to study how:
- ASD can arise due to genetic and environmental factors, as well as their interactions
- Specific genes can affect brain development, and whether they can be targeted as therapeutics
- Environmental exposures during pregnancy and development can increase the risk of ASD, and whether they can be avoided
Conclusion
In conclusion, despite higher order cognitive deficits associated with ASD, behavioral correlates and assays exist in animal models. They are useful tools in studying the mechanisms of ASD and in attempts to find treatments.
Since ASD is a highly heterogeneous and multifaceted disorder, various genetic and environmental models have been generated to cover a wide spectrum of causes. Most models discussed here are capable of replicating the core symptoms of ASD, while also covering some other associated deficits.
To score behaviors in these models, both traditional and specially-developed assays are used. Overall, given the many promising models and armed with tools for behavioral assessment, we can expect ongoing breakthroughs in ASD research including the expansion into pharmaceutical testing.
References
- Ornoy, A., Weinstein-Fudim, L. & Ergaz, Z. Prevention or Amelioration of Autism-Like Symptoms in Animal Models: Will it Bring Us Closer to Treating Human ASD? Int J Mol Sci 20, doi:10.3390/ijms20051074 (2019).
- Crespi, B. J. Autism As a Disorder of High Intelligence. Front Neurosci 10, 300, doi:10.3389/fnins.2016.00300 (2016).
- Gnanavel, S. & Robert, R. S. Diagnostic and statistical manual of mental disorders, fifth edition, and the impact of events scale-revised. Chest 144, 1974, doi:10.1378/chest.13-1691 (2013).
- Silverman, J. L., Yang, M., Lord, C. & Crawley, J. N. Behavioural phenotyping assays for mouse models of autism. Nat Rev Neurosci 11, 490-502, doi:10.1038/nrn2851 (2010).
- Gharani, N., Benayed, R., Mancuso, V., Brzustowicz, L. M. & Millonig, J. H. Association of the homeobox transcription factor, ENGRAILED 2, 3, with autism spectrum disorder. Mol Psychiatry 9, 474-484, doi:10.1038/sj.mp.4001498 (2004).
- Park, H. R. et al. A Short Review on the Current Understanding of Autism Spectrum Disorders. Exp Neurobiol 25, 1-13, doi:10.5607/en.2016.25.1.1 (2016).
- Kazdoba, T. M. et al. Translational Mouse Models of Autism: Advancing Toward Pharmacological Therapeutics. Curr Top Behav Neurosci 28, 1-52, doi:10.1007/7854_2015_5003 (2016).
- Hrabovska, S. V. S., Y.T. Animal Models of Autism Spectrum Disorders and Behavioral Techniques of their Examination. Neurophysiology 48, doi:DOI 10.1007/s11062-017-9613-2 (2016).
- Argyropoulos, A., Gilby, K. L. & Hill-Yardin, E. L. Studying autism in rodent models: reconciling endophenotypes with comorbidities. Front Hum Neurosci 7, 417, doi:10.3389/fnhum.2013.00417 (2013).
- Yang, M., Zhodzishsky, V. & Crawley, J. N. Social deficits in BTBR T+tf/J mice are unchanged by cross-fostering with C57BL/6J mothers. Int J Dev Neurosci 25, 515-521, doi:10.1016/j.ijdevneu.2007.09.008 (2007).
- Wahlsten, D., Metten, P. & Crabbe, J. C. Survey of 21 inbred mouse strains in two laboratories reveals that BTBR T/+ tf/tf has severely reduced hippocampal commissure and absent corpus callosum. Brain Res 971, 47-54, doi:10.1016/s0006-8993(03)02354-0 (2003).
- Faraji, J., Karimi, M., Lawrence, C., Mohajerani, M. H. & Metz, G. A. S. Non-diagnostic symptoms in a mouse model of autism in relation to neuroanatomy: the BTBR strain reinvestigated. Transl Psychiatry 8, 234, doi:10.1038/s41398-018-0280-x (2018).
- Schroeder, J. C., Reim, D., Boeckers, T. M. & Schmeisser, M. J. Genetic Animal Models for Autism Spectrum Disorder. Curr Top Behav Neurosci 30, 311-324, doi:10.1007/7854_2015_407 (2017).
- Gadad, B. S., Hewitson, L., Young, K. A. & German, D. C. Neuropathology and animal models of autism: genetic and environmental factors. Autism Res Treat 2013, 731935, doi:10.1155/2013/731935 (2013).
- Nicolini, C. & Fahnestock, M. The valproic acid-induced rodent model of autism. Exp Neurol 299, 217-227, doi:10.1016/j.expneurol.2017.04.017 (2018).
- Shultz S.R., MacFabe D.F. Propionic Acid Animal Model of Autism. In: Patel V., Preedy V., Martin C. (eds) Comprehensive Guide to Autism. Springer, New York, NY (2014).
- Patterson, P. H. Immune involvement in schizophrenia and autism: Etiology, pathology and animal models. Behavioural Brain Research, 204(2), 313–321. https://doi.org/10.1016/j.bbr.2008.12.016 (2009).
- Choi, Gloria B, Yim, Yeong S, Wong, Helen, Kim, Sangdoo, Kim, Hyunju, Kim, Sangwon V, … Huh, Jun R. The maternal interleukin-17a pathway in mice promotes autism-like phenotypes in offspring. Science (New York, N.Y.), 351(6276), 933–939. https://doi.org/10.1126/science.aad0314 (2016).