Introduction
What is Drug Addiction?
Despite the various mechanisms of action of the many abusable drugs, drug addiction looks largely the same from a behavioral standpoint. Drug addiction is a chronic, relapsing brain disease characterized by compulsive drug use despite the negative consequences of intake. It is produced by the adaptive changes in the brain as a result of the chronic exposure of the drug. These brain-wide changes promote repeated consumption of the drug such that the sufferer loses control and compulsively approaches the awarding effects of the drug or avoids the negative effects of the absence of the drug depending on the stage of addiction cycle they are in.
Human Symptomatology of Drug Addiction
Drug addiction is characterized by a variety of behavioral symptoms in humans. According to the DSM V criteria of drug addiction, the set of symptoms associated with drug addiction in the human condition include:
- The need for an increased quantity of the substance to achieve its intoxicating or desired effects or the experience of a diminished effect with the continual use of the same quantity. Key behavioral parameter: tolerance.
- The presence of withdrawal syndrome or the use of a substance to relieve or avoid withdrawal symptoms. Key behavioral parameters: depression, anxiety, and anhedonia.
- Persistent desire to use a substance and/or unsuccessful attempts to eliminate the use of the substance. Key behavioral parameters: drug craving/seeking, relapse.
- The prolonged use or the use of large quantities of the substance. Key behavioral parameters: impulsivity, compulsivity, loss of control of substance use, escalation of drug intake.
- Reduced or absent participation in important social, occupational, or recreational activities because of substance use. Key behavioral parameters: impulsivity, compulsivity.
- A large amount of time spent in activities necessary to obtain, use, or recover from the effects of substance use. Key behavioral parameters: impulsivity, compulsivity, habit
- Continual substance use despite negative physical and psychological consequences that are likely caused or exacerbated by the use of the substance. Key behavioral parameters: compulsivity, resistance to adverse consequences of drug intake.
While drug addiction cannot be modeled in whole within the laboratory due to its complexity, many analogous rodent models have been developed that incorporate major elements of addiction in the form of key behavioral parameters, including compulsive drug seeking, drug craving, physical dependence, relapse, loss of control/impulsivity, and vulnerability to drug use.
Research Techniques for Studying Addiction
Due to the controversial nature of the issue, addiction research has faced many obstacles over the decades. Current techniques strive to be simple enough so that internal validity and reproducibility are maximized, but also holistic enough so that translational potential to human situations is preserved.[1]
In preclinical studies, unlike in clinical studies, variables such as behavior before and after drug exposure, environmental context, dose, and frequency of use can be controlled easily, allowing for high-powered, high face validity studies that can later inform future pharmacological and behavioral treatment options.
Mouse models used to study addiction address either a specific symptom, such as a specific neurobiological or psychological feature like compulsivity, or address several symptoms of the pathology as described in humans (that is, as defined in the DSM V or ICD 10). The first type is especially helpful for delineating the neurobiological mechanisms underlying the disease, while the second type is helpful in testing pharmacological treatments or conducting longitudinal behavior studies where multiple symptoms are potentially interacting. Overall, rodent models of drug addiction can help clarify
- The mechanisms at the molecular level by which drug use becomes compulsive drug abuse
- The mechanisms underpinning the risk of long-term relapse
- The mechanisms in which behavior and motivation become controlled by the abused drug, or cues associated with it, despite negative consequences of use
Drug-induced reinforcement can be assessed with classical conditioning-based paradigms such as conditioned place preference as well as operant or non-operant self-administration methods. In all cases, data generalizability and robustness are key features that must be preserved to ensure high effect sizes and insightful translational results to inform addiction treatment and understanding in humans.
Mechanisms Underlying Drug Addiction
The addiction process is characterized by the drug’s selective action on the reward circuitry in both humans and other mammals. This is largely understood as the mesolimbic pathway, which involves the projections from the midbrain Ventral Tegmental Area (VTA) to the forebrain, including the Nucleus Accumbens (NA) in the ventral striatum and Prefrontal Cortex (PFC).
This pathway is composed of a network of dopaminergic neurons which respond when stimuli indicative of a reward are present. The release of dopamine from the mesolimbic pathway regulates motivation and desire for rewarding stimuli and facilitates reinforcement and reward-related motor function learning, as well as the subjective perception of pleasure. Drug acquisition can thus be blocked by dopamine antagonists, which disrupt this pathway, or through direct lesioning of this pathway. Similarly, downregulation of dopamine receptors in the mesolimbic pathway has been implicated with decreased sensitivity to the reinforcing effects of drug use.[2]
The NA is associated with the initiation and elicitation of conditioned behaviors. It has been found to be involved in the increased sensitivity to drug use as the addiction process progresses. Molecular studies have shown that overexpression of the ΔFosB protein in the NA is a crucial modulator of positively reinforced behavior, and so underlies the development of all forms of drug addiction. Moreover, drug-seeking behavior has been shown to be induced by glutamatergic projections from PFC to NA and dorsolateral striatum. This behavior can be eliminated by the inhibition of AMPA glutamate receptors and glutamate release in the NA. [3]
The PFC is also an important area in drug addiction because it is implicated in the integration of information to help determine whether a reinforced behavior will be elicited. It is thought to be important for forming associations between the rewarding effects of the substance and cues in the environment. As seen in the incubation for drug-craving and reinstatement rodent models, cues are strong modulators of drug-seeking behavior and can facilitate drug relapse months or years after abstinence.
Classical Conditioning Paradigm
Conditioned Place Preference: Modeling Drug-Seeking and Drug-Avoiding Behavior
Conditioned Place Preference (CPP) is a model helpful in understanding the psychological and neurobiological mechanisms that underlie drug addiction. In CPP, the experimenter associates the rewarding effect of the drug with a particular compartment in a box, as seen here. The mouse is free to roam both compartments in the first phase. In the second phase, the drug is injected repeatedly over days in one compartment, allowing an association to develop between the drug effects and the compartment, while a control solution such as saline is injected in the other compartment. CPP is tested when the animal has access to both compartments again, who will then be motivated to move towards the compartment they have associated with the rewarding effects of the drug. The behavioral correlate of the rewarding properties of the drug will be evident then as measured by time spent in the chamber as a function of drug dosage. If a particular compartment was associated with a drug with negative effects, the model can be useful to assess withdrawal effects by the development of conditioned place aversion. In summary, CPP is useful in determining if a drug creates reward/aversion as indicated by its ability to create approach/avoidance behaviors.
This model has good construct validity, as otherwise neutral reinforcers such as environmental context play an important role in drug-seeking behavior in human addicts as well. It’s important to note CPP is well-suited for mice in the context of alcohol research, as rats commonly show indifference in this case. The model is particularly adept at classifying drugs according to their emotional valency but less well-suited to classify drugs according to their potency. Being a passive contextual Pavlovian model, it can’t by itself account for the active drug-seeking and drug-taking behavior demonstrated in both rodents and humans, which is better modeled through self-administration studies.
Self-Administration Paradigms
Operant Self-Administration: An Overview
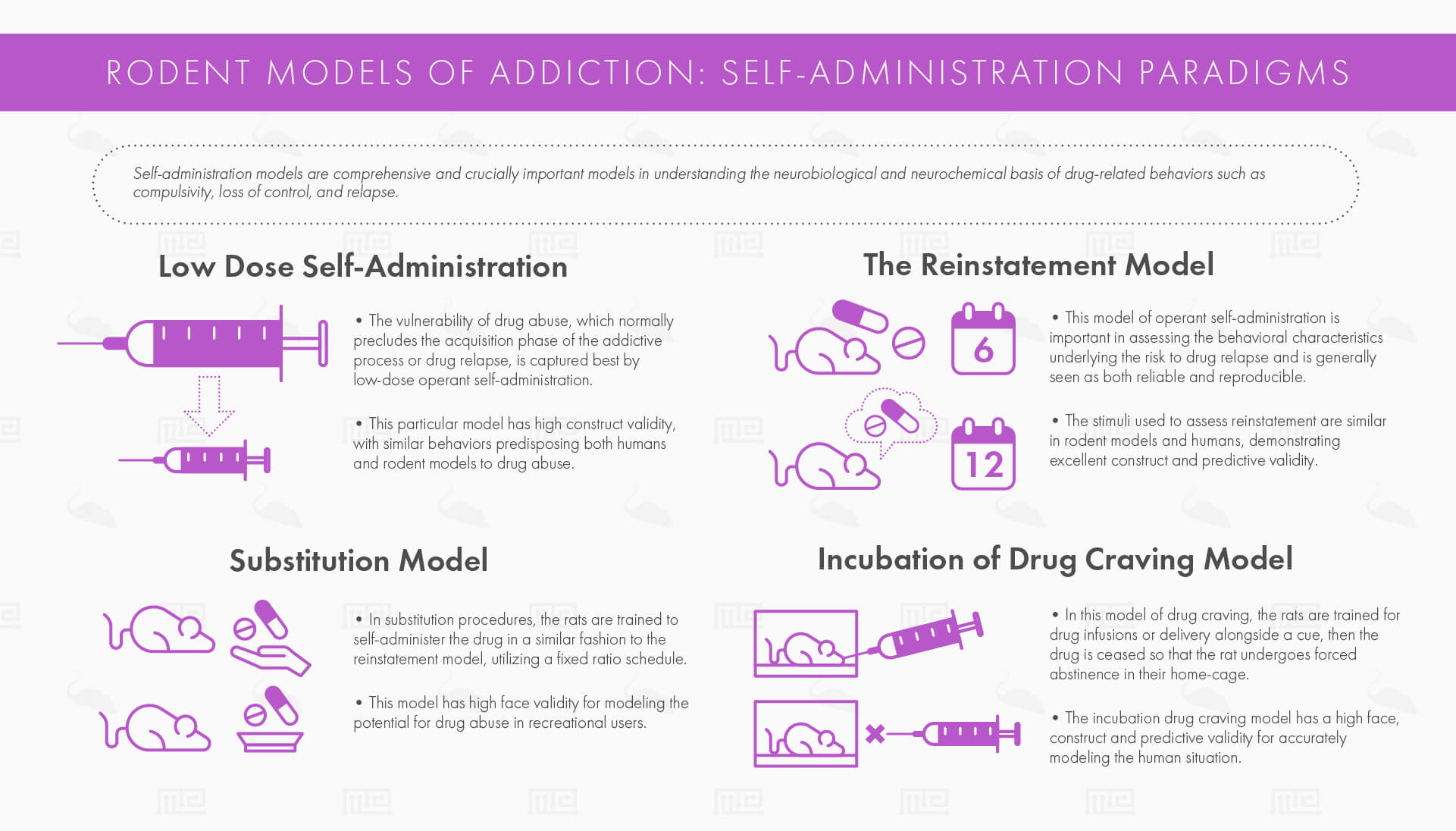
Self-administration models are comprehensive and crucially important models in understanding the neurobiological and neurochemical basis of drug-related behaviors such as compulsivity, loss of control, and relapse. The concept of reinforcement is central to the self-administration model. Reinforcement, broadly speaking, refers to the idea that behavior is controlled by its consequences. A behavior may be strengthened by the result of that behavior, as is the case when the reward circuitry of the brain is activated under the influence of an addictive drug.
Reinforcement then, in utilizing the reward pathway, amounts to patterns of behavior that promote further drug consumption and seeking relative to a control. Many schedules can be used to assess different behavioral parameters. A fixed-ratio schedule, where the drug delivery occurs every time after a fixed number of responses, is particularly useful in understanding the transition from controlled drug use to compulsive and uncontrolled drug use and is usually administered in unlimited access or in discrete trials during the day/night diurnal cycle conditions. For instance, in the FR1 schedule, which is useful in assessing the development of addictive behavior, a single active-lever response delivers a small, previously defined quantity of a drug. In a progressive-ratio schedule, the requirements for obtaining the reinforcer increases progressively, usually in a predefined arithmetic manner. The progression may occur after each reinforcement is delivered, such that each administration of the drug requires more lever presses (in the case of rats) or nose pokes (in the case of mice) until the animal stops responding. This schedule is useful in noting the animal’s motivational status for the drug. The maximum lever presses until non-responding are known as the “break point”, which can be compared across drugs to assess the reinforcement efficacy of the drug.[3]
Generally speaking, the model is created by training rats to self-administer an abusable drug (such as cocaine, nicotine, or alcohol) by lever-pressing or nose-poking in an operant chamber, usually after learning the behavior on a training reward such as water or food. The drug, in this case, is delivered intravenously from an implanted intravenous catheter, leading to a near-immediate CNS reaction. Once trained, the rats are given a testing drug to assess drug abuse liability. The model has excellent face validity, as a lever press is directly indicative of the reinforced behavior through unambiguous rewarding effects. Furthermore, clearly specified variables are tested for in operant self-administration studies, such as the dose-response relationship (# of doses/hour versus unit dose of the drug). Construct validity and predictive validity are also high, as the translational potential to human situations is great. In general, abused drugs by humans are self-administered by rodents, and drugs that are not abused are not self-administered by rodents, showing that addictive behavior is common across mammalian species.[2]
Low Dose Self-Administration: Modeling Vulnerability to Drug Use
Vulnerability to drug use has many behavioral correlates in humans, such as anxiety, impulsivity, and novelty-seeking. This feature of drug abuse, which normally precludes the acquisition phase of the addictive process or drug relapse, is captured best by low-dose operant self-administration. In this case, the study is designed such that one can identify which animals can detect the reinforcing effects of the low dose of the drug. This is heavily influenced by multiple factors such as feeding conditions, drug use history, sex, strain, sweet preference, and reactivity to novelty. The behavioral correlates of vulnerability to drug use can be modeled using procedures like the elevated plus maze, where variables like time spent in the open and closed arms and self-grooming can be indicative of anxious behavior. Impulsivity can be modeled in several ways as outlined in this article, which can inform preclinical study design, as impulsivity has been heavily implicated in the behavioral shift to compulsive drug taking.[3] Thus, this particular model has high construct validity, with similar behaviors predisposing both humans and rodent models to drug abuse.
The Reinstatement Model: Modeling Drug Craving and Relapse
The reinstatement model is useful in assessing factors that may contribute to the risk of drug relapse. In this model of operant self-administration, the rats are trained and then the behavior is made extinct by stopping drug delivery. The rats are then tested under several stimuli without the drug’s reinforcement to see if they reinstate drug-seeking behavior, such as repeated non-reinforced operant responses of either lever presses or nose pokes depending on the animal used. These stimuli include drug priming with small doses of the drug, as well as drug-free conditions like presenting stressful cues and other conditioned stimuli. This model is important in assessing the behavioral characteristics underlying the risk to drug relapse and is generally seen as both reliable and reproducible. The stimuli used to assess reinstatement are similar in rodent models and humans, demonstrating excellent construct and predictive validity. However, the abstinence period made through extinguishing the reinforcement does not accurately mimic the human situation, who generally don’t go through an extinction phase. Moreover, the model does not effectively model the negative consequences of drug use. Since the abused drug is not actually resumed in full, the model better captures drug craving and cue reactivity than drug relapse as seen in human users.
Substitution Model: Modeling Physical Dependence
In substitution procedures, the rats are trained to self-administer the drug in a similar fashion to the reinstatement model, utilizing a fixed ratio schedule. After the rat is trained with a limited-access training drug (a positive control), the training drug is substituted for various new testing drugs. If the training drug is responded to above the control drug’s response threshold, then this drug is noted as a positive reinforcer. Substitution models are useful to assess physical dependence and withdrawal. Once dependence is made on the training drug, a novel testing drug can be introduced to see if dependence still continues. In a similar study design, the training drug is substituted for an antagonist that produces withdrawal symptoms, then a novel testing drug is introduced to see if this drug can reverse the withdrawal symptoms. In this way, drugs with proposed similar mechanisms of action (such as the various opioids or GABA-agonists like benzodiazepines) can be substituted for each other and then the animal can be studied for symptoms of physical dependence and/or withdrawal. This model has high face validity for modeling the potential for drug abuse in recreational users, but its predictive validity is influenced heavily by the animal’s history of drug use prior to receiving the testing drug.
Incubation of Drug Craving Model: Modeling Drug Craving
In human users, environmental stimuli such as friendships, music, location, and the presence of other drugs can contribute to drug craving even after long periods of abstinence, and this craving has been shown to increase over time. In rats, cocaine seeking elicited by cues has been shown to increase during the first couple of months after abstinence. In the incubation model of drug craving, the rats are trained for drug infusions or delivery alongside a cue, then the drug is ceased so that the rat undergoes forced abstinence in their home-cage. Then in the testing condition, drug-associated cues (for instance, a light flash or tone cue that accompanied the cocaine administration) are presented at different days after abstinence, causing non-reinforced responding of either a nose poke or a lever press, indicative of increased drug craving. The incubation drug craving model has a high face, construct and predictive validity for accurately modeling the human situation. The model’s results can be accounted for most probably by neuroplastic changes in the mesolimbic reward system in both humans and rodents. One weakness of the model is that it does not capture the negative consequences of drug use, only increased the craving for the drug after a period of abstinence or withdrawal.
Non-Operant Self-Administration: Modeling Addiction and Uncontrolled Drug Use
This model is helpful in elucidating the transition from controlled drug use to uncontrolled drug abuse and distinguishing between addicted and non-addicted animals. Non-operant versions of self-administration are common in alcohol and caffeine research, where the amount of alcohol or caffeine consumed, the preference of concentration, and the total intake is measured. Both are known to be relatively weak reinforcers, so long-term voluntary exposure to the drug is needed to create compulsive behavior during relapse. In these models, a free-choice oral route of administration is used, with one bottle in the home cage containing water, and another bottle containing an aqueous solution of alcohol. Oftentimes, ascending concentrations of alcohol are used to train the rats or inclusion of a sweetener like sucrose will be used to make higher concentrations more tolerable. Irregular circadian drinking rhythmicity or voluntarily drinking alcohol with an added bitterant such as quinine are both behavioral measures of compulsivity. Additionally, in long-term alcohol drinking rats deprived of alcohol for several weeks, re-exposure leads to increased drinking frequency, especially of higher concentration alcohol solutions, and a loss of diurnal drinking rhymicity.
This model has high construct validity, as altered circadian rhythms and drinking patterns are common behaviors in alcoholics.[1] Factors influencing face, construct, and predictive validity, in this case, include rodent strains, as some rodents lines such as UChB and AA have in-born alcohol preference, making these studies very useful in assessing specific genetic risk alleles that predispose humans to excessive alcohol consumption.[4][5] Predictive validity is informed by compulsivity being reduced by certain pharmacological treatments like naltrexone or acamprosate, which are commonly used in treating alcoholics. Strengths of this model are that it is non-invasive, relatively simple, and uses the route of administration that humans do.
Final Thoughts
Overall, different models are used to model different parts of the addiction process. Self-administration studies may be the “gold standard” pre-clinical study design due to its overall high validity. The acquisition phase of addiction is best modeled with operant self-administration models that directly show the awarding reinforcing effects of abusable drugs. Reinstatement models and incubation of drug craving models demonstrate, with high translational efficacy, both forced and voluntary relapse and drug craving. Substitution studies can accurately model physical withdrawal and dependence.
Modeling the majority of the behavioral parameters of addiction, including compulsive use, relapse, and loss of control over drug use, is best done with outbred mice, maximizing the variation in response to a particular treatment and mirroring the human situation better. More sophisticated models will have to be used to account for all the subtleties and complexities of the addictive process and its multi-symptomatic nature. This includes multi-faceted behaviors such as vulnerability to drug use, drug use motivated by the desire to avoid negative withdrawal effects, as well as the interplay with psychosocial factors that influence drug acquisition, maintenance, and relapse.
References
- Spanagel R. (2017). Animal models of addiction. Dialogues in clinical neuroscience, 19(3), 247–258.
- Lynch, W. J., Nicholson, K. L., Dance, M. E., Morgan, R. W., & Foley, P. L. (2010). Animal models of substance abuse and addiction: implications for science, animal welfare, and society. Comparative medicine, 60(3), 177–188.
- Belin-Rauscent, A. and Belin, D. (2012). Animal Models of Drug Addiction, Addictions – From Pathophysiology to Treatment, David Belin, IntechOpen, DOI: 10.5772/52079.
- Sommer, W., Hytia, P., Kiianmaa K. (2006). The alcohol-preferring AA and alcohol-avoiding ANA rats: neurobiology of the regulation of alcohol drinking. Addiction Biology, 11(3-4), 289-309.
- Quintanilla, M.E., Israel, Y., Sapag, A., Tampier, L. (2006). The UChA and UChB rat lines: metabolic and genetic differences influencing ethanol intake. Addiction Biology, 11(3-4), 310-23.