Introduction
The endeavor to understand the relationship between observed behaviors and underlying brain activity has important implications for the treatment of many diseases in humans and has thus played a central part in neuroscience. To investigate how the patterns of firing in the brain’s neurons make possible the execution of specific behaviors, it is necessary not only to record brain activity but also to be able to precisely and deliberately manipulate this activity in animal models.
In this article, we will focus on an exciting new paradigm for the manipulation of neural activity: magnetogenetics. We will discuss historical methods for controlling activity in the brains of humans and model organisms, including more traditional techniques such as electrical stimulation, and newer techniques such as optogenetics, before examining in detail how magnetogenetics compares to these, and what experimental evidence exists to validate its use.
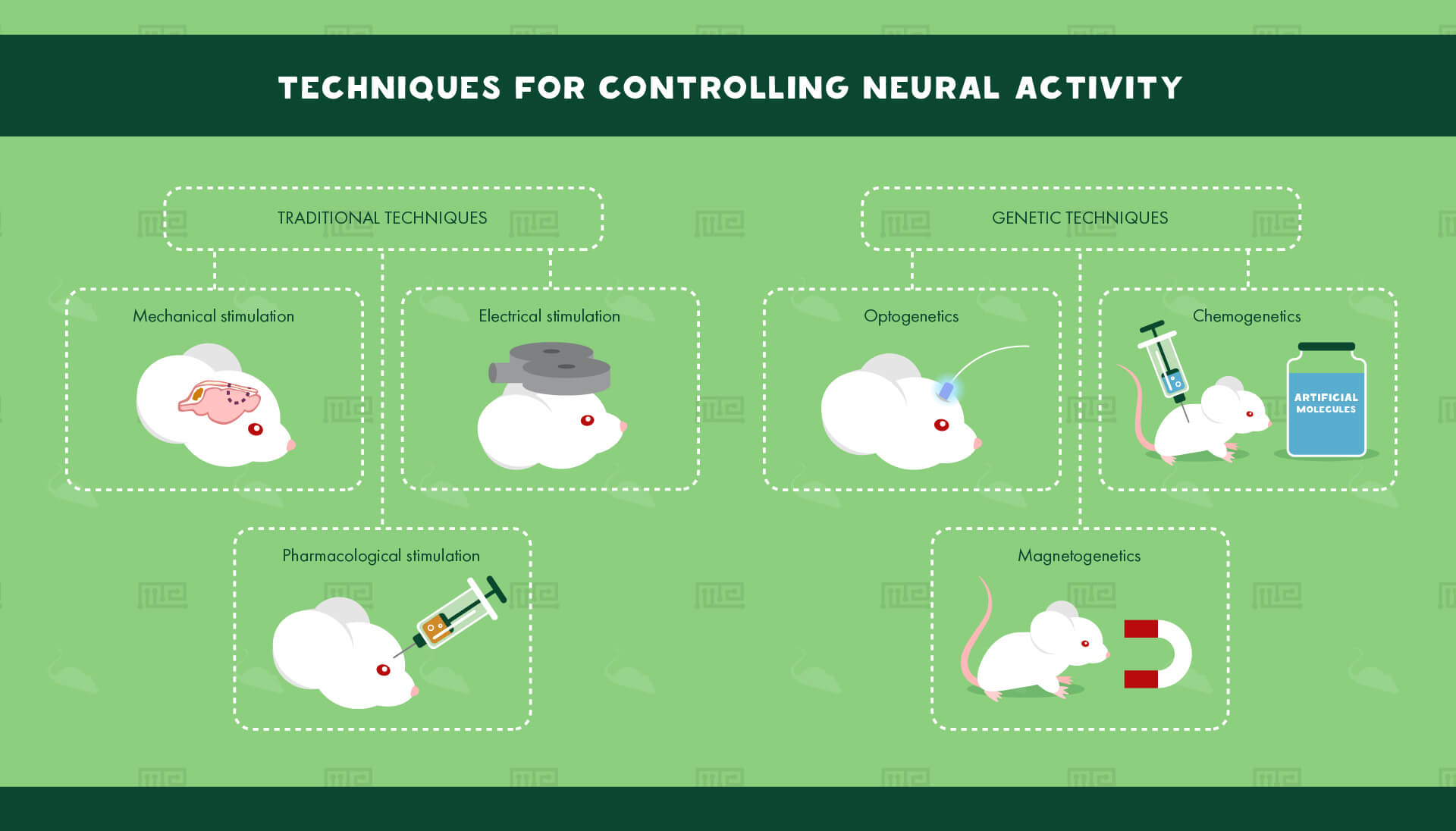
Traditional Techniques for Controlling Neural Activity
A range of techniques has been developed over the course of the last century to allow for the manipulation of neural activity in scientific experiments. Historically, the most popular methods for altering neural activity have been mechanical, pharmacological or electrical in nature.
Many scientists have relied on physical lesioning, where a part of an organism’s brain is permanently destroyed, and then its behavior is examined in comparison to an intact organism of the same species. This method requires extensive surgical intervention and tends to be very imprecise. Critics question the real extent of brain damage, as well as confounding caused by the side effects of the surgery. Lesioning also raises severe ethical quandaries.
Electrical stimulation via electrodes is superior to permanent lesioning but still suffers from similar pitfalls. Non-invasive techniques such as transcranial magnetic stimulation are highly imprecise. Invasive techniques, such as stimulation with implanted arrays, require potentially damaging surgery, and encounter biocompatibility issues. Electrode arrays are only capable of indiscriminately stimulating a large population of neurons.
Drugs have been used in one form or another to alter brain activity for millennia. Pharmacological control of brain activity has the advantage of not requiring surgery. However, drugs can have diverse confounding side effects, which may not be fully understood. They are without a doubt the least precise method of experimental neural control.
Dissatisfaction with mechanical, electrical and pharmacological methods of neural control provided the impetus for the development of genetic methods in recent years.
Genetic Techniques for Controlling Neural Activity
With the advent of much easier genetic engineering in the laboratory, it has become possible to control neural activity through genetic techniques. Genes can be inserted into neurons, allowing for the specific expression of manipulable ion channels. The activity of these neurons can then be controlled by activating the ion channels altering the neurons’ permeability to various ions in real time.
Three major techniques based on this paradigm have now been developed: optogenetics, chemogenetics, and magnetogenetics. These techniques employ light, artificial molecules, and magnetic fields respectively to alter the voltage across neuronal membranes, and thus increase or decrease the probability of action potentials firing.
The key advantage of genetic techniques is that they allow different kinds of neurons in the same location to be treated differently. Whereas lesioning and electrical stimulation tend to have an imprecise and indiscriminate effect on neurons in a wide area, genetic techniques have a higher spatial resolution, can be targeted to particular kinds of neurons, and allow for the inhibition and stimulation of different kinds of neurons at the same time.[1]
The Development of Magnetogenetics
The basis for magnetogenetic control of neurons first emerged from the laboratory of Ali D. Güler at the University of Virginia, US. In 2016 the Güler lab published an article in the journal Nature Neuroscience, describing the work of Michael Wheeler and colleagues on a new genetic technique.[2]
The authors of the study note that a number of previous studies have shown how the cation channel TRPV1 can be linked with the protein ferritin or inorganic paramagnetic particles, allowing it to be manipulated by radio waves and magnetothermal heating. Inspired by this, Wheeler and colleagues engineered a fusion of ferritin with the channel TRPV4, creating the first-ever magnetogenetic system for neural control. They named it “Magneto”.
Calcium imaging experiments in HEK239 cells confirmed that Magneto responds to an electromagnetic field. A second model—Magneto 2.0—was developed to remedy issues with trafficking to the membrane. This model was shown to elicit action potentials in the brain of a mouse and was as effective at this as the current injection.
The researchers then proceeded to test Magneto 2.0’s ability to alter behavior. First, they used the actuator to increase coiling behavior in zebrafish larvae. They then expressed Magneto 2.0 in the striatum of mice, an expression whose effects were clearly seen on electrophysiological measurements. The researchers were then able to control reinforcing behaviors, carrying out an experiment that they claim would have been considerably more difficult with optogenetic technology.
Electromagnetic Control of Feeding Behaviors
A paper published in the journal Nature later in the same year describes how Sarah Stanley and associates used a magnetic technique to control feeding behavior in mice.[3] The researchers wanted to improve upon previous research into the role of the hypothalamus in mouse feeding, looking into the contribution of particular cell types and felt that magnetogenetics would be superior in this regard to optogenetics.
A TRPV1-ferritin fusion was localized to glucokinase neurons (which are believed to sense glucose levels) in the hypothalamus of mice. The researchers were then able to raise and lower glucose levels in the bloodstream by controlling the activation of these neurons. This meant that the researchers could effectively switch a mouse’s feeding behavior on and off at will. They compared their results favorably to similar experiments using optogenetics, and speculate about how magnetogenetics could serve as a non-invasive method for deep brain stimulation in human patients.
Benefits and Downsides of Magnetogenetics
The prime advantage of magnetogenetics over optogenetics is its lack of invasiveness. For optogenetics to work, optical fibers must be implanted into the brain; the light may not penetrate far enough through the brain tissue, and the heat can cause neuronal death. Magnetogenetics requires no implanted devices, and so circumvents all of these issues.
Magnetogenetics also offers advantages over chemogenetics, delivering superior temporal resolution. Chemogenetics, in addition, suffers from an efficiency issue due to the need to repeatedly administer the molecules involved. Magnetogenetics then, at least potentially, compares well to previous genetic techniques.[1] However, it does come with some notable downsides.
First, no-one understands to any great extent how the technique actually works at the molecular level. Meister (2016)[4] asserts that none of the proposed explanations are “even remotely plausible”, and castigates researchers for not being mathematically rigorous. While ferritin-based actuators show promising results, biophysical theory suggests that ferritin nanoparticles should not be capable of activating channels. There is thus concern that the observed results of magnetogenetics studies might be mere artifacts, and robust replication is required to assuage these doubts.
Second, the powerful magnetic fields needed to carry out the technique are much harder to generate than the simple visible light stimuli required for optogenetics. The equipment required is expensive, hard to use, releases a lot of heat, and risks introducing artifacts. The oscillating magnetic fields also make electrophysiology difficult, as they interfere with the electric fields across the electrodes. It is hoped that better coil systems will mitigate these issues.
Nimpf and Keays (2017)[5] argue that, despite apparent protestations to the contrary on the part of those who use it, magnetogenetics is not especially functional at present compared to other genetic techniques. They claim that it is very cumbersome to carry out and that it is slower than optogenetics at inducing action potentials in cell culture by several orders of magnitude. Nonetheless, they cite research which suggests that there is scope for making magnetogenetics considerably faster and more efficient.
Conclusion
Magnetogenetics offers exciting prospects as a new technique for neural control that avoids the downsides of optogenetics and chemogenetics. Magnetogenetics could allow for more precise and less invasive control of behavior in experimental mice, as well as having direct applications in human patients. However, the technique is in the embryonic stages of its development and suffers from difficulty in execution, as well as a lack of any solid theoretical grounding. It is hoped that the replication of prior results, as well as the development of better electromagnetic technologies, will allow for magnetogenetics to prove itself as a major behavioral neuroscience technique in the coming years.
References
- Magnetogenetics – Brain behavior research | Noldus. 2018. Magnetogenetics – Brain behavior research | Noldus. [ONLINE] Available at: https://www.noldus.com/animal-behavior-research/magnetogenetics. [Accessed 07 November 2018].
- Wheeler, M.A.; Smith, C.J.; Ottolini, M.; Barker, B.S.; Purohit, A.M.; Grippo, R.M.; Gaykema, R.P.; Spano, A.J.; Beenhakker, M.P.; Kucenas, S.; Patel, M.K.; Deppmann, C.D.; Güler, A.D. 2016. Genetically targeted magnetic control of the nervous system. Nature Neuroscience, 19, 756-761.
- Stanley, S.A.; Kelly, L.; Latcha, K.N.; Schmidt, S.F.; Yu, X.; Nectow, A.R.; Sauer, J.; Dyke, J.P.; Dordick, J.S.; Friedman, J.M. 2016. Bidirectional electromagnetic control of the hypothalamus regulates feeding and metabolism. Nature, 531, 647-666.
- Meister, M. Physical limits to magnetogenetics. eLife. 2016. 5:e17210.
- Nimpf S, Keays DA. Is magnetogenetics the new optogenetics?. EMBO J. 2017. 36(12):1643-1646.