Need equipment for learning and memory tests?
Procedural memory is a part of everyday life. It’s a cognitive process that enables us to carry out complex behaviors without even noticing. Just thinking about how you get reading in the morning, tying your shoes, putting clothes on, making a sandwich, and driving to work. All of those actions are examples of procedural memory.
Procedural memory is a type of long-term implicit memory that is involved in the encoding, storage, and retrieval of skills not available to conscious awareness. This memory system develops via repeated and gradual procedural learning.
In this way, procedural memory encodes procedures, which are finely tuned motor patterns, behaviors, and motor skills. This is in contrast to declarative memories, or episodes, which correspond to facts that are verbally describable and consciously recalled.
Eventually, procedural learning results in the automation of the procedure once all the collaborating neural systems work together to produce the desired outcome. Because procedural memories form along simple reflex pathways that link sensory inputs to movements, they are different from declarative memories. Compared to declarative memory, procedural memory:
Brain Regions Involved in Procedural Memory
Procedural memory can be further broken down into three main types: cognitive, motor, and perceptual, which are supported by different brain regions.
Cognitive Procedural Memory
Cognitive procedural memory is implicated in learning sequences of decisions and actions. This includes the learning and processing of sequences and rules in language, especially probabilistic rules.
Some examples include predicting the next item in a sequence or the output of a given linguistic rule. In this way, procedural memory allows us to speak without giving conscious thought to the rules of grammar and syntax.[3][4]
Cognitive procedural memory is supported by the basal ganglia as well as the frontal, parietal, and cerebellar regions. The basal ganglia play a key role in the initial learning and consolidation, while frontal regions may be more involved in the processing of procedures that have been learned already.[3]
Motor Procedural Memory
Motor procedural memory is acquired and consolidated under conditions of repeated practice of a motor skill. Skill learning, or motor sequence learning, takes place under cognitive procedural memory routines, i.e. formed within the context of a selected action dictated by cognitive procedural memory.
Some examples of activities supported by motor procedural memory include riding a bike and walking in humans or grooming movements in rodents.
Motor procedural memory is largely dependent on the striatum, basal ganglia, and cerebellum.[3]
Perceptual Procedural Memory
Perceptual procedural memory is utilized in the gradual formation of visuospatial skills, such as the ability to read words on a page or their mirror image in humans or the ability to associate a visual cue with a reward in rodents.
This type of procedural memory is thought to be maintained by cortical regions and the bilateral fusiform area.[3]
Procedural Memory and Declarative Memory are Dissociated Systems
Procedural memory and declarative memory rely on distinct neural systems. The most famous evidence comes from Patient HM, an amnesiac patient who could form new procedural habits despite not being able to form new declarative memories.
Although procedural memory is supported by different neural systems than declarative and working memory systems are, it also interacts with both. For instance, procedural working memory holds representations of what to do with the objects stored in declarative working memory.[5]
Additionally, some functions that are supported by procedural memory can be effectively handled by declarative memory systems. This is true in the case of route learning and navigation in rodents and humans. In this case, hippocampal “place” learning is subserved by striatal “response” learning. That is to say, the animal can shift from allocentric (environment)-based navigational strategies (for instance, water maze place navigation) to egocentric navigational strategies (such as intra-pool cue responding).[6]
Let’s take a closer look at the crucial brain regions supporting procedural memory.
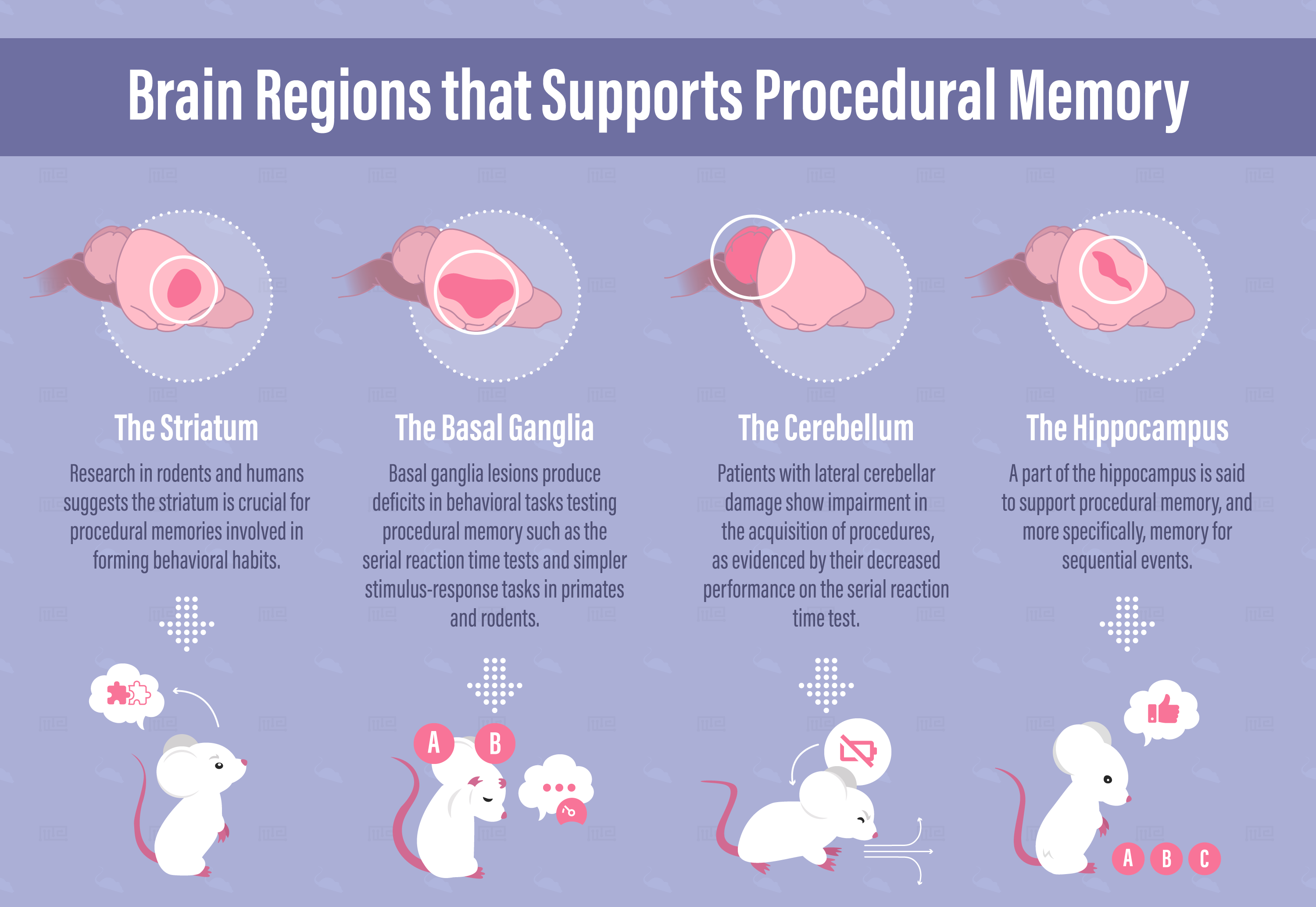
Brain Regions Supporting Procedural Memory
Striatum
Research in rodents and humans suggests the striatum is crucial for procedural memories involved in forming behavioral habits. The striatum is made up of two elements of the basal ganglia called the caudate and putamen. The striatum sits at a crucial location within the motor loop. As the major input region of the basal ganglia, it receives input from the somatosensory and motor cortices and its fibers project to the thalamus and cortical areas that control voluntary movement. In this way, the striatum takes in highly processed sensory information and outputs signals involved in motor responses.
Striatal neurons code for the sequence of behaviors initiating a task, corresponding ultimately to an unconscious procedural habit in the animal. For instance, in a rodent T-maze task, neural responses are initially most active in the striatum when the rat is turning to retrieve the food reward. Over repeated trials, striatal neural responses begin to preferentially fire at the beginning and ending of the procedure and gradually begin to fire over multiple stages of the task. In this way, neural response patterns in the stratum shift to “map” an action template for the procedure once an association with the food reward has been made.
Naturally, lesions to the striatum disrupt the formation of these learned behavioral habits. For instance, rats with dorsal striatal lesions show impairments in stimulus-response learning in Water Maze studies where the escape platform is moved to a novel location after task acquisition.[6]
Basal Ganglia
The basal ganglia are a group of brain structures lying deep within both brain hemispheres. These structures include the striatum, subthalamic nuclei, putamen, globus pallidus, and substantia nigra. Generally speaking, the basal ganglia is implicated in numerous behavioral processes in rodents and humans. This includes motor sequence learning, decision making, habit formation, and reward-based learning.
The basal ganglia circuit has an inhibitory lock on thalamic input, which has excitatory projections to the motor cortex where movement is ultimately executed. This motor loop plays a key role in regulating motor activity by gating the proper initiation and suppression of movement during a goal-directed habit.
Basal ganglia lesions produce deficits in behavioral tasks testing procedural memory, including serial reaction time tests and simpler stimulus-response tasks in primates and rodents.[6]
Cerebellum
Cerebellum has been studied mostly in the context of classical eye-blink conditioning, but it isn’t clear how these insights translate to complex sensorimotor learning. However, it is known that the cerebellum is involved in sensory-motor coordination of ongoing movement and the automation of movement patterns. The cerebellum has extensive connections with the spinal cord and the cerebrum. The left side of the cerebellum controls movements on the left side of the body, while the right side of the cerebellum controls movements on the right side of the body.
Lesions to the cerebellum may interrupt the proper relearning of motor skills and may halt the automation of a process used when learning a procedure.[7] Patients with lateral cerebellar damage show impairments in the acquisition of procedures, as evidenced by their decreased performance on the serial reaction time test.[19]
In available rodent studies, interfering with long-term depression in the cerebellum through pharmacological and genetic means does not dramatically impair the acquisition of new motor skills. Additionally, lesions in the cerebellar-dentate nucleus result only in small learning deficits. With the wide availability of transgenic mouse lines, the genetic targeting of specific pathways and mechanisms involving the cerebellum will further reveal its significance in complex sensorimotor learning and procedural memory.[7]
Hippocampus
The hippocampus is most known for its role in place cell-based navigation and episodic memory. However, procedural memory and more specifically, memory for sequential events, is thought to initially depend in part on the hippocampus. Hippocampus is preferentially recruited for early or immediate procedural learning, then cortical and subcortical structures are gradually recruited.
Evidence from amnesiacs and functional imaging suggests the medial temporal lobe is involved in the learning of higher-order sequences during the acquisition of serial reaction time tests. Hippocampal formation lesions in rats were found to impair accuracy on first and second-order sequences in a simple cued operant task. These results suggest that the hippocampal formation plays an essential role in rapid acquisition of higher-order sequence representations.
Corroborating these findings, functional MRI studies in humans have shown hippocampal activation during explicit and implicit motor skill learning on a serial reaction time test.[8]
Behavioral Assays of Procedural Memory in Rodents
In this section, we will see which behavioral assays are used by behavioral neuroscientists to test procedural memory in rodents and in humans.
Illuminated Radial Arm Maze
In an illuminated variant of the standard Radial Arm Maze designed to test procedural memory, lights are placed at the entrance of each radial arm that contains food, while unlit arms have no food. In this way, the rat learns a consistent association between lights on the maze and the presence of food, so that declarative memory is not required to discriminate among the arms or which arms have been previously entered. Instead, the rats learn a procedural habit based on the association between light and reward.
In this version, hippocampal lesions disrupt maze performance in the standard radial arm maze but not the lighted version. On the other hand, striatum lesions impair maze performance in the lighted version but has little impact on standard maze performance.[6]
Double H Water Maze
The Double H Maze is a water-filled maze that is constructed of three parallel arms intersecting a central corridor. Similar in design to a Morris Water Maze, an escape platform is submerged at one corner of the maze. This maze is flexible in that it allows the experimenter to choose whether they want to test the rodent’s procedural or declarative memory as strategies used during the training phase. Basically, the experimenter can change the navigational strategy of the rat depending on where they decide to have the starting position for each trial.
In a procedural paradigm, the maze assesses learning patterns such as left-right choices, or acquiring the escape platform’s location. Procedural patterns are assessed when the researcher initiates the task from the same starting position, so that the animal can gradually learn the procedural patterns to reach the platform location. This maze is ideal for studying procedural memory disruptions due to neurodegenerative diseases or drugs.[9]
Reaction Time Tests
Reaction Time Tests are procedural memory tasks involving sequences that engage corticostriatal and cortico-cerebellar processes underlying the temporal organization of behavior.
Most commonly, this may involve selecting an appropriate response when shown a visual cue, and the resulting response time reflects procedural processes (i.e. interpreting stimulus, retrieving information from memory, and initiating a muscle response). In these tasks, complexity can be adjusted by varying the number of sequenced movements and the degree of temporal precision enforced.
For example, the five-choice serial reaction time task (5CSRTT) is a type of operant conditioning chamber that has a platform with 5 holes/apertures. A mouse is exposed to a light and it must quickly identify from which hole the light came from in order to receive a sugar reward. Between each trial, there is a short pause where the mouse is expected to withhold any response. A response during this short interval is considered to be a failure of inhibitory control or, in other words, a sign of impulsivity. The apparatus can be modified to have any number of holes/apertures to perform the task.
Serial Reaction Time Test in Humans
In a serial reaction time test, patients see a visual cue at one of four positions arranged horizontally on a computer screen. Each position corresponds to a response pad so that patients can select the appropriate response when a given cue is shown. The cues are typically shown in trial blocks in a repeating sequence or randomly. Procedural learning results in faster response times for repeated than random sequences of cued locations.[11]
Behavioral Assays of Procedural Memory in Humans
Pursuit Rotor Task
The Pursuit Rotor Task in humans is a visual-motor tracking task that tests hand-eye coordination and visual-motor tracking skills. In this task, subjects follow a moving object with a cursor on a computer screen or turntable while researchers assess both procedural memory performance (over repeated trials) and fine-motor skills. The task performance is calculated by time-on and time-off the object. While amnesic patients perform well on this task, task performance is impaired by sleep loss, especially stage 2 of NREM, and drug use.[10]
Mirror Star Tracing
Mirror Star Tracing is a visual motor learning task that assesses the acquisition of a motor task. In this task, patients learn to trace an image of a star and try to stay within the boundaries of the image. The results are then plotted as the number of errors within each session.
Amnesic patients, such as the famous patient HM, are able to learn and retain this task as well as show improvements over subsequent sessions. Similarly, Alzheimer’s disease patients show deficits in hippocampal-dependent declarative memory but demonstrate retained procedural memory on this task.
Weather Prediction Task
In this task of cognitive procedural memory, patients have to guess which combination of one, two, or three out of four possible visual cues are associated with a prediction of sunny or rainy weather. With one of 14 possible combinations, the experimenter assigns different probabilities to each cue signaling sun or rain. Patients begin to build up an association between the cues and weather when they are told whether they made correct or incorrect guesses.
This task uses procedural learning by drawing on the formation of a stimulus-response habit.
Declarative memory can be tested when the experimenter uses a questionnaire in which the patients answer multiple-choice questions about the appearance of the cues on the screen.[6]
Diseases Affecting Procedural Memory
Alzheimer’s Disease
Alzheimer’s disease is characterized by widespread cortical atrophy, including subcortical striatal structures important for procedural memory. Alzheimer’s dementia patients have difficulty acquiring and retaining procedural memory compared to age-matched controls and amnesic patients.
While this disease tends to preferentially degrade episodic memory more than procedural memory, this memory system is still disrupted compared to healthy controls. Although the mechanism underlying the disruptions is unclear, current research suggests that it may be a consequence of changes in enzyme activity in brain regions supporting procedural memory, including the hippocampus and cerebellum.[12]
Parkinson’s Disease
Parkinson’s disease is characterized by degeneration of dopaminergic substantia nigra inputs to the striatum. Parkinson’s patients show difficulty in learning the weather forecasting task but perform at normal levels in a weather forecasting questionnaire assessing declarative memory.
In one study of 20 Parkinson’s patients, patients with advanced symptoms showed impaired Rotary Pursuit Learning compared to age-matched controls and less improvement across days than controls for Mirror Reading Tasks.
Huntington’s Disease
In advanced stages of Huntington’s, the striatum is damaged as well as important corticostriatal pathways. Huntington’s disease patients show difficulty in learning tasks in which a motor response is associated with a given stimulus, or stimulus-response learning.
In 15 HD patients undergoing a simple maze-learning task on a computer, the group was found to have impairments in both generalizing cognitive skills across mazes and improving maze performance on predictable routes. Researchers concluded that these skills were contingent on basal ganglia structures, which are preferentially attacked by the disease.[13]
Drugs Affecting Procedural Memory
Cocaine
Cocaine exerts its effects in the brain mainly through the inhibition of the reuptake of neurotransmitters called monoamines, resulting in its high abuse potential and euphoric effects. These monoamines include dopamine, norepinephrine, and serotonin. D1 receptors are important receptors for long-term procedural memory consolidation and mediate cocaine-induced physiological and behavioral responses.[14]
In one 2008 study published in Neuroscience, researchers administered trace amounts of cocaine prior to a Running Wheel Task paradigm in rats. They found it stabilized motor memory consolidation by enhancing striatal D1 receptor-mediated molecular processes. Similarly, when cocaine was given in conjunction with a D1 receptor antagonist, it reversed the inhibition of skill learning produced by the antagonist and enabled intact skill performance.[15]
Amphetamine
Amphetamine is a psychostimulant with high abuse potential and a similar mechanism of action to cocaine. It increases dopamine signaling through various mechanisms, including inhibiting dopamine uptake and causing the release of dopamine from presynaptic terminals.
In one rodent study investigating conditioned emotional responses in a training chamber, procedural memory consolidation was enhanced in a striatal site-specific manner by intracerebral infusions of amphetamine. When researchers injected amphetamine into the ventrolateral dorsal striatum, this led to an enhancement of procedural memory involving olfactory sensory information, while infusion into the poster-ventrolateral dorsal striatum increased procedural memory performance with respect to visual sensory information.[16]
Similarly, posttraining injection of amphetamine in the intradorsal striatum enhances procedural memory performance in both a Cued Water Maze and Illuminated Radial Arm Maze tasks. As mentioned previously, both tasks were designed for rats to acquire rewards through a procedural learning mechanism.[17]
D1-Receptor Antagonists
While dopamine agonists appear to enhance procedural learning strategies and procedural memory consolidation, dopamine receptor antagonists reverse these effects. Thus, intact dopamine signaling is a crucial element of habit formation and procedural memory consolidation.
Injection of the dopamine receptor antagonist flupenthixol into the dorsal striatum of rats produces deficits in procedural memory in the Radial Arm Maze. Similarly, the D1 receptor antagonist SCH-23390 impaired skill learning on a Running-Wheel task in mice, but this effect was attenuated by concurrent cocaine administration[18]
Cannabinoids
The endocannabinoid system is an evolutionarily ancient biological system that plays an important regulatory role in learning and memory processes of the dorsal striatum. Most endocannabinoids (cannabinoids produced within the body) and phytocannabinoids (such as THC from marijuana) bind to both CB1 and CB2 receptors. CB1 receptors are found on both glutamatergic corticostriatal projections and GABAergic medium spiny neurons within the striatum, while CB2 receptors are found in certain peripheral tissues.[17]
Rodent studies have shown that both enhancing and disrupting the endocannabinoid system can impair procedural learning. Mice with a CB1 receptor null mutation show impairments in habit formation in an instrumental learning task, and these results were validated by a systemic injection of a CB1 antagonist into wild-type mice. Acute infusions of either CB1 agonists or antagonists in the dorsolateral striatum has been found to reduce the use of procedural learning strategies in the T-Maze.
Acute systemic or intra-dorsolateral striatum infusions of CB1 receptor agonists and antagonists have been associated with an impaired procedural learning and reduced memory consolidation in several versions of the T-Maze and Plus-Maze tasks designed to test procedural memory. In contrast to acute infusions, chronic, long-term treatment of endocannabinoids has been found to actually enhance procedural learning. [17]
Conclusion
Procedural memory is a vitally important memory system for unconscious daily activities and, more generally, the ability to interact with the world. Memories for gradually-learned habits and memories for facts are supported by different neural substrates and both are vulnerable to several common neurodegenerative disorders in humans.
Rodents don’t have some of the cortical specializations that support high-level visuomotor skills and dexterity as found in mammals, but they do have in common the fundamental neural architecture for motor control. Thus, rodent models of human disease offer an insightful window into how procedural memory is affected by diseases and drugs. Gradual response-based learning and procedural memory performance can be effectively assessed in rodents with the Illuminated Radial Arm Maze and Double H maze.
In humans, the pursuit rotor, serial reaction time, mirror star tracing, and weather prediction tasks allow careful experimental control and are all widely-used assessments of procedural learning and memory. As such, these tasks are sensitive to disruptions in the underlying subcortical neural systems caused by common neurodegenerative disorders and certain drugs. With respect to the latter, drugs that act on the dopaminergic and endocannabinoid systems can disrupt or enhance procedural memory as a consequence of the mediating effects of these systems on striatal plasticity.
Summary
- Procedural memory is a type of long-term implicit memory that is involved in the encoding, storage, and retrieval of skills not available to conscious awareness
- Procedural memory can be broken down into three main types: cognitive, motor, and perceptual, which are supported by different neural systems.
- Cognitive procedural memory is implicated in learning sequences of decisions and actions. It relies on the basal ganglia, cerebellum, frontal and parietal regions.
- Motor procedural memory is involved in the gradual learning of motor skills. This system is supported by the basal ganglia and cerebellum.
- Perceptual procedural memory supports the gradual learning of visuospatial skills such as reading the mirror image of words. This system relies on cortical regions and the bilateral fusiform region.
- Procedural and declarative memory are supported by distinct neural systems.
- The striatum is a crucial structure for forming behavioral habits, gradually coding for the sequence of motor behaviors initiating a task.
- The basal ganglia are important for motor sequence learning, decision making, habit formation, and reward-based learning.
- The cerebellum is involved in the sensory-motor coordination of ongoing movement and the automation of movement patterns.
- The hippocampus is recruited in early procedural memory and plays an important role in the acquisition of higher-order sequence representations.
- Procedural memory is assessed in rodents using the illuminated radial arm maze, double H water maze, and serial reaction time tests.
- In humans, procedural memory can be assessed using serial reaction time, pursuit rotor, mirror star tracing, and weather prediction tasks.
- Neurodegenerative diseases such as Alzheimer’s, Parkinson’s, and Huntington’s disease impair procedural memory.
- Drugs affecting the dopaminergic system such as amphetamine and cocaine can enhance procedural memory consolidation.
- D1-receptor antagonists and cannabinoids can disrupt procedural learning and procedural memory consolidation.
References
- Churchill, J. D., Stanis, J. J., Press, C., Kushelev, M., & Greenough, W. T. (2003). Is procedural memory relatively spared from age effects? Neurobiology of Aging, 24, 883–892.
- Steidl, S., Mohi-uddin, S., & Anderson, A. K. (2006). Effects of emotional arousal on multiple memory systems: evidence from declarative and procedural learning. Learning & memory (Cold Spring Harbor, N.Y.), 13(5), 650–658. doi:10.1101/lm.324406
- Mochizuki-Kawai, H. (2008). [Neural basis of procedural memory]. Brain and Nerve, 60(7), 825-832.
- Shrivastava, Rahul and Tripathi, Sudhakar and Kumar, Prabhat, Modeling of Basal Ganglia to Incorporate the Procedural Memory (March 9, 2019). Proceedings of 2nd International Conference on Advanced Computing and Software Engineering (ICACSE) 2019.
- Magallón, S., Narbona, J., & Crespo-Eguílaz, N. (2016). Acquisition of Motor and Cognitive Skills through Repetition in Typically Developing Children. PloS one, 11(7), e0158684. doi:10.1371/journal.pone.0158684
- Bear, M. F., Connors, B. W., & Paradiso, M. A. (2015). Neuroscience: Exploring the brain (4th ed.). Philadelphia, PA: Wolters-Kluwer
- Ölveczky, B. P. (2011). Motoring ahead with rodents. Current Opinion in Neurobiology, 21(4), 571–578.
- Ergorul, C. (2006). Essential Role of the Hippocampal Formation in Rapid Learning of Higher-Order Sequential Associations. Journal of Neuroscience, 26(15), 4111–4117.
- Kirsch, R.D., Pinnell, R.C., Hofmann, U.G., Cassel, J.C. The Double-H Maze: A Robust Behavioral Test for Learning and Memory in Rodents. J Vis Exp. 2015 Jul 8; (101):e52667.
- Ewert J, Levin HS, Watson MG, Kalisky Z. Procedural Memory During Posttraumatic Amnesia in Survivors of Severe Closed Head Injury: Implications for Rehabilitation. Arch Neurol. 1989;46(8):911–916.
- Robertson EM. The serial reaction time task: Implicit motor skill learning. J Neurosci. 2007;27:10073–10075.
- Kawai H, Kawamura M, Mochizuki S, et al. [Longitudinal study of procedural memory in patients with Alzheimer-type dementia]. No to Shinkei = Brain and Nerve. 2002 Apr;54(4):307-311.
- Bylsma FW, Brandt J, Strauss ME. Aspects of procedural memory are differentially impaired in Huntington’s disease. Archives of Clinical Neuropsychology. 1990;5:287–297.
- Willuhn, I., & Steiner, H. (2009). Skill-memory consolidation in the striatum: critical for late but not early long-term memory and stabilized by cocaine. Behavioural brain research, 199(1), 103–107.
- Willuhn I, Steiner H. Motor-skill learning in a novel running-wheel task is dependent on D1 dopamine receptors in the striatum. Neuroscience. 2008;153:249–258
- Viaud, M.D., and White, N.M. (1989). Dissociation of visual and olfactory conditioning in the neostriatum of rats. Behav. Brain Res. 32,
- Byrne, J. H. (2008). Learning and memory: A comprehensive reference. Amsterdam: Elsevier.
- Willuhn I, Steiner H. Motor-skill learning in a novel running-wheel task is dependent on D1 dopamine receptors in the striatum. Neuroscience. 2008;153:249–258.
- Torriero S, Oliveri M, Koch G, Lo Gerfo E, Salerno S, Petrosini L, et al. Cortical networks of procedural learning: evidence from cerebellar damage. Neuropsychologia. 2007;45(6):1208–1214.