Pain
Pain is a physiological and emotional response to a noxious stimulus. Pain receptors (nociceptors) throughout the body convey sensation from mechanical and chemical stimuli over dedicated nerve fibers to the brain, where they are interpreted as pain. These messages can be modified by other concurrent experiences[1] or by endogenous opioids within the body[2]. Chronic pain is a condition that occurs when the pain continues to be perceived after the stimulus has been removed. While this is perceived as pain, the mechanism seems to involve hyperactivity of the glial cells rather than re-stimulation of the nociceptors[3]. While some understanding of pain has come from human studies, much of our current understanding of pain and ways to treat pain come from animal models of pain.
Methods of Inducing Pain in Mice
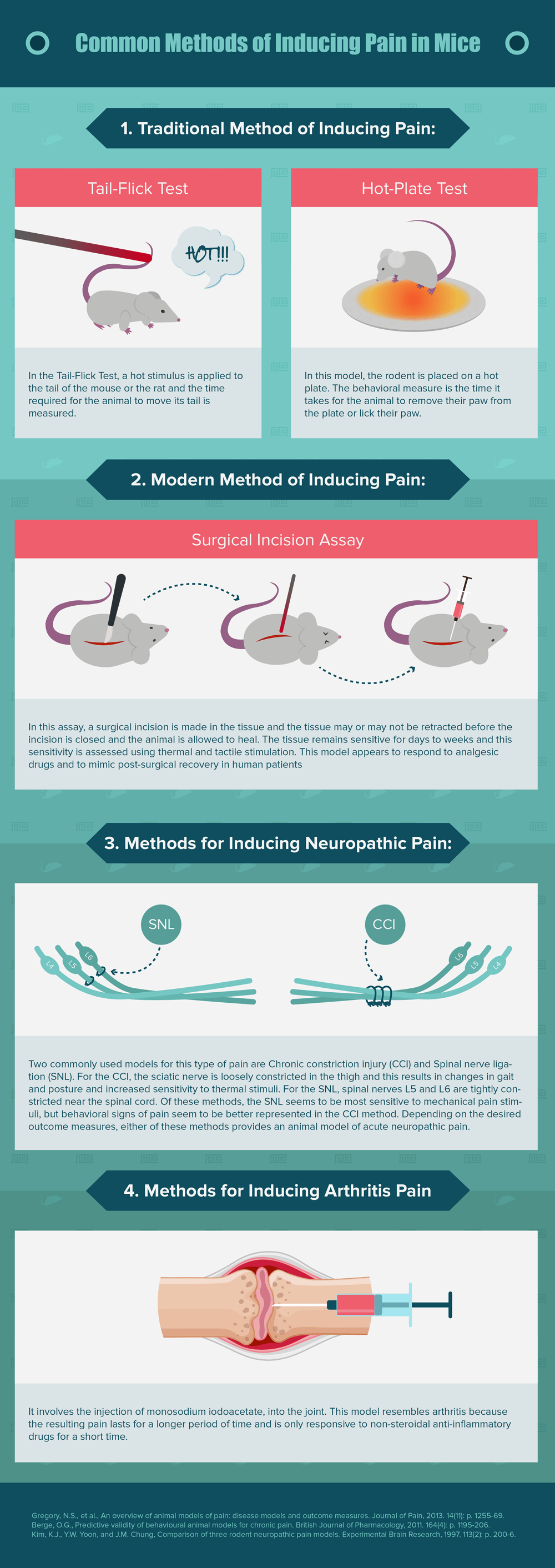
Traditional Method
Animal models of pain have been used for more than 50 years to understand the human condition. In the classic technique, a stimulus such as heat, mechanical force, noxious chemicals, or electric shock is applied to a part of the body[4, 5]. This generates a behavioral reaction, which is observed and scored. To use such techniques, researchers might employ the
In the Tail-Flick Test, a hot stimulus is applied to the tail of the mouse or the rat and the time required for the animal to move its tail is measured. This is a reflex controlled by the spinal cord, but the reflex can be altered by brain stem centers[4].
Another similar technique is the Hot-Plate Test, in which the rodent is placed on a hot plate. The behavioral measure is the time it takes for the animal to remove their paw from the plate or lick their paw[4]. Mechanical stimuli can also be used to apply force to the skin or to muscles or joints.
Most commonly, the assessment for these tests involves the time until the animal withdrawals away from the painful stimulus.
Modern Method
Human patients experience many different forms of pain, and that subjective experience varies between people, probably due to differences between patients in terms of sensation, cognition, and emotion. Clinical experience demonstrates that modern analgesics, while effective in animal models, are only effective for some patients in some conditions. This suggests that animal models of pain do not reflect accurately the variability in symptoms and pathology found in the human conditions. Pain researchers argue that a more accurate model of pain and analgesia would be effective for only some of the animals in some of the conditions[6, 7].
To address these concerns, newer pain assays have been proposed. This includes a Surgical Incision Assay. In this assay, a surgical incision is made in the tissue and the tissue may or may not be retracted before the incision is closed and the animal is allowed to heal[6]. The tissue remains sensitive for days to weeks and this sensitivity is assessed using thermal and tactile stimulation. This model appears to respond to analgesic drugs and to mimic post-surgical recovery in human patients[6].
Methods for Inducing Neuropathic Pain
Neuropathic pain, such as patients might experience from compressed nerves, diabetes, or shingles, can also be modeled in animals. Two commonly used models for this type of pain are
- Chronic constriction injury (CCI)
- Spinal nerve ligation (SNL).
For the CCI, the sciatic nerve is loosely constricted in the thigh and this results in changes in gait and posture and increased sensitivity to thermal stimuli[6]. For the SNL, spinal nerves L5 and L6 are tightly constricted near the spinal cord[6]. Of these methods, the SNL seems to be most sensitive to mechanical pain stimuli, but behavioral signs of pain seem to be better represented in the CCI method[8]. Depending on the desired outcome measures, either of these methods provide an animal model of acute neuropathic pain.
Methods for Inducing Arthritis Pain
In addition to acute pain, chronic pain such as arthritis is a common complaint in patients. Classically, models of long-term inflammation involved injecting a chemical such as capsaicin or Freund’s complete adjuvant into the joint[9]. The amount of pain experienced by the animal is assessed by monitoring their behavior. The degree to which animals avoid using an injured paw or limb is also used to assess deep pain in the limbs or joints. The more precise model of arthritis involves the injection of a different chemical, monosodium iodoacetate, into the joint. This model more closely resembles arthritis because the resulting pain lasts for a longer period of time and is only responsive to non-steroidal anti-inflammatory drugs for a short time[6]. The degree of pain is assessed in this model by quantifying how the animal moves and how they react to physical stimulation. While this cannot capture the long-term pain experienced by human patients with arthritis, it is an improvement on previous chemical injections.
Measuring Pain In Animal Models
Because animals cannot self-report about their pain, researchers have developed ways to quantify the sensory experience of the animals that go beyond behavioral observations and try to assess cognitive perception of pain by the animals. Some of these rely on classic methods from psychology, such as conditioned place preference[4, 5]. In such models, the animal is placed in a chamber that has two distinct regions. In one, they are administered a painful stimulus. The animal is returned to that same chamber at a later time and the amount of time spent in the region where they received the painful stimulus can be compared with the amount of time spent in the region not associated with the stimulus. In this conditioned place preference or conditioned place avoidance task, the animal avoids areas that are associated with unpleasant experiences and seeks out areas that are more pleasurable. This is viewed as a measure of how painful or unpleasant the experience was for the animal, and may provide an assessment of the animal’s experience that goes beyond behavioral observations.
Clinical observations in humans highlight that chronic pain also impacts cognitive functions, such as loss of pleasure in other activities, anxiety, loss of appetite, memory impairments, sleep disruption, and impoverished social interactions[4, 5]. The newer animal models of pain observe and quantify such quality-of-life impairments in animals. Changes in stereotypical behaviors, such as rearing, grooming, and movement behaviors, can be used to describe the pain state of the animals[4, 5] and the conditioned place preference task provides insight into how pain changes the decisions and preferences of the animal.
Relevance of Animal Models of Pain to Human Conditions
Regardless of the means of inducing or measuring pain, animal models of pain need to be relevant to a human clinical condition. New models of pain are being developed to look at particular conditions, not to model pain in general. These models include the acute pain associated with post-operative recovery from surgery and the chronic pain associated with arthritis. Other models have been developed specifically for burns, chemotherapy and cancer, labor pain, spinal cord injury, or sickle-cell anemia[5, 6, 10]. Researchers have argued that as their understanding of pain increases, it is now more important than ever to develop animal models of pain that are based on the human clinical experience and that use behavioral outcomes that are characteristic of the way humans experience the disease[4]. Such models need to consider chronic pain that has been present for years, or for the animal equivalent of multiple years for a human. They will also need to address the method of delivery of the drug.
Currently, experimental treatments are typically delivered in a single large dose whereas human treatments are typically delivered in multiple doses to achieve the therapeutic dose[6]. The resulting animal models of pain will be sensitive to the specific pain profile of the condition and sensitive enough that they will reflect the variable effectiveness of analgesics observed in humans.
References
- Campbell, T.S., J.A. Johnson, and K.A. Zernicke, Gate Control Theory of Pain, in Encyclopedia of Behavioral Medicine, M.D. Gellman and J.R. Turner, Editors. 2013, Springer New York: New York, NY. p. 832-834.
- Holden, J.E., Y. Jeong, and J.M. Forrest, The endogenous opioid system and clinical pain management. AACN Clin Issues, 2005. 16(3): p. 291-301.
- Gosselin, R.D., et al., Glial cells and chronic pain. Neuroscientist, 2010. 16(5): p. 519-31.
- Gregory, N.S., et al., An overview of animal models of pain: disease models and outcome measures. Journal of Pain, 2013. 14(11): p. 1255-69.
- Mogil, J.S., Animal models of pain: progress and challenges. Nat Rev Neurosci, 2009. 10(4): p. 283-94.
- Berge, O.G., Predictive validity of behavioural animal models for chronic pain. British Journal of Pharmacology, 2011. 164(4): p. 1195-206.
- McGonigle, P., Animal models of CNS disorders. Biochemical Pharmacology, 2014. 87(1): p. 140-9.
- Kim, K.J., Y.W. Yoon, and J.M. Chung, Comparison of three rodent neuropathic pain models. Experimental Brain Research, 1997. 113(2): p. 200-6.
- Fehrenbacher, J.C., M.R. Vasko, and D.B. Duarte, Models of Inflammation: Carrageenan- or Complete Freund’s Adjuvant-Induced Edema and Hypersensitivity in the Rat. Current Protocols in Pharmacology, 2012. 56(5): p. 5.4.1 – 5.4.4.
- Lei, J., et al., Comparative Analysis of Pain Behaviours in Humanized Mouse Models of Sickle Cell Anemia. PLoS One, 2016. 11(8): p. e0160608.