The Hebb-Williams Maze
Mazes are among the most popular behavioral tests used by researchers that work with rodents. Many varieties of mazes exist to investigate distinct questions regarding spatial learning and memory, intelligence, as well as other rodent behaviors. Just like any tool, it is important to choose the maze that is best suited for a given experiment. If you’re looking to add a dimension of problem solving to your experiment, look no further than the Hebb-Williams maze.
The Basics
The Hebb-Williams maze was introduced in 1946[1]. It was designed to be a test of intelligence in rats, although mouse versions are common as well. While several mazes rely on spatial cues to guide rodents (e.g. Morris Water maze, Radial Arm maze), the Hebb-Williams maze provides no spatial cues to assist the subject. Instead, the maze is designed to require the subject to rely on problem-solving skills.
Radial Arm maze), the Hebb-Williams maze provides no spatial cues to assist the subject. Instead, the maze is designed to require the subject to rely on problem-solving skills.
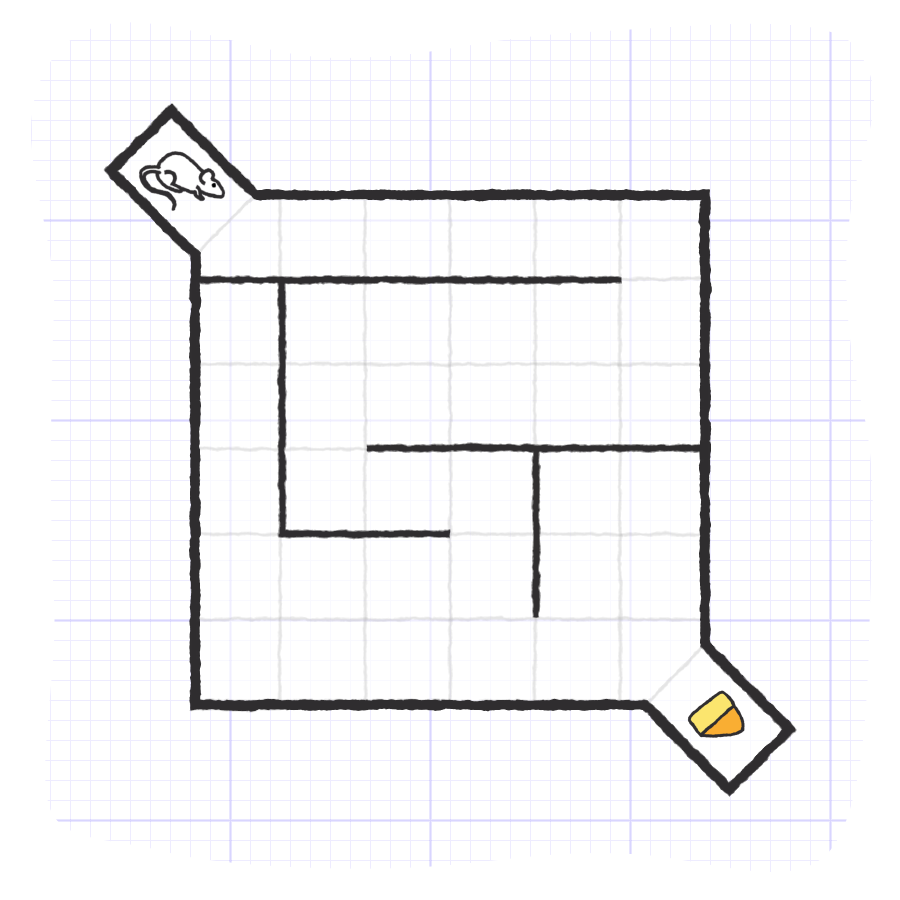
The maze is a square arena, and the start and finish points are always located in the same corners. The interior of the arena is dynamic. A different arrangement of interior walls is assembled for each test, such that the subject relies on the knowledge of the start and end locations to navigate the new interior. It would be difficult to yield results from a dynamic maze that could have the start and end change in between experiments, but fortunately, two scientists named Rabinovitch and Rosvold created a set of standardized interior mazes in 1951[2]. The result is a battery of pre-designed mazes available to everyone that are all still in use today.
Specifications of the Arena
The square arena of Hebb-Williams maze is meant to remain consistent throughout testing. It generally constructed with plexiglass or another opaque plastic, approximately 60 cm x 60 cm x 10 cm[3]. Start and goal boxes should be placed at opposite corners of the maze and remain in those corners for the duration of experimentation.
The interior walls should be made of the same opaque material as the exterior walls of the arena, although they should be moveable. It is common for the floor of maze arenas to be made of transparent plexiglass, and a transparent lid may be used, but the walls should be opaque.
How it Works
Hebb-Williams mazes are performed in three phases: an acclimation phase, an acquisition phase, and a testing phase.
Phase 1 – Acclimation
In the acclimation or habituation phase, the animal is introduced to the arena. This phase is common to many behavioral tests. The animal is placed in the arena (facing the same direction every time, for the duration of all testing phases), and allowed to explore the arena for a set amount of time, often about 5-10 minutes[4], without any interior walls in place.
After several habituation trials in an empty arena, a food reward may be placed in the goal box for some more “baited” habituation trials. For this test, a food reward in the goal box is common practice. This phase should generally last about 3-4 days, with the first trials un-baited and the last few trials baited. After the animal has been sufficiently acclimated to the arena and to the reward box, the next phase of experimentation may begin.





Phase 2 – Aquisition
The second phase is known as the acquisition phase. This is the first phase of experimentation that will utilize the interior walls of the maze. Rabinovitch and Rosvold’s standardized set of mazes includes six acquisition mazes (A-F) that are considered very simple to allow animals to learn how to successfully navigate a maze.
The mazes are generally run in a consistent order and in two sessions per day. Food rewards should be used in this phase, but because multiple mazes are run per session it is important not to overfeed (around 10mg of reward should be sufficient for mice). Although the exact paradigm may vary, each acquisition session should generally consist of around nine trials. Progress through the series of six mazes in order (i.e. A-B-C-D-E-F-A- B-C, or D-E-F-A-B-C-D-E-F)[4].
These acquisition trials should be timed, and if the animal has not reached the goal in around 300 seconds, a piece of opaque plexiglass (that matches the walls) should be used to guide the subject to the finish. The acquisition phase is complete when each animal can complete the full battery of nine trials in less than one minute total for two consecutive sessions. The number of sessions before reaching criterion should be noted for each animal, as individuals will vary in acquisition time.
If only a few animals have not met criterion within a given cohort, one does not need to run all animals in every session. Maintain the performance of the other animals by running them in a few sessions per week until all animals have met criterion.
Phase 3 – Testing
Once all animals have met criterion, the testing phase may begin. There are twelve standardized mazes (1-12) for this phase, and once again they should be tested in the same order for all subjects. Unlike the acquisition phase, only one maze should be used per session. Each subject should have around five trials per session with a given maze.
After a trial, the animal should be given some set amount of time to enjoy their reward before being transported back to the start in a small container. After five trials, they may be returned to the home cage.
How does the Scoring Work?
In the acquisition phase, the primary measures of performance are time to complete a maze and number of sessions until criterion has been met. Many mazes have infrared beams in the start and end boxes to most precisely measure trial time.
In the testing phase, time to goal is not often the primary measure of performance, as it may be more indicative of locomotive capabilities than spatial learning in some cases[4]. It is, however, common practice to note this metric regardless. The number of errors made is often the primary scoring metric. Any entry into a portion of the maze that does not lead to the goal box is considered an error. Other behaviors, such as pauses, should be noted, but are not considered errors.
To be considered a successful trial, the rodent should progress through the maze quickly with no errors. As with many behavioral tests, automated scoring programs exist to assist with evaluation. Many researchers choose to manually score behavior as well to ensure accuracy.
A Multi-Use Test
As with many behavioral tests, the Hebb-Williams maze may be used to assess problem solving differences between rodent strains, drug treatments, and models of disease, among other manipulations. Perhaps one of the most notable qualities of the Hebb-Williams maze is its potential for cross-species application.
As any rodent researcher knows, one of the most persistent obstacles in rodent behavioral research is generalizing the results of a rodent test to human conditions and behavior. In 2001, Shore and colleagues developed a virtual Hebb-Williams maze that allows for the comparison of rodent performance on the maze to human performance on a virtual maze[5].
What You Should Know
Tests with cross-species validity have become increasingly popular, as they are considered to provide a better representation of human conditions and learning mechanisms. Add to that a maze that is sensitive to many natural and manipulated factors, such as sex, environmental enrichment, drug administration, and genetic manipulations, and the Hebb-Williams maze is an attractive candidate for cross-species assessments of problem-solving and intelligence.
References
- Hebb, D.O., and Williams, K.A. (1946). A method of rating animal intelligence. J Gen Psychol, 34(59): 59-65.
- Rabinovitch, M.S., and Rosvold, H.E. (1951). A closed-field intelligence test for rats. Can J Psychol 5(122): 122-128.
- Pritchett, K., and Mulder, G.B. (2004). Hebb-Williams Mazes. Contemporary Topics by the American Association for Laboratory Animal Science 43(5): 44-45.
- Stanford, L., and Brown, R.E. (2003). MHC-congenic mice (C57BL/6J and B6-H- 2K) show differences in speed but not accuracy in learning the Hebb-Williams Maze. Behavioural Brain Research 144: 187-197.
- Shore, D.I., Stanford, L., MacInnes, W.J., Klein, R.M., and Brown, R.E. (2001). Of mice and men: Virtual Hebb-Williams mazes permit comparison of spatial learning across species. Cognitive, Affective, & Behavioral Neuroscience 1(1): 83-89.
