Introduction
In order to effectively navigate through the world, forage for food, avoid danger, and seek shelter, all animals rely on the crucial brain processes that constitute spatial memory. Spatial memory allows us to remember routes to make our way through an ever-changing environment as well as the locations of items within this dynamic environment.
When spatial memory is disrupted, we aren’t able to form representations of space from landmarks that aid in goal-directed behavior, nor are we able to orient ourselves and our passage through space in relation to our surroundings.
Spatial memory has representations in short term and long term memory. The former is denoted by spatial working memory. This system is used for the temporary storage, maintenance, and manipulation of spatial information over short time intervals. For rodents, this is temporary spatial information indicating, for instance, that a certain maze arm is not baited, which will not be relevant in the next trial. On the other hand, spatial reference memory refers to spatial information that has been consolidated and is maintained for long time periods. For instance, this may refer to the trial-independent locations where food may be found in a maze. In rodent experiments, spatial working memories are formed within a single, specific trial which could last from seconds to minutes, while spatial reference memories are created over a succession of trials from the constant circumstances of the task and may last days, weeks, or even longer.
Spatial working memory is commonly assessed using the corsi block tapping test in humans and alternation tasks such as the T-maze or Y-maze in rodents. Spatial reference memory can be assessed with water mazes such as the Morris Water Maze in rodents and using virtual reality environments in humans. The radial arm maze can be used to evaluate both spatial working and reference memory in rodents.
The study of spatial memory in animals has led to important translational findings in humans due to the straightforward spatial paradigm used between all model species.
Brain Regions Associated with Spatial Memory
As animals navigate the world, multiple sensory inputs are combined into a seamless whole that forms a spatial representation of the surroundings. The hippocampal formation is a network of key brain regions involved in forming this coherent representation of external space. The hippocampal formation is located bilaterally in the medial temporal lobe, encompassing the hippocampus, the dentate gyrus, and the subicular cortex.
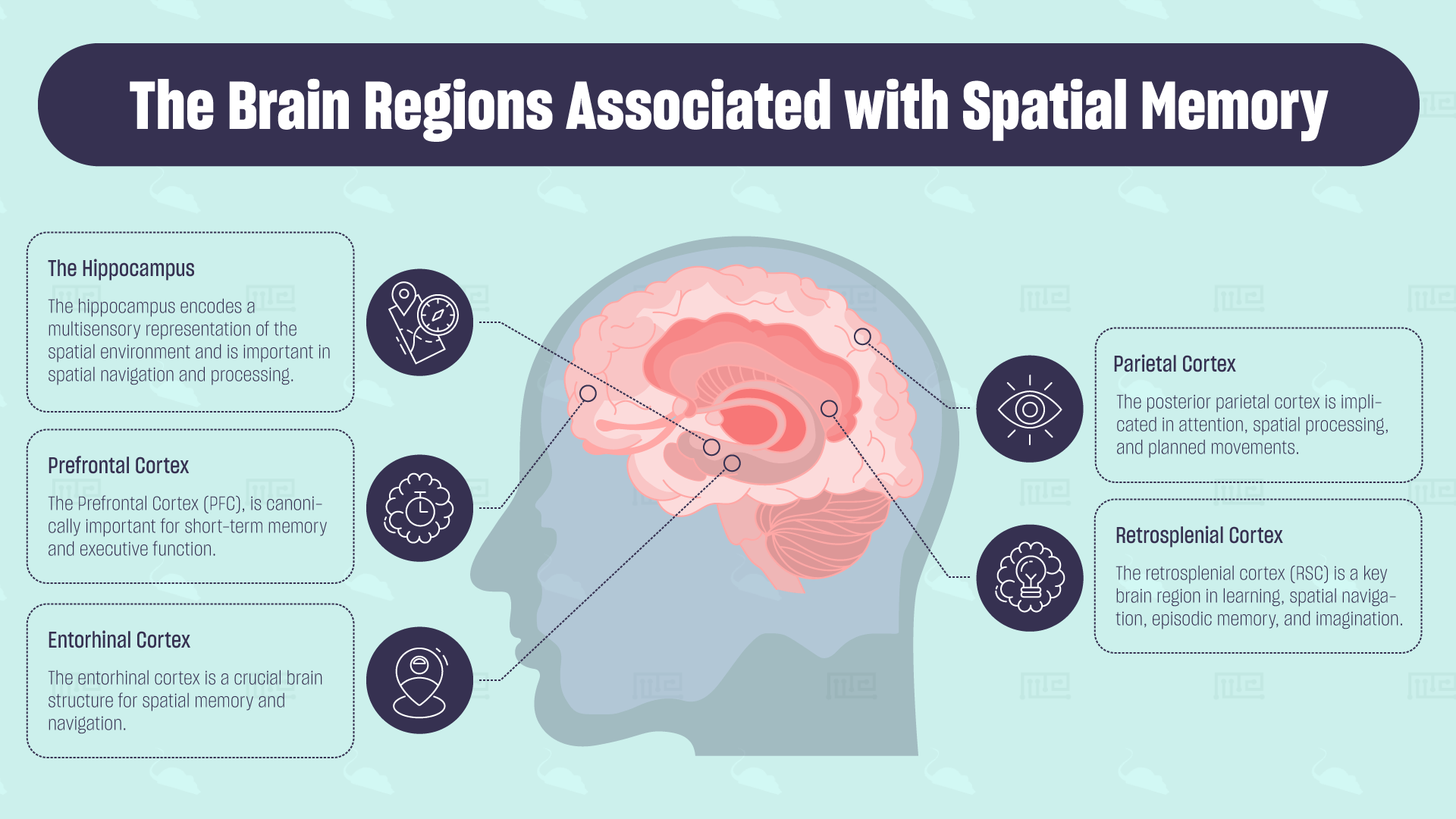
The Hippocampus
John O’Keefe was the first researcher to recognize that the hippocampus proper encodes a multisensory representation of the spatial environment. Many rodent studies utilizing mazes have later confirmed its importance in spatial navigation and processing. When the hippocampus is lesioned, rats have an impaired ability to navigate to rewards or remember specific locations.
The hippocampus contains place cells, which are neurons that fire in relation to the animal’s position in space. In this way, the brain reduces its surroundings to small, overlapping areas in the environment called place fields. These place fields form a mosaic that is represented by the firing of specific place cells in the hippocampus.[1][2]
The activity of hippocampal cells, specifically in the CA3 region of the hippocampus, is also informed by environmental context, so that specific spatial cues can activate the entire neural ensemble encoding the spatial memory. The spatial map constructed by the coordinated firing of place cells is “learned” in a matter of minutes once the animal has navigated a new environment.[1] Modulatory processes like attention further contribute to the long-term stability of these maps.
When the hippocampus is lesioned or plasticity is inhibited, problems with spatial navigation and goal-directed behavior result. Lesions to the dorsal hippocampus create deficits in the retrieval and processing of short-term memory and disrupt the encoding of long-lasting spatial representations. Indeed, dorsal lesions lead to impairments in delayed alternation tasks in rats, while lesions to the ventral hippocampus do not alter spatial memory but instead appear to causally influence fear and defensive behavior.[1]
Entorhinal Cortex
The entorhinal cortex is another crucial brain structure for spatial memory and navigation. The brain region is the main interface between the neocortex and the hippocampus. The dorsolateral medial entorhinal cortex projects heavily to the hippocampus, relaying spatial information important for the formation of long-lasting representations of the environment. The entorhinal cortex contains grid cells, which constitute a hexagonal topographical map of the spatial environment. The periodic firing patterns of these cells are important for the encoding of geometric and spatial properties of the environment and path integration, which is the animal’s use of self-motion cues to update an estimate of their current position and direction. Lesions to the entorhinal cortex produce delay-dependent impairments in spatial navigation and alter the processing of spatial information in exploration tasks.[3]
Retrosplenial Cortex
Found at the back of the corpus callosum, the retrosplenial cortex (RSC) is another key brain region in learning, spatial navigation, episodic memory, and imagination. It projects heavily to the hippocampus, providing important visual and spatial information. Like the hippocampus, it contains place cells that encode important geometric properties of the environment. Lesions to the area induce impairments in spatial navigation abilities as measured by a deficit in the ability to find a fixed-platform in the Morris Water Maze and impairments in the radial arm maze in normal light conditions.[4]
Parietal Cortex
While the hippocampal formation is associated with stable spatial representations of the environment, the locations of objects relative to the animal are represented in the parietal lobe. The posterior parietal cortex is implicated in attention, spatial processing, and planned movements. Damage to this area can cause impairments in perception and memory of spatial relationships, inaccurate grasping, and hemispatial neglect. Hemispatial neglect causes deficits in the awareness and attention of one-half of the visual field.[5]
Prefrontal Cortex
The PFC is canonically important for short-term memory and executive function. The PFC is implicated in task-specific behavior involving selective attention, organization, planning, and coordination of multiple information sources. This area has heavy reciprocal connections with the hippocampus since contextual representations from the ventral hippocampus are sent to the medial PFC. It is therefore thought to play a key role in the retrieval of spatial information encoded in the hippocampus for goal-directed navigation. This is supported by lesioning studies, where medial PFC lesions disrupt performance on a previously trained radial arm maze task and memory retrieval under partial-cue conditions in a hidden platform task.[6]
Development of Spatial Memory
From simple invertebrates to human beings, the development of spatial memory is central to behavior and crucial for survival. The majority of what we know about the neural correlates of spatial memory and its development is derived from rodent studies due to its relative intractability in humans.
In these studies, the development of spatial maps has been parametrized by egocentric and allocentric coordinate frames. The egocentric representations involve the location of objects relative to the animal’s body. These are simpler representations, for instance, the representation of the location of a source of light relative to the fovea, or the source of an odor relative to the animal.
The more simplistic sensory mapping of egocentric representations provides a starting point to understand multisensory representations of the environment. Allocentric spatial representations, on the other hand, involve the location of objects relative to each other and the broader environment independent of the animal’s location. Allocentric representations are likely mediated by non-aggregate interactions between hippocampal and extra-hippocampal regions rather than a specific brain region.[7]
Spatial information received from the external environment must reach the brain through sensory receptors in an inherently egocentric manner. This leads to the question of how the brain learns to transform egocentric sensory representations into allocentric ones, a function that is necessary for the long-term storage of spatial memories. Both the retrosplenial cortex and entorhinal cortex have been implicated in this transformation, but this is still an active area of inquiry.[7]
This problem has been delineated by characterizing the several types of spatially-tuned neurons distributed across the brain. Each of them is a key player in the formation of coherent spatial maps and long-term spatial memory across all vertebrate species. In addition to the place cells discussed above, there are three other spatial neurons classes that allow for functional networks that support the development of spatial memory and navigation.
Head-direction cells are distributed across the midbrain, hypothalamus, thalamus, and hippocampal formation and selectively respond when the animal orients its head in a certain direction, irrespective of the animal’s location.
Grid cells are recorded from parahippocampal regions and fire in broad, triangular arrays that span the environment, with firing patterns that are robust to alterations in the environment.
Boundary cells are found in the medial entorhinal cortex and fire when the animal is along environmental borders that make up barriers to movement, such as the enclosed walls of mazes or edges.[8]
Recent evidence indicates that each type of spatially-tuned neuron matures at different postnatal days in the rat. Head direction cells emerge earliest before the visual system is activated by environmental cues and mediates early homing behavior. Place cells and boundary responsive cells gradually emerge next, followed by grid cells by postnatal day 21. Allocentric spatial navigation develops around this time, and true hippocampus-dependent spatial navigation emerges at around the same time as spontaneous active exploration, at the end of the third postnatal week. Throughout this process, spatial memory may emerge as a function of multilevel coordination of molecular, cellular, and network-level changes that converge at around the time of weaning.
Future work will have to further delineate the roles of specific spatial cells in the development of spatial behavior as well as how mutually independent the egocentric and allocentric representations formed by these spatial cells really are.[8]
Behavioral Assays for Spatial Memory
Due to its fundamental importance across all animals, spatial memory research, including its experimental paradigms, is highly translational across model species.
Alternation Tasks
The T-maze and Y-maze take advantage of the natural tendency for rodents to explore novel environments and locate food rewards. The maze takes into account the alternating behavior of rats as they search for food in two arms. It is popularly used to investigate spatial working memory, and so is sensitive to lesions in the hippocampus.
The total amount of arm entries and the sequence of entries is used to calculate the percentage of alternation. The percentage of alternation can be used to assess hippocampal dysfunction due to lesions, the effects of drugs on spatial learning and memory, and cognitive deficits in transgenic mice used to model human neurological and neuropsychiatric deficits. In a modified T-maze, researchers can join many simple T-mazes together to create a more complex task which can access place learning and cognitive mapping.
Advantages of the maze are ease of setup, simplicity of learning, and minimal stress on the rat. In some paradigms, the rat must be food-deprived, which may be a drawback. Since the trials are usually short and spatial memories are only used by the rat from the previous trial to make the correct choice for the food reward, the maze has limited value in assessing spatial reference memories, that is, information that is useful on any day of testing.
Radial Arm Maze
Created in the mid-1970s, the radial arm maze is a common behavioral assay for spatial memory in rodents. Throughout the maze, the rodent must remember the location of the arms it has already visited for the reward and which arms it has not searched. Therefore, the Radial Arm Maze assesses the spatial memory of the rat as it navigates the maze and remembers the correct and incorrect pathways.
This task requires several days or weeks of training and does not use distant visual cues to aid spatial learning, as in the Morris Water Maze. The maze allows for researchers to control several variables, including the type of food reward hidden, the locations of the reward, the olfactory cues that may confound results, and the retention interval, which is defined as the time between the exposure of information (goal reward locations) and the testing of the retention of that information.[9]
Morris Water Maze
The Morris Water Maze (MWM) is another commonly used task designed by Richard Morris in the 1980s to assess hippocampal spatial learning and memory in rodents. Unlike the binary choice paradigm found in the T-maze or Y-maze, the Morris Water Maze requires the animal to continually choose its route to find the reward. Many aspects of the task are derived from the activity of place cells in the hippocampus, cells whose activity represents specific locations in the maze.
Across trials, which may be multiple days, the animal under normal circumstances progressively finds quicker routes to the submerged platform in part through the use of visual and other external cues. In the process, it forms spatial reference memories quantified as reduced escape latency, or the time it takes to find the platform or an increased search in the correct quadrant when the exposed platform is removed.[9]
This maze is frequently used when assessing the effects of age-related cognitive decline, hippocampal formation lesions, and pharmacological agents on spatial memory. The maze is particularly sensitive to hippocampal dysfunction, as evidenced by impaired performance in rodents with lesions to the dorsal region of this area. The Morris Water Maze has several advantages compared to other tasks, namely, little pre-training required, no food restrictions, ease-of-use, quick learning times, and flexible control of cues, including the removal of undesired olfactory cues. Additionally, the animal’s motivation is equal across tasks, as opposed to food-motivated tasks where hunger and satiety can influence its results. There are some concerns about confounds such as the effects of stress on performance. [9]
Virtual Environments
Virtual reality has the advantage of awarding researchers great control over the test environment and the possibility of creating environments that one wouldn’t normally find in the real world. In humans, virtual reality tasks are often used in conjunction with imaging techniques such as functional MRI. This allow researchers to monitor real-time activity of regions associated with spatial memory while the subject navigates through virtual mazes such as the virtual Morris Water Maze or virtual radial arm maze. More complicated virtual arenas and large-scale worlds can also be created to easily assess hippocampal function.
Corsi Block tapping test
The Corsi block-tapping test is a commonly encountered spatial memory task for humans. This test is modeled after the digit span test created by Hebb for assessing working memory, but in this case, the numerical items are replaced by spatial test items, namely, blocks. In this task, the experimenter taps on blocks in a particular pattern that the subject then must replicate. The pattern increases in complexity as the number of blocks increases. The test usually reaches a maximum of nine blocks, and the average number of blocks an individual can reach is five.
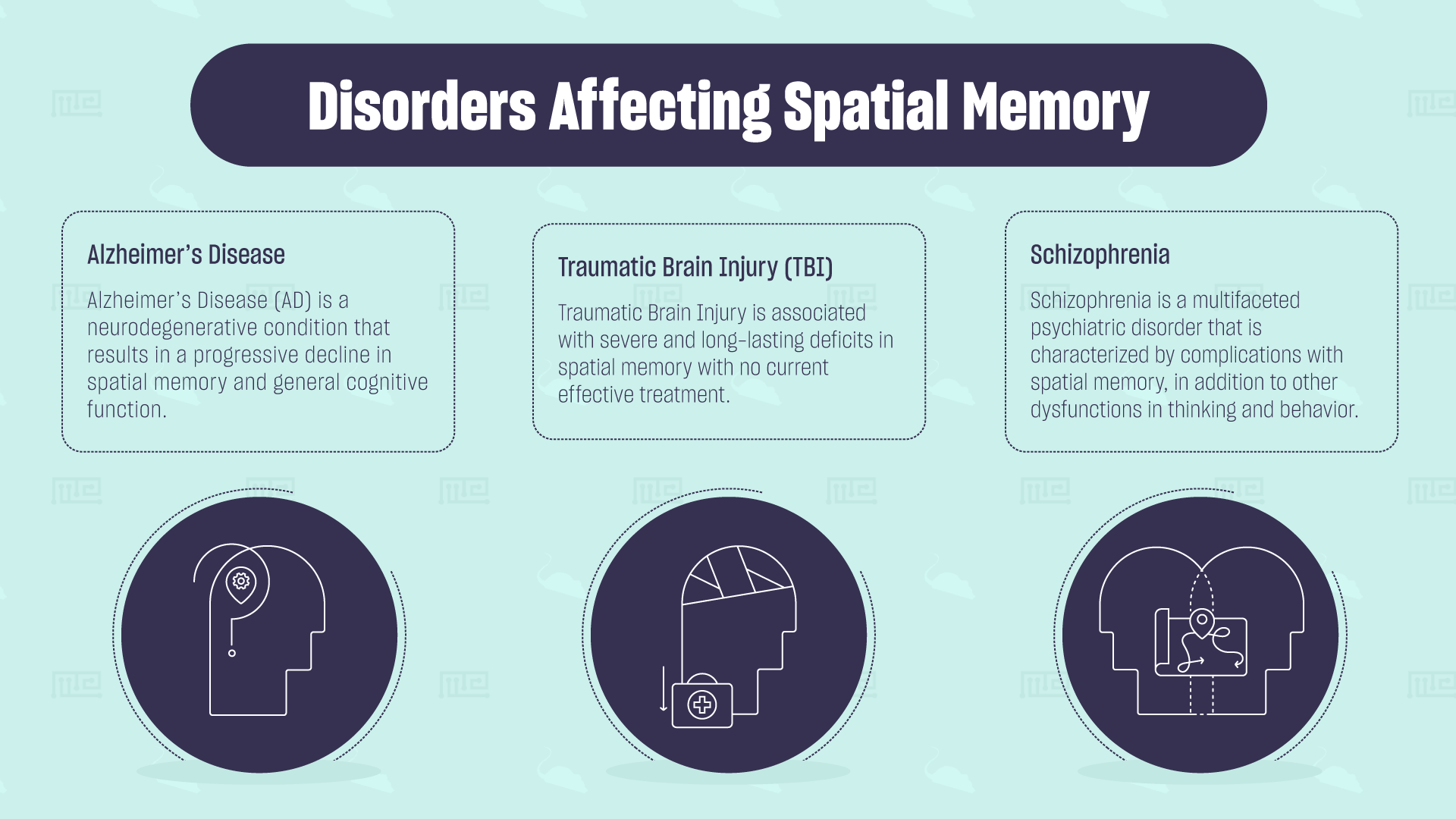
Disorders Affecting Spatial Memory
Alzheimer’s Disease
Alzheimer’s Disease (AD) is a neurodegenerative condition that results in a progressive decline in spatial memory and general cognitive function. The disease is associated with the accumulation of an abnormal protein called amyloid-beta that forms tangles (called neurofibrillary tangles) which lead to progressive cell loss. Degeneration of the hippocampal formation is an early feature of Alzheimer’s disease. These patients progressively show impairments in spatial orientation and navigation. Perhaps one of the earliest indicators of AD and Mild Cognitive Impairment is dysfunction in the retrosplenial cortex and entorhinal cortex. This is seen as reduced activity in these regions as measured by Positron Emission Tomography (PET) in humans.
Mouse models of AD have been instrumental in assessing pharmacological treatment strategies and further understanding the mechanisms behind the pathophysiology of the disease. While no current model entirely recapitulates the human symptomatology of AD, more than 100 genetic models of AD currently exist in mice. Generally speaking, these models are created to increase the rate of amyloid precursor protein (APP) cleavage, leading to a build-up of amyloid beta, or to overexpress mutated tau proteins, resulting in neurotoxic neurofibrillary tangles. Two of the most common models in mice are the PDAPP model and TGGRND8 model. Both models exhibit impaired performance in Morris Water Maze and deficits in decision making in rewarded alternation tasks.[10]
Traumatic Brain Injury (TBI)
Moderate to severe Traumatic Brain Injury is associated with severe and long-lasting deficits in spatial memory with no current effective treatment. TBIs are associated with learning-memory deficits as well as general impairments in executive function that scales with the severity of the injury.
Traumatic Brain Injury can be modeled in rodents with lateral fluid percussion injury and controlled cortical impact (CCI). The latter method is preferred and utilizes an impactor to deliver a precise force onto intact dura exposed after a craniectomy. It can induce TBIs ranging from mild to severe. CCI is the easiest to control, more accurate, and produces TBIs similar to what is found in humans. You can read more about it in our article on animal models of TBI
In rats with moderate and severe TBI, spatial memory and learning impairments as measured by the Morris Water Maze were caused by structural changes in the hippocampus, such as cell loss in CA3, as well as white matter injury in cortex, striatum, and corpus callosum. As such, future therapies may selectively protect and restore the white matter to attenuate the impairments found in TBI patients.[11]
Schizophrenia
Schizophrenia is a multifaceted psychiatric disorder that is characterized by complications with spatial memory, in addition to other dysfunctions in thinking and behavior. In human studies, schizophrenia patients show cognitive impairments in a virtual Morris water maze task relative to healthy controls.[12]
Rodent models of schizophrenia include four categories: developmental, drug-induced, lesion, or genetic manipulation models. Certain rodent models replicate the spatial deficit symptoms of schizophrenia, including NMDA antagonist injections in mice (such as PCP or MK-801), neonatal hippocampal lesions, or transgenic lines such as DISC0-1 Knockouts. These mice show deficits in T-maze performance, impaired spatial working memory, and deficits in spatial object recognition.[13]
Drugs Affecting Spatial Memory
Spatial memory formation can be augmented by drugs that increase synaptic plasticity in the brain regions associated with spatial memory and learning, notably the hippocampus and prefrontal cortex. Glutamatergic, dopaminergic, and cholinergic transmission is all critical to increase synaptic strength and mediate long-term potentiation in the hippocampus. For instance, D1 receptor agonists infused in the medial PFC improves performance on a delayed variant of a radial arm maze task in rats and object recognition before memory retrieval.
Acetylcholinesterase inhibitors such as Donepezil are commonly prescribed in early Alzheimer’s Disease and work by inhibiting the breakdown of acetylcholine in the synaptic cleft. They have been shown to attenuate deficits in recognition memory as well as scopolamine-induced and alcohol-induced spatial memory deficits in rodent studies with mazes.[14]
Many drugs of abuse, including methamphetamine, ketamine, MDMA, alcohol, and cocaine, affect spatial processing and memory. Frequent ketamine and opiate users show reduced activation in hippocampal and parahippocampal regions, resulting in spatial memory deficits as measured in a virtual reality task with concurrent fMRI analysis.
In general, the drugs of abuse are known to recruit the hippocampus, which processes contextual drug associations and plays a role in the acquisition of drug-related associative memories. Drug abuse is thought to involve the disruption of hippocampal long-term potentiation caused by certain features of drug addiction such as context-dependent withdrawal. In rodents, these drugs of abuse, even with their markedly different mechanisms of action, induce cognitive impairments in hippocampal-dependent spatial navigation tasks such as the T-maze, radial arm maze, and Morris water maze.[15]
Polyunsaturated Fatty Acids and Spatial Memory
Polyunsaturated fatty acids promote or inhibit immune and inflammatory responses. Omega-3s show anti-inflammatory properties and promote immune functions, which can attenuate the effects of in utero inflammation caused by maternal immune activation.
Omega-3 PUFA deficiency induces spatial memory deficits and results in altered lipid compositions in the brains of adult mice within a lipopolysaccharide model of maternal immune activation. The deficiency is thought to enhance pro-inflammatory cytokines which have been associated with spatial memory modulation in several animal models.[16]
In humans, treatment with omega-3 polyunsaturated fatty acids can improve learning-memory functions in TBI patients by reducing oxidative stress and increasing brain-derived neuronal factor, leading to enhanced neuronal survival.[17]
Conclusion
Spatial memory is a critical brain process for any properly functioning animal. It is impacted by various drugs and common neurodegenerative and neuropsychiatric conditions. Spatial memory may be assessed in a variety of ways, and many of the maze-based tasks in rodents lend themselves to quick learning times in the animals as well as easy setup and great control of task parameters. Virtual reality tasks are increasingly being used with great success in rodents and humans due to its unique and flexible ability to control the task environment. Understanding the neural and behavioral correlates of spatial memory in rodents under varying task conditions is an effective springboard to understanding how certain diseases and drugs may affect spatial memory in humans.
References
- Broadbent NJ, Squire LR, Clark RE. Spatial memory, recognition memory, and the hippocampus. Proc. Natl. Acad. Sci. USA. 2004;101(40):14515–20.
- Moser, M. B., Rowland, D. C., & Moser, E. I. (2015). Place cells, grid cells, and memory. Cold Spring Harb. Perspect. Biol., 7(2), a01808.
- T. Van Cauter, J. Camon, A. Alvernhe, C. Elduayen, F. Sargolini, E. Save. Distinct roles of medial and lateral entorhinal cortex in spatial cognition. Cereb. Cortex, 23 (2013), pp. 451-459
- Vann, S. D., Aggleton, J. P., & Maguire, E. A. (2009). What does the retrosplenial cortex do? Nature Reviews Neuroscience, 10(11), 792–802.
- Husain M, Nachev P. Space and the parietal cortex. Trends Cogn Sci. 2007;11:30–36.
- Kessels R, Pastma A, Wijnalda E, Haan E (2000a) Frontal-lobe involvement in spatial memory: evidence from PET, fMRI, and lesion studies. Neuropsych Rev 10:101–113
- Ekstrom, A. D., Arnold, A. E., & Iaria, G. (2014). A critical review of the allocentric spatial representation and its neural underpinnings: toward a network-based perspective. Frontiers in human neuroscience, 8, 803.
- Tan, H. M., Wills, T. J., & Cacucci, F. (2016). The development of spatial and memory circuits in the rat. Wiley Interdisciplinary Reviews: Cognitive Science, 8(3), e1424.
- Vorhees, C. V., & Williams, M. T. (2014). Assessing spatial learning and memory in rodents. ILAR journal, 55(2), 310–332.
- Webster, S. J., Bachstetter, A. D., Nelson, P. T., Schmitt, F. A., & Van Eldik, L. J. (2014). Using mice to model Alzheimer’s dementia: an overview of the clinical disease and the preclinical behavioral changes in 10 mouse models. Frontiers in genetics, 5, 88.
- An C, et al. Severity-Dependent Long-Term Spatial Learning-Memory Impairment in a Mouse Model of Traumatic Brain Injury. Transl Stroke Res. 2016
- Folley, B. S., Astur, R., Jagannathan, K., Calhoun, V. D., & Pearlson, G. D. (2010). Anomalous neural circuit function in schizophrenia during a virtual Morris water task. NeuroImage, 49(4), 3373–3384.
- Young, J. W., Powell, S. B., & Geyer, M. A. (2012). Mouse pharmacological models of cognitive disruption relevant to schizophrenia. Neuropharmacology, 62(3), 1381–1390. doi:10.1016/j.neuropharm.2011.06.013
- Gawel K, Labuz K, Gibula-Bruzda E, Jenda M, Marszalek-Grabska M, Filarowska J, Silberring J, Kotlinska JH (2016) Cholinesterase inhibitors, donepezil and rivastigmine, attenuate spatial memory and cognitive flexibility impairment induced by acute ethanol in the Barnes maze task in rats. Naunyn Schmiedeberg’s Arch Pharmacol 389(10):1059–1071.
- Taubenfeld SM, Muravieva EV, Garcia-Osta A, Alberini CM. Disrupting the memory of places induced by drugs of abuse weakens motivational withdrawal in a context-dependent manner. Proc. Natl. Acad. Sci. U. S. A. 2010;107:12345–12350.
- Labrousse, V. F., Leyrolle, Q., Amadieu, C., Aubert, A., Serea, A., Coutureaud, E., et al. (2018). Dietary omega-3 deficiency exacerbates inflammation and reveals spatial memory deficits in mice exposed to lipopolysaccharide during gestation. Brain Behav. Immun. 73, 427–440.
- Kumar, P. R., Essa, M. M., Al-Adawi, S., Dradekh, G., Memon, M. A., Akbar, M., & Manivasagam, T. (2014). Omega-3 Fatty acids could alleviate the risks of traumatic brain injury – a mini review. Journal of traditional and complementary medicine, 4(2), 89–92.