There are hardly any tasks that can be completed without using short term memory.
Indeed, as you are reading this sentence, you are using your short term memory to maintain the earlier words as you read along and understand the entire meaning of the sentence. Short term memory is crucial for many basic tasks in life, such as remembering a new phone number or performing arithmetic. In fact, you may be familiar with the experience of reading a telephone number and mentally rehearsing that number in your head until you can dial it. This one strategy helps the number remain in short term memory for longer, and can even facilitate its storage in long-term memory.
In non-human animals, short-term memory plays a critical role in understanding new environments, allowing goal-directed behavior, and generally speaking, providing a substantial survival advantage. Without it, animals couldn’t successfully avoid predators and search for prey, build nests, and assimilate and understand novel environments within their existing models of the world.
In this article, we’re going to take a look at what short-term memory is, including what brain regions are involved in it and some models that have been created to explain it. Then, we’ll explore the different ways short-term memory is commonly assessed in humans and rodents. We’ll end discussing some common diseases and drugs that affect short-term memory.
What is Short Term Memory?
Short-term memory is a memory system that encodes limited amounts of information in a highly accessible state temporarily. This information could be sensory events, movements, or cognitive information such as digits, words, or names. In contrast to working memory, short-term memory does not include executive processes acting on the stored items, such as the manipulation or organization of information in short-term storage.[1]
In this way, short-term memory is akin to a temporary workspace that allows us to carry out operations necessary for nearly every cognitive task. Nonetheless, working memory always includes a short-term memory component in its assessment, and they are commonly used interchangeably in the literature, especially in relation to tasks that are conducted to measure them.
Duration and Capacity of Short-term Memory
Short-term memory has a duration of about 15 to 30 seconds, and these memories can be easily disrupted until they are consolidated into long-term memory. Once in long-term memory, these sensitive memory traces become much more durable and can persist for an indefinite amount of time.
It’s well-known that the average person can hold about seven plus or minus two items in short-term memory, brought to the forefront from the famous paper published by George Miller in 1956. It’s important to note this number was found under conditions where participants could use mnemonic strategies like chunking and rehearsal to aid in storage. When a participant uses meaning-laden chunking strategies in memorization, they can drastically increase the number of items they are able to memorize since this strategy uses additional information from long-term memory. Therefore, in 2001, Cowan and colleagues revised this number to four plus or minus one. This was devised in part from the sharp performance drop-offs they noted after 3 to 5 items.[2]
Theoretical Models of Short-term Memory
Many models of short-term memory have been devised over the last decades. The so-called multistore models break down short-term memory into storage buffers that are specific for different forms of information.
Atkinson and Shiffrin proposed the influential modal model in 1968, which attempted to explain how perceptions move from short-term memory to long-term memory in discrete stages. Under this model, information flows from the environment through a parallel series of sensory memory systems into a limited-capacity short-term store. This short-term memory store forms a bottleneck between long-term memory and sensory perception.
One implication of this model, which has been supported experimentally, is that the durability of memory is a function of its depth of processing. Simple visual appearance leads to the least learning, phonological (verbal) processing in terms of sound leads to better learning, and semantic processing of meaning leads to the best retention.[3]
This model was then revised by Baddeley and colleagues in 1974, who sought to sub-divide the short-term memory store into multiple components. They modeled short-term memory in terms of a verbal buffer, which they called the phonological loop, which holds information that is rehearsed verbally. The phonological loop stores auditory memory traces and contains a rehearsal component (sometimes known as the articulatory loop) that can aid in the revival of memory traces. Another buffer, the visuospatial sketchpad, maintains visuospatial information and is further divided into spatial stores and visual/object stores, a separation that parallels the dorsal stream and ventral stream pathways of the visual system. These buffers are controlled by a central executive, the modality-free “supervisory system” that directs attention to govern the buffer systems.[4]
The buffers described above were informed by various neuropsychological studies. The researchers noticed there is a dissociation of verbal and visuospatial short-term memory, which became evident from “dual-task interference” paradigms. In these tasks, if one is learning a list of words and then asked to repeatedly rehearse a random word unrelated to the list in the delay period, this process interrupts verbal short-term memory retention (by disrupting rehearsal of the list items). In a similar manner, visuospatial tasks in the delay period (such as tapping in a square pattern repeatedly) interfere with visual short-term memory retention but not verbal short-term memory.[4]
One consequence of these multi-store models is that it views short-term memory and long-term memory as distinct systems with separate representations. However, on the other extreme, researchers have proposed unitary-store models, where short-term memory and long-term memory are not separable.
The unitary model proposes that these memory systems differ not in terms of their representations, but on the activation of these representations and the processes that act on them. Under this model, therefore, short-term memory and long-term memory are not dependent on two distinct underlying neural systems.[3] In multi-store models, forgetting implies the spontaneous decay of the memory trace, but in unitary models, it reflects interference between the items retained.
Short-term Memory and the Brain
At the cellular level, short-term memory results from changes to the function and structure of neurons in various brain regions. New synapses can form or existing synapses can be strengthened to allow for heightened sensitivity in the communication between the neurons involved in the memory. At the molecular level, short-term memory is mediated by alterations in pre-existing proteins. This involves the recruitment of enzymatic signaling pathways such as the cyclic-AMP pathways which enhance neurotransmitter release to strengthen synapses involved in the memory.
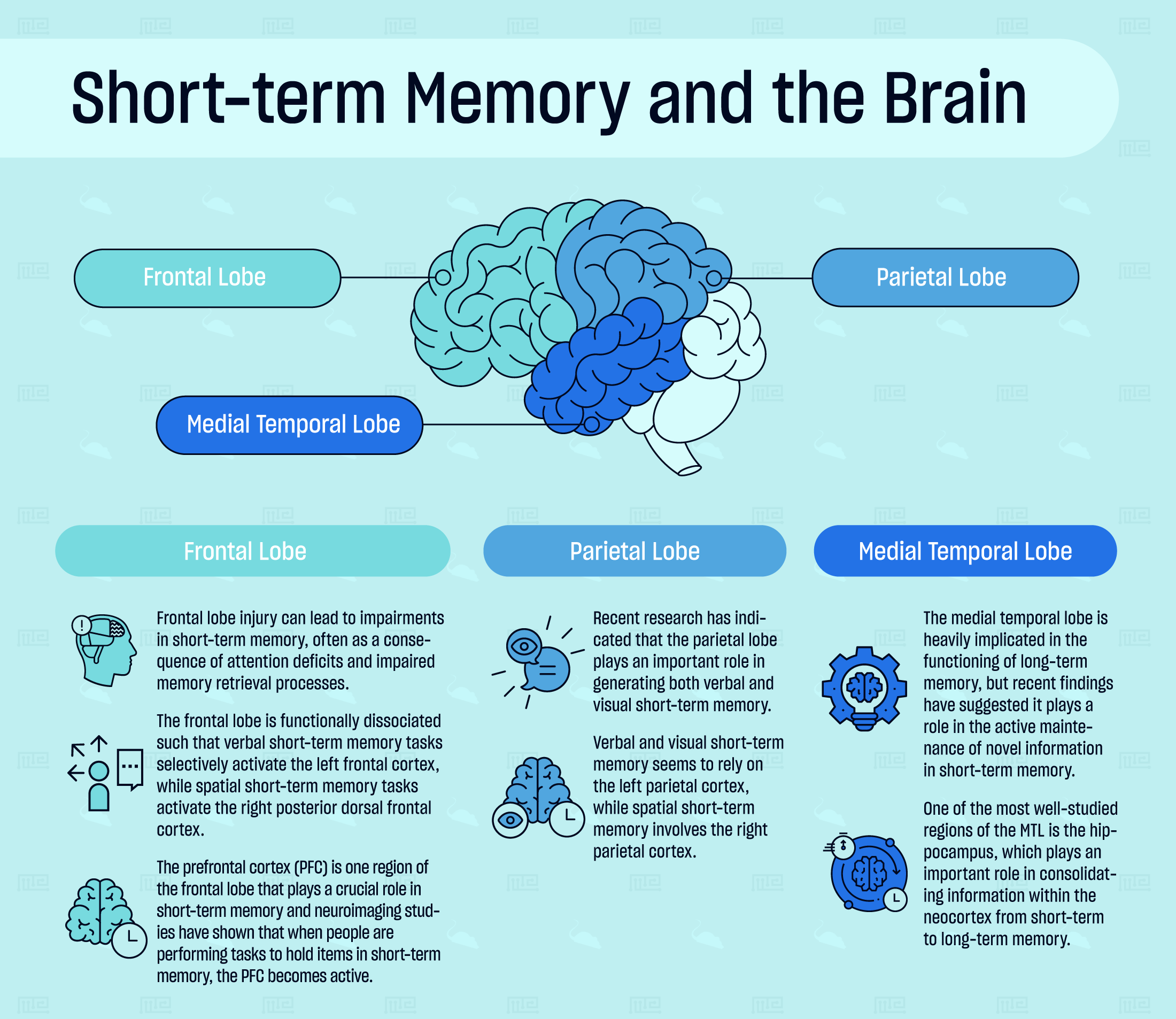
Neuropsychological and neuroimaging studies have demonstrated that short-term memory is supported by neural activity in a network of brain regions, including the frontal lobe, parietal lobe, and medial temporal lobe.
Frontal Lobe
Frontal lobe injury can lead to impairments in short-term memory, often as a consequence of attention deficits and impaired memory retrieval processes. The frontal lobe is functionally dissociated such that verbal short-term memory tasks selectively activate the left frontal cortex, while spatial short-term memory tasks activate the right posterior dorsal frontal cortex. During a visual short-term memory task, the left inferior frontal cortex is activated.[3]
The prefrontal cortex (PFC) is one region of the frontal lobe that plays a crucial role in short-term memory. It covers the front of the frontal lobe and it is the most recent addition to the mammalian brain. Neuroimaging studies have shown that when people are performing tasks to hold items in short-term memory, the PFC becomes active. Like the rest of the frontal lobe, there is a functional dissociation between the hemispheres of the PFC. Verbal short-term memory seems to preferentially activate the left, while spatial short-term memory activates the right side.[5]
Parietal Lobe
The parietal lobe is well-known to be involved in sensory processing, object identification, and understanding spatial relationships. Recent research has indicated that this region of the brain plays an important role in generating both verbal and visual short-term memory. Left parietal lobe damage can produce verbal short-term memory deficits (in the context of aphasia), while right parietal lobe damage can lead to hemispatial neglect and concurrent spatial short-term memory deficits.[6]
Verbal and visual short-term memory seems to rely on the left parietal cortex, while spatial short-term memory involves the right parietal cortex. Additionally, activity in the posterior parietal cortex is tightly correlated with the capacity limits of visual short-term memory.[7] Visual short-term memory emerges from multiple systems acting together, including regions of the parietal lobe such as the inferior intra-parietal sulcus and the super intra-parietal sulcus, but also parts of the occipital lobe.[8]
Medial Temporal Lobe
The medial temporal lobe (MTL) encompasses the hippocampal formation (the dentate gyrus, CA fields, and subiculum), perirhinal, entorhinal, and parahippocampal cortices. The medial temporal lobe is heavily implicated in the functioning of long-term memory, but recent evidence has suggested it plays a role in the active maintenance of novel information in short-term memory. More evidence from studies on MTL-damaged individuals reported impaired performance in verbal and visual memory tasks that had delay periods of a few seconds.[9] In mild cognitively impaired individuals, cortical thinning along the MTL was associated with impaired performance in visual short-term memory tasks.[10]
The structures of the medial temporal lobe are crucial for binding memory items to their context and successfully retrieving those items when context is no longer within one’s attention. One of the most well-studied regions of the MTL is the hippocampus, which plays an important role in consolidating information within the neocortex from short-term to long-term memory. In addition to consolidating long-term memories, the hippocampus has more recently been found to play an important role in the encoding and retrieval of associative information in short-term memory. Interestingly, the retrieval of very short-term memories, on the order of 10 seconds or less, has been found to be exclusively controlled by the subiculum, a region that lies between the hippocampus and entorhinal cortex.[11][12]
Network-level Oscillations Correlate to Short-term Memory Capacity
Recent research has suggested that the capacity of short-term memory may intimately depend on theta (4-7Hz) and gamma (25-50Hz) bands of the brain’s electrical activity. These waves can functionally couple, and their coupling predicts short-term memory performance.
For instance, the amplitudes of these waves increase when people are actively storing information in short term memory. Research published in 2011 from the Nencki Institute of Experimental Biology in Warsaw found that short term memory capacity is correlated to the length of the theta-to-gamma cycle ratio in a modified digit span task. They found that the longer the theta cycles (as measured by EEG), the more items the subjects could remember; the longer the gamma cycle, the less the subject remembers.[13]
Behavioral Assays of Short-term Memory
Many of the behavioral assays of working memory are also used to evaluate short-term memory. In this section, we will overview some of the most common short-term memory tasks in animals and humans. Broadly speaking, short term memory research is dominated by verbal tasks because the material is easy to record and manipulate.
Delayed Alternation
Delayed alternation is a classic paradigm for assessing short term memory in rodents. These tasks exploit the natural tendency for rats to explore and choose alternate maze arms after re-exposure to the task. Since the rodents have to remember their first response to select the alternative response, this task is a short term memory task. The most common delayed alternation task is the T-maze, where the animal begins the task at the stem of the “T” and must choose between two arms, one of which has a food reward.
Mice with deficits in short-term memory show decreased spontaneous alternation on this task. In this way, these tasks are sensitive to aging, hippocampal lesions, and amnestic drugs. Memory load can be increased in delayed alternation by increasing the delay period. Rats with lesioned hippocampi perform at chance level with delay periods as short as fifteen seconds, but above-chance performance has been seen in delay periods up to five minutes using the T-maze.
Match-to-sample and Match-to-place
Generally speaking, match-to-sample tasks require an animal to remember the identity or location of a stimulus for more than a few seconds. In the delayed match-to-sample task used to assess short-term memory retention, the subject is shown a stimulus (such as a colored light or visual pattern), followed by a probe item after a varying delay. The subject is then asked to recall if the probe item was the same as the stimuli presented before. The length of the delay can be varied to gain insight into how long the animal retains the information in short term memory.
In rodents, this task concept is adapted to maze environments, such as delayed non-matched-to-place in the T-maze or water maze. In these tasks, the rat is cued to make a choice response based on a past representation in order to obtain the escape platform location or a food reward. The delayed match-to-sample task activates brain regions associated with short term memory, such as the dorsolateral prefrontal cortex and posterior parietal cortex. In this way, these tasks can explore the effects of lesions in these areas and the effects of associated drug treatments.[14]
Contextual Fear Conditioning
Contextual fear conditioning is a task where the animal is conditioned with an aversive event and then tested for recollection. Mice are usually given a foot shock (unconditioned aversive stimulus) within a specific environment (conditioned neutral stimulus), such that after training the mice will freeze when placed back in the environment. To test short-term memory, the mice are placed in the shock environment up to one hour after training. In an Alzheimer’s disease mouse model, the mice with AD-associated pathologies showed reduced freezing incidences compared to normal mice. Similarly, reduced freeze responses are also noted in sleep-deprived and aged mice, two other conditions that can impair short-term memory.[15][16]
This assessment has the advantage of involving rapid one-trial associative learning. Brain regions such as the amygdala, hippocampus, frontal cortex, and cingulate cortex are thought to be involved in contextual fear conditioning. Research utilizing this task may inform novel pharmacotherapies for conditions such as phobias, anxiety, and post-traumatic stress disorder.
Memory Span Tasks
In the memory span test, participants are shown a sequence of items that they must attempt to reproduce in the presented order. These items can be presented visually or verbally and are commonly digits, consonants, or words in lengths ranging from 0.5s to 2s per item. Performance on these tasks is measured in terms of the maximum level achieved. The disadvantage is that data collection typically occurs in item lengths where performance is perfect, which can provide limited information. Studies utilizing memory span have found performance with unrelated words to be about 5 or 6, but meaningful sentences can create spans of 16 or more words.[4]
Free Recall
The free recall paradigm is another common assessment of short-term memory in humans. In this task, subjects are presented with a list (usually made of words) that they must then recall in any order they wish. Earlier items depend upon long-term memory while more recent items reflect short-term memory, which gives credence to the idea that they are separable systems. Good recall on the first and last few words has been dubbed the recency and primary effect, respectively. Performance is typically measured in terms of the total recalled items on the list. [4]
Pattern Span Tests
Pattern span tests are the visual equivalent of verbal span tests. In pattern span test, a matrix pattern is shown to the subject where certain cells are filled, and then a second matrix is presented to reproduce the pattern after a delay period of varying length. The test may begin with a simple two-by-two matrix with half of the cells filled, and then it will increase in complexity until the subject can’t accurately reproduce the pattern. For a normal adult, this is around 16 cells.
Given the dissociation of verbal and non-verbal short term memory, pattern span tests have been successfully employed for years to measure the visual component of short term memory. However, their face validity alone is not sufficient grounds for clinical use, and so their reliability and validity are established with rigorous psychometric methods, and ideally, concurrent brain imaging work.[17]
Corsi Block Tapping Test
Along the same lines as pattern span tests, the Corsi block-tapping test assesses visual and spatial short-term memory in humans. In this task, the experimenter taps on blocks in a specific pattern that the subject then must replicate. After successful trials, the tapping pattern gets longer and more complex as the number of blocks increases.
The test usually reaches a maximum of nine blocks, and the average number of blocks an individual can reach is five. This test is sensitive to short-term memory impairments commonly found in anterograde amnesia, Alzheimer’s disease, Parkinson’s disease, and frontotemporal dementia.[18]
Diseases and Conditions That Affect Short-term Memory
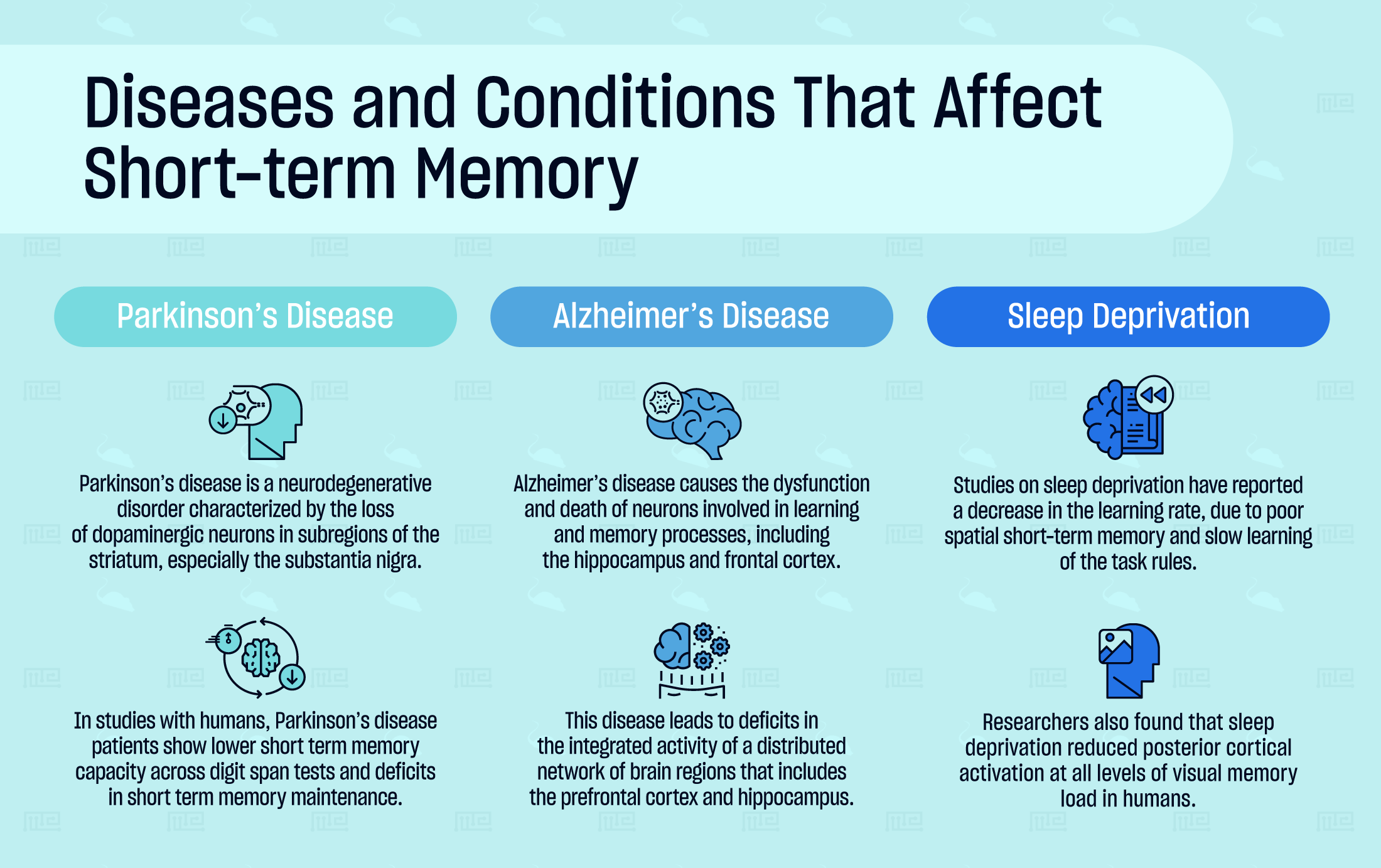
Parkinson’s Disease
Parkinson’s disease is a neurodegenerative disorder characterized by the loss of dopaminergic neurons in subregions of the striatum, especially the substantia nigra. The disease involves widespread motor impairments, such as tremors and rigidity, and cognitive deficits such as dementia and reduced attention.
LRRK2 BAC transgenic lines are a popularly used rodent model of Parkinson’s disease. These animals develop progressive deficits in dopamine circuit function and subsequent progression of motor and non-motor symptoms. LRRK2 BAC transgenic rats show impaired spatial short-term memory as measured by decreased performance on the alternating T-maze test.[19]
In studies with humans, Parkinson’s disease patients show lower short term memory capacity across digit span tests and deficits in short term memory maintenance. Similarly, in “n-back” tasks, a common test of spatial working memory, Parkinson’s disease patients perform below age-matched controls. Levodopa, a precursor to dopamine, was noted to increase performance only on spatial working memory tasks in one study.[20]
Alzheimer’s Disease
Alzheimer’s disease causes the dysfunction and death of neurons involved in learning and memory processes, including the hippocampus and frontal cortex. The disease is associated with mitochondrial alterations, oxidative stress, and the accumulation of neurotoxic amyloid-beta protein that causes widespread neuronal death.
Alzheimer’s disease leads to deficits in the integrated activity of a distributed network of brain regions that includes the prefrontal cortex and hippocampus. In a McGill transgenic rat model of Alzheimer’s disease, the animals demonstrate significant reductions in spontaneous alternation behavior in the Y-maze or T-maze paradigms.[21]
Sleep Deprivation
Sleep is fundamental for optimal physical and mental health as well as overall vitality. Sleep deprivation is associated with a slew of negative health consequences, including disrupted cardiovascular health, immunity, and widespread deficits in cognition. Much of the sleep deprivation studies in behavioral neuroscience have centered around sleep deprivation’s negative effects on memory processing.
In 2006, researchers investigated the effects of 72 hours of REM sleep deprivation in rats using a modified version of the Morris water maze called the place learning set task. In this task, differences in escape latency between two identical trials reflect measures of short-term memory or working memory. The researchers reported a decrease in the learning rate, due to poor spatial short-term memory and slow learning of the task rules.[22]
In one human study, researchers assessed the effects of sleep deprivation on visual short-term memory load using a delayed match-to-pattern task. With concurrent brain imaging, they found that sleep deprivation reduced posterior cortical activation at all levels of visual memory load. The negative effects were attributed to declines in visual attention and/or visual processing. Additionally, the researchers reported that donepezil, an acetylcholinesterase inhibitor, can reduce the decline of visual short-term memory and visual attention in individuals that are vulnerable to the effects of sleep deprivation.[23]
Drugs That Affect Short Term Memory
Although there are a number of drugs that affect short term memory with widely different mechanisms of action, this section overviews four drugs that have been found in human and rodent studies to influence short-term memory. We will discuss two that negatively impairs short term memory, and two that has shown evidence to improve short-term memory.
Alcohol
While “blackouts” caused by binge drinking can cause serious memory loss, chronic alcoholism is also associated with significant impairments in memory and learning. One mechanism through which memory impairments arise in chronic alcohol exposure is thiamine deficiency. When severe, this can produce Wernicke-Korsakoff syndrome, a brain disorder characterized by amnesia, deficits in executive functioning, and delirium. In a study from 1971 of alcoholics who drank for 12-14 days, researchers found that short-term memory is progressively impaired depending on the level of intoxication.[24]
In another study involving a rodent model of alcoholism, rats were given alcohol for 20 weeks. After a 2-month alcohol-free period, short-term memory was evaluated with a discrete-trial temporal-alternation task assessing short-term memory. Alcohol-treated rats showed severe impairments in 50-second long trial retention intervals compared to rats who were not chronically exposed to ethanol.[25]
Benzodiazepines
Benzodiazepines are popularly prescribed drugs that work by enhancing GABA signaling at the GABAA receptor, resulting in its hypnotic, sedative, anticonvulsant, and anxiolytic properties. Apart from its potential addictiveness and associated withdrawal symptoms, frequent, high doses of benzodiazepines are associated with significant memory impairments. This includes disruptions in both memory acquisition and consolidation as well as the generation of anterograde amnesia.[26] One 2004 meta-analysis that evaluated 13 studies in humans found that benzodiazepines produce a widespread effect on several variables of cognition, including deficits in verbal memory, nonverbal memory, attention, and sensory processing.[27]
Modafinil
Modafinil is classified as a “wakefulness-promoting agent” and is thus prescribed primarily for narcolepsy and shift work sleep disorder, but it is increasingly being used off-label for its cognitive-enhancing effects. Modafinil’s arousal-promoting properties are primarily due to its action as a selective and weak dopamine reuptake inhibitor, which indirectly releases orexin neuropeptides and histamine from several subregions of the hypothalamus. One 2003 study from The University of Cambridge involving 60 healthy males found that 100-200mg of modafinil significantly enhanced their performance on common tests of short term memory, including both digit span and visual pattern recognition tasks.[28]
Rodent studies have also pointed towards the short term memory-enhancing effects of Modafinil. One study found that high doses (64mg/kg) of Modafinil produced a delay-dependent increase in spontaneous alternation performance in rats on a T-maze task.[29] Similar dosage ranges used in other studies similarly found performance increases in a delayed nonmatching to position task in a modified water maze.[30]
PRL-8-53
PRL-8-53 is a synthetic cognitive enhancement supplement derived from benzoic acid and phenylmethylamine. Supplementation of PRL-8-53 in rats was able to improve avoidance learning in doses ranging from 5-20mg/kg.[31] In a human study of this nootropic, participants took a memory test consisting of twelve monosyllabic words in a specific order. The test was conducted three times, once immediately after supplementation, a day after supplementation, and four days after supplementation. People over the age of 30 with poor short-term memory showed slight improvements in acquisition and retention of verbal information from supplementation, quantified as a 30-45% improvement in the number of words retained.[32]
Although its mechanism of action is unclear, some researchers suggest possible cholinergic properties and dopamine potentiating effects. No adverse side effects were seen in the human study, and preliminary rodent evidence suggests it is safe. However, more well-designed studies will have to be conducted to validate its beneficial effects on short-term memory.
Conclusion
Short-term memory has become part and parcel of the general study of cognition in psychology and behavioral neuroscience. While most modern research into this topic has honed in on working memory, the two concepts share significant overlap in terms of the neurobiological systems underlying them and the behavioral tasks used to measure them. Short-term memory can be divided into verbal and visuospatial components, and behavioral assessments are designed to capture short-term memory performance in a modality-specific way. Memory span and free recall paradigms can accurately assess verbal spatial memory, while pattern span, Corsi block tapping, and match-to-sample tasks can assess visuospatial memory.
In rodents, match-to-place and delayed alternation tasks can assess spatial short-term memory, while contextual fear conditioning can evaluate short-term recall of fear memories. Understanding short-term memory in tasks such as these can inform novel pharmacotherapies and diagnostics for a wide range of diseases and conditions that negatively affect this memory system.
References
-
- Norris D. (2017). Short-term memory and long-term memory are still different. Psychological bulletin, 143(9), 992–1009.
- Cowan, N. (2001). The magical number 4 in short-term memory: A reconsideration of mental storage capacity. Behavioral and Brain Sciences, 24, 87–114.
- Jonides, J., Lewis, R.L., Nee, D.E., Lustig, C.A., Berman, M.G., & Moore, K.S. (2008). The mind and brain of short-term memory. Annual Review of Psychology, 59, 193–224
- Tulving E, Craik FIM, eds. 2000. The Oxford Handbook of Memory. New York: Oxford Univ. Press
- Fuster JM. The prefrontal cortex—an update: time is of the essence. Neuron. 2001;30:319–333.
- Berryhill ME, Phuong L, Picasso L, Cabeza R, Olson IR. Parietal Lobe and Episodic Memory: Bilateral Damage Causes Impaired Free Recall of Autobiographical Memory. J Neurosci. 2007;27:14415–14423.
- Todd JJ, Marois R. Capacity limit of visual short-term memory in human posterior parietal cortex. Nature. 2004;428:751–754.
- Xu Y, Chun MM. Dissociable neural mechanisms supporting visual short-term memory for objects. Nature. 2006;440:91–95.
- Jeneson, A., & Squire, L. R. (2011). Working memory, long-term memory, and medial temporal lobe function. Learning & memory (Cold Spring Harbor, N.Y.), 19(1), 15–25.
- Das SR, Mancuso L, Olson IR, Arnold SE, Wolk DA. (2015) Short-Term Memory Depends on Dissociable Medial Temporal Lobe Regions in Amnestic Mild Cognitive Impairment. Cereb.Cortex. 26(5):2006-17.
- Kumaran D. Short-term memory and the human hippocampus. Journal of Neuroscience. 2008;28:3837–3838.
- Roy DS, Kitamura T, Okuyama T, Ogawa SK, Sun C, Obata Y, Tonegawa S (2017) Distinct neural circuits for the formation and retrieval of episodic memories. Cell 170:1000–1012.
- J. Kamiński, A. Brzezicka, A. Wróbel (2011). Short-term memory capacity (7 ± 2) predicted by theta to gamma cycle length ratio. Neurobiol. Learn. Mem., 95; pp. 19-23.
- Daniel, T. A., Katz, J. S., & Robinson, J. L. (2016). Delayed match-to-sample in working memory: A BrainMap meta-analysis. Biological psychology, 120, 10–20.
- Qureshi MF, Jha SK (2017) Short-term total sleep-deprivation impairs contextual fear memory, and contextual fear-conditioning reduces REM sleep in moderately anxious Swiss mice. Front Behav Neurosci 11:239.
- Moyer JR., Jr. Brown TH. Impaired trace and contextual fear conditioning in aged rats. Behav Neurosci. 2006;120(3):612–24.
- Della Sala S, Gray C, Baddeley A, Allamano N, Wilson L. (1999). Pattern span: a tool for unwelding visuospatial memory. Neuropsychologia 37: 1189–99.
- Guariglia C. C. (2007). Spatial working memory in Alzheimer’s disease: A study using the Corsi block-tapping test. Dementia & Neuropsychologia, 1(4), 392–395.
- Sloan, M., Alegre-Abarrategui, J., Potgieter, D., Kaufmann, A. K., Exley, R., Deltheil, T., … Wade-Martins, R. (2016). LRRK2 BAC transgenic rats develop progressive, L-DOPA-responsive motor impairment, and deficits in dopamine circuit function. Human molecular genetics, 25(5), 951–963.
- Beato R, Levy R, Pillon B, Vidal C, du Montcel ST, Deweer B, Bonnet AM, Houeto JL, Dubois B, Cardoso F. Working memory in Parkinson’s disease patients: clinical features and response to levodopa. Arq Neuropsiquiatr. 2008;66:147–151.
- Petrasek, T., Vojtechova, I., Lobellova, V., Popelikova, A., Janikova, M., Brozka, H., … Stuchlík, A. (2018). The McGill Transgenic Rat Model of Alzheimer’s Disease Displays Cognitive and Motor Impairments, Changes in Anxiety and Social Behavior, and Altered Circadian Activity. Frontiers in aging neuroscience, 10, 250.
- Colavito, V., Fabene, P. F., Grassi-Zucconi, G., Pifferi, F., Lamberty, Y., Bentivoglio, M., & Bertini, G. (2013). Experimental sleep deprivation as a tool to test memory deficits in rodents. Frontiers in systems neuroscience, 7, 106.
- Chuah LYM, Chee MWL. Cholinergic augmentation modulates visual task performance in sleep-deprived young adults. Journal of Neuroscience. 2008;28(44):11369–11377.
- Tamerin, J. S., Weiner, S., Poppen, R., Steinglass, P., and Mendelson, J. H., 1971. Alcohol and memory: Amnesia and short-term function during experimentally induced intoxication, Amer. J. Psychiat. 127 (12): 1659.
- Walker D.W. and Hunter B.E. (1978). Short-term memory impairment following chronic alcohol consumption in rats. Neuropsychologia; 16, 545-553.
- Beracochea D: Anterograde and retrograde effects of benzodiazepines on memory. ScientificWorldJournal. 2006, 6: 1460-1465.
- Barker MJ, Greenwood KM, Jackson M, Crowe SF: Cognitive effects of long-term benzodiazepine use: a meta-analysis. CNS Drugs. 2004, 18: 37-48.
- Turner DC, Robbins TW, Clark L, Aron AR, Dowson J, Sahakian BJ. Cognitive enhancing effects of modafinil in healthy volunteers. Psychopharmacology (Berl) 2003;165:260–9
- Beracochea D, Cagnard B, Celerier A, le Merrer J, Pere M, Pierard C. First evidence of a delay-dependent working memory-enhancing effect of modafinil in mice. Learning and Memory. 2001;12:375–378
- Ward CP, Harsh JR, York KM, Stewart KL, McCoy JG. Modafinil facilitates performance on a delayed nonmatching to position swim task in rats. Pharmacol Biochem Behav. 2004;78:735–741.
- Hansi, N., A novel spasmolytic and CNS active agent: 3-(2-benzyl-methylamino ethyl) benzoic acid methyl ester hydrochloride. Experientia, 30 (1974) 271–272.
- Hansi, N. and Mead, B., PRL-8–53: Enhanced learning and subsequent retention in humans as a result of low oral doses of new psychotropic agent. Pschopharmacol. 56 (1978) 249–253.