What is Phenibut?
Phenibut, originally known as phenigamma and sometimes written as fenibut, was developed in St. Petersburg, Russia during the 1960’s. Clinically, phenibut is not used in some countries while in others it is used as a medicine for a wide range of purposes. For example, phenibut is clinically used in Russia to relieve instances of fear, tension, and anxiety, and to improve sleep in neurotic patients. Also, it has been given to patients experiencing post-traumatic stress or disorders characterized by depression.[1] However, in the European Union, Australia, or the United States, phenibut is not a licensed medicine used by doctors but is still obtained by the general public via the internet which has been a growing issue of concern lately.[2]
Despite its frequent clinical use overseas, phenibut is a nootropic for which research is scarce. The involved mechanisms of action, as well as the effects on behavior and cognition, remain to be elucidated through the use of animal models and behavioral research.
Reports and case studies have been on the increase, demonstrating that the general public’s consumption of phenibut is on the rise. Subsequently, more instances of toxicity and overdose are also occurring. Therefore, more research is necessary in order to establish exact parameters of how phenibut affects the body and to put in place limitations for safe intake.[2]
Phenibut’s Chemical Composition
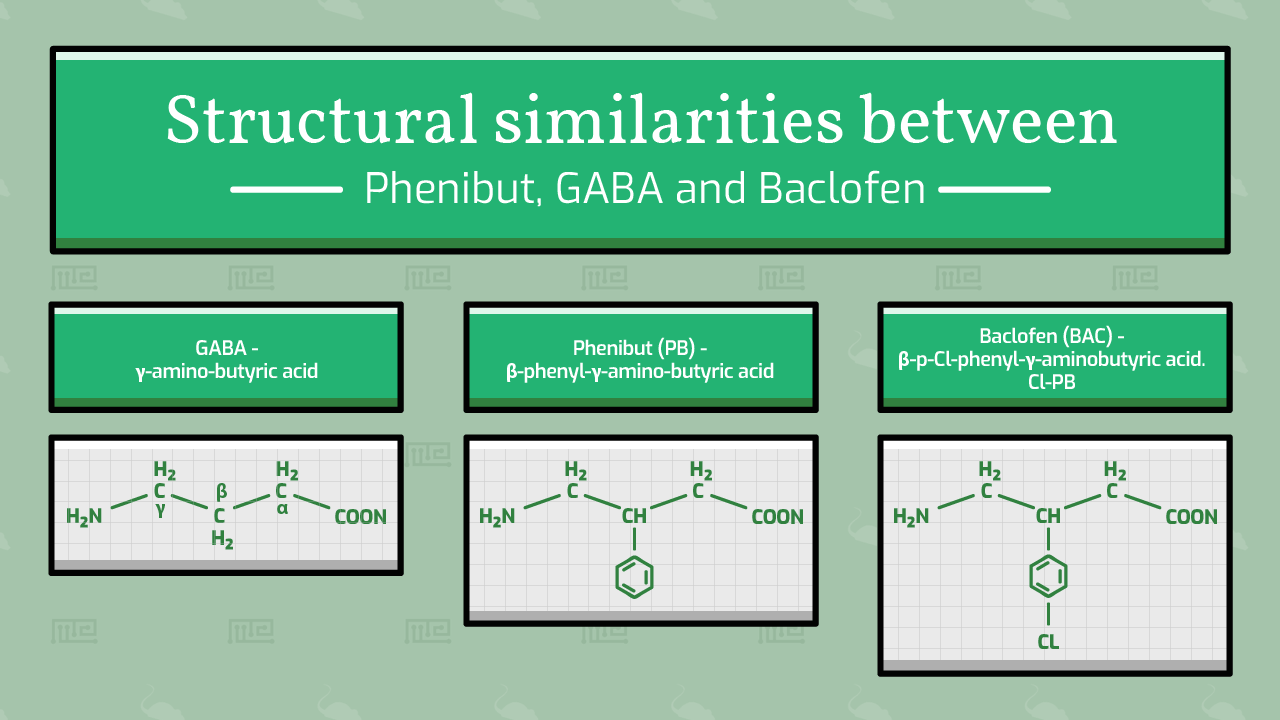
Phenibut, also known as β-phenyl-γ-aminobutyric acid, mimics GABA, the primary inhibitory neuron in the mammalian central nervous system. Due to its similarities to one of the brain’s major neurotransmitters, phenibut is viewed to be able to affect cognition and behavior in ways that GABA would otherwise be involved in.[1]
Furthermore, phenibut has an extra phenyl ring and that is the reason that the molecule is believed to enter the brain so easily.[1]
Structurally, phenibut is similar to baclofen (p-Cl-phenibut), an anxiety-reducing muscle relaxant currently in clinical trials for treating anxiety, and beta-phenylethylamine (PEA), an anxiogenic.[3] Baclofen and phenibut are believed to act on GABAB receptors. Phenibut may have more ability to affect the central GABAB receptors over the peripheral ones and to be able to show some effect on GABAA receptors as well.[3]
Phenibut and the Brain
Since phenibut mimics GABA, it is considered to be a GABA analogue.
Phenibut has been marketed as potent due to its supposed ability to cross the blood-brain barrier (BBB), an action believed to be impossible for GABA. However, recent research is questioning the long-held notion that GABA is unable to cross the BBB. Physiological studies are beginning to note that there are special GABA transporters present in the BBB.[4] Due to the mixed notions and findings, more research is needed to show how phenibut interacts with the brain.
Phenibut’s neuronal effects were first studied using cat cerebral neurons.[5] Some literature exists comparing phenibut’s pharmacological properties with other supplements, such as diazepam or piracetam, and placing a special focus on benzodiazepine receptors, as well as GABAA, GABAB, and dopaminergic receptors.[6]
Phenibut’s General Neuropharmacological Effects
In Russia, phenibut may be used medically and is prescribed to treat clinical cases of insomnia or anxiety. In the United States and Europe, phenibut is predominantly marketed as a nootropic or supplement and is not a pharmaceutical substance.
The following findings provide a general idea of phenibut’s neuropharmacological abilities and should serve as a starting point for future research seeking to expand and verify these effects.
Phenibut and its Anticonvulsant Properties
Phenibut is reported to be able to antagonize hyperkinesis in seizures induced by arecoline-induced tremor and barbiturates, but not seizures induced by bemegride, nicotine, electroshock, strychnine, or pentylenetetrazol.[7]
Also, it is reported that in penicillin-induced seizures, phenibut can reduce hyperactivity in young, immature animals, but not in adult animals.[7] Furthermore, in rats that have been genetically manipulated to be prone to audiogenic seizures, phenibut may be able to potentiate the effects of anticonvulsants such are phenytoin, ethosuximide, trimethadione, and barbiturates.[9]
Typically, when mice or rats are treated with convulsants, they die quickly. This is believed to be due to hypoxia. The rodents that are treated with phenibut apparently have prolonged life and reduced lethality after combining convulsants with phenibut, suggesting a protective effect. Therefore, phenibut may have antihypoxic properties.[6] This finding should be further studied and verified using models of hypoxia.
In one recent study, R-phenibut, which specifically targets GABAB receptors, was shown to have mixed results. Up to 100 mg/kg, it was unable to affect pentylenetetrazole-induced seizures. But, when its anti-nociceptive effects were measured using the formalin-induced paw-licking test, R-phenibut pre-treatment was associated with a dose-dependent decrease in the nociceptive response.[8] Paw licking is a behavioral response to pain and can also be tested using the Hot/Cold Plate apparatus.
Phenibut May Have Tranquilizing Effect by Reducing Anxiety
In one experiment, phenibut or diazepam was given to rodents which were then subjected to the Elevated-Plus Maze. This maze is an industry standard for evaluating anxiety-related behaviors.
Tranquilizing, anxiolytic effects were observed for doses of phenibut at 10 to 25 mg/kg i.p. and for diazepam from 0.5 to 1.0 mg/kg.[6] Such similarities between phenibut and diazepam indicate that phenibut has some of the anxiolytic properties for which diazepam is so well known for.
However, what differentiated the two supplements was the mice’s performance in the social interaction test. In the social interaction test, a popular assessment used in experiments focusing on anxiety triggered by social interactions and encounters phenibut, at doses of 10 to 50 mg/kg i.p., did not increase the duration or rate of contact between the paired mice while diazepam, at 1 mg/kg i.p., did.[9]
Phenibut May Have a Nootropic, Cognition-Enhancing Effect
The same study mentioned above also demonstrated that phenibut can impact memory and facilitate the formation of the conditioned reflex in the Passive Avoidance Test.[5] At doses 5 and 10 mg/kg i.p, phenibut-treated mice had significantly higher latency times, indicating that they could remember which chamber was conditioned with adverse stimuli.[5] While the control mice had a latency of 154 +/- 91 seconds to enter into the dark chamber, the mice receiving phenibut at 5 mg/kg i.p. had a latency of 284 +/- 45 seconds. Furthermore, the total time that the control mice spent in the dark chamber was significantly longer compared to the phenibut-treated animals; 108 +/- 28 seconds and 38 +/- 12 seconds, respectively.[1][6]
Furthermore, in the same study, at doses of 10 and 20 mg/kg i.p., phenibut was found to enhance the mice’s performance in the Rotarod Test. The rotarod test is a measure used to quantify motor performance, balance, and coordination and this may serve as a starting point for translational research. Future studies should take these findings and expand on them, in order to address needs that may exist in the muscle-building or exercise community, since phenibut is often marketed to them as an enhancer or recovery supplement.[2]
Phenibut, due to its structural similarity to baclofen which has muscle-relaxant properties and a tranquilizing effect, is beginning to get attention from scientists that study anxiety and its mechanisms of action and effects on behavior.
Although phenibut is not a clinically prescribed or doctor-recommended medicine in the United States, Australia, and Europe, people will still try to acquire it in order to self-medicate for anxiety. It may be common to hear praise and positive comments about phenibut’s anxiolytic effects (especially through the internet) but that is quickly countered by doctors’ voiced opinions that phenibut can easily be abused without proper regulation. Experts suggest that more research is necessary in order to unveil the exact mechanisms and effects that it has on anxiety and establish proper regulations.[2] Therefore, through the use of animal models, the effects that phenibut has on anxiety can be objectively studied and established.
To assess anxiety-related behaviors, a group of researchers subjected male rats to behavioral training. The rats were trained using the Light/Dark Box to avoid the bright compartment. Using a specially fitted platform which was installed in the chambers to deliver footshock at 45 V, rats would receive a shock when entering the light compartment and the shock would switch off automatically as soon as they went into the dark chamber. Upon establishing a stable reflex response, the rats were re-tested again a few days later upon receiving an injection of phenibut (10 μg).[10]
When compared to the controls with received saline, the phenibut group had much lower scores on the Light/Dark Box anxiety measure. When placed in the experimental apparatus, the phenibut group ended up spending a significantly higher amount of time in the light sector. Such behavioral findings indicate that despite the learned anxiety and conditioned aversion to the light compartment, phenibut still demonstrated anxiolytic properties.
Phenibut: A cause of concern
Phenibut comes with its fair share of side-effects. Several case studies exist reporting this supplement as being addictive, labeling it as a “nootropic of abuse.”[11] Some reports even tie psychosis to the withdrawal episodes experienced while weaning off of phenibut.[12] In fact, the first seizure associated with phenibut intake was detected in 2011 in Sweden.[2] Therefore, the potential side-effects of this nootropic should be addressed by increased research combined with attempts to raise public awareness.
Phenibut is considered to have low acute toxicity. The dosage level that kills 50% of laboratory animals is 700 mg/kg with intraparietal injections (i.p.) in rats and 900 mg/kg i.p. in mice.[1] Still, in the human population, cases associated with seizures[2], psychosis[12], and withdrawal due to substance abuse[11] have been on the increase.
Although phenibut is prescribed in some places in the world for conditions such as anxiety, more information and regulations are necessary in order to ensure consumer safety.
Conclusion
To date, a few studies exist demonstrating the physiological and cognitive effects of phenibut through the use of animal models and mazes. Although some findings do exist, much more research is needed in order to support, solidify, and verify these claims. Research needs to continue in the direction of mood and anxiety disorders, in order to establish concretely (and in-depth) the mechanisms and the behavioral impact that phenibut had.
Despite the fact that phenibut is used clinically in some countries, more behavioral research is needed in order to inform the general public about this nootropic’s true effects
References
- Lapin, Izyaslav. “Phenibut (β‐Phenyl‐GABA): A Tranquilizer and Nootropic Drug.” CNS drug reviews 7.4 (2001): 471-481.
- Van Hout, Marie Claire. “A narrative review of the naturally occurring inhibitory neurotransmitter gamma-aminobutyric acid (GABA) called phenibut in dietary supplements.” Performance enhancement & health 6.1 (2018): 33-35.
- Holloway, Garrett Blake. “Neutraceutical-Based Topical Anxiolytic Agent and Method of Use.” U.S. Patent Application No. 12/949,746.
- Takanaga, Hitomi, et al. “GAT2/BGT-1 as a system responsible for the transport of γ-aminobutyric acid at the mouse blood–brain barrier.” Journal of Cerebral Blood Flow & Metabolism 21.10 (2001): 1232-1239.
- Davies, J., and J. C. Watkins. “The action of β-phenyl-GABA derivatives on neurones of the cat cerebral cortex.” Brain Research 70.3 (1974): 501-505.
- Mehilane, L. S., L. K. Rago, and L. H. Allikmets. “Pharmacology and clinic of phenibut.” Tartu: Izd. TGU (1990).
- Khaunina RA, Prakhie I. B. Effect of phenibut on the action of anticonvulsants. Trudy Psikhonevrol InstBekhtereva 1969;52:382–384 (in Russian with English summary).
- Zvejniece, Liga, et al. “R-phenibut binds to the α2–δ subunit of voltage-dependent calcium channels and exerts gabapentin-like anti-nociceptive effects.” Pharmacology Biochemistry and Behavior 137 (2015): 23-29.
- Lapin, I. P., and M. V. Slepokurov. “The anxiogenic activity of phenylethylamine in the social isolation test on mice.” Farmakologiia i toksikologiia 54.6 (1991): 9-11.
- Talalaenko, A. N., et al. “Neurochemical mechanisms of the dorsal pallidum in the antiaversive effects of anxiolytics in various models of anxiety.” Neuroscience and behavioral physiology 36.7 (2006): 749-754.
- Ahuja, Tania, et al. “Phenibut (β-Phenyl-γ-aminobutyric Acid) Dependence and Management of Withdrawal: Emerging Nootropics of Abuse.” Case reports in psychiatry 2018 (2018).
- Högberg, Lovisa, István Szabó, and Jaan Ruusa. “Psychotic symptoms during phenibut (beta-phenyl-gamma-aminobutyric acid) withdrawal.” Journal of Substance Use 18.4 (2013): 335-338.