Cannabis is the most widely used illicit drug in the world and its usage continues to grow in tandem with a widespread reevaluation of its legal status and social acceptability.[1] While a great deal of research has been conducted on its effects at both a pharmacological and behavioral level, many questions remain unanswered. Here, we will briefly discuss how cannabis and cannabinoids exert their effects on cognition and behavior through the endocannabinoid system. Then, we will outline a novel and pressing field of investigation: the effects of cannabis exposure during pregnancy and how it is modeled in rodents.
The Endocannabinoid System
Cannabis and other synthetic cannabinoids function largely through the endocannabinoid system (ECS), a complex network of receptors, enzymes, and neurotransmitters present in all mammals (in addition to others).[2] Most notably, the primary components of the ECS are the neurotransmitters anandamaide (AEA) and 2-Arachidonoylglycerol (2-AG). These two neurotransmitters are synthesized and degraded by an array of enzymes including diacylglyerol lipase (DAGL), NAPE, fatty acid amide hydrolase (FAAH) and MAG lipase (MAGL). While the primary receptors involved in the ECS are known as the cannabinoid receptors (type 1 and 2, termed CB1R and CB2R), the endocannabinoids have been shown to interact directly with a host of other receptors in the brain including the transient receptor potential channel TRPV1 and the so-called orphan receptors GPR55 and GPR88.[3] Of important note, CB1R have been shown to couple to other types of receptors (known as heterodimers) wherein the regulation of their activity directly regulates the activity of the coupled receptors, as well. Such examples include opioid,[4] dopamine,[5] and serotonin[6] receptors amongst others.
The most commonly discussed target of the ECS is the CB1R. Located presynaptically, CB1R functions as a regulatory mechanism that acts similarly to a rheostat on a light-switch. This means that rather than functioning like post-synaptic receptors which activate or inactivate neurons, CB1R serves as a functional moderator that decreases the release of neurotransmitters from the presynaptic neuron. This occurs when the postsynaptic neuron releases endocannabinoids, which then travel back to the presynaptic neuron and activate CB1R, who in turn reduce neurotransmitter release. This process occurs at nearly every synapse in the brain, which explains why cannabis and other cannabinoid-targeting drugs exhibit such a wide array of effects.[7]
Once the endocannabinoids have done their job, the aforementioned enzymes break them back down into the fatty molecules (lipids) from which they were originally synthesized.[8] These degrading enzymes, FAAH and MAGL, have been the subject of great interest for the development of pharmaceutical drugs due to their higher specificity and comparatively lower side-effect profile than direct modulators of the cannabinoid receptors. There exists a wide range of behavioral effects which occur when these enzymes are inhibited, leading to increased levels of the endocannabinoids. Indeed, FAAH-targeting pharmaceuticals have been proposed for the treatment of everything from anxiety and eating disorders to chronic pain and insomnia.
Although cannabis contains an array of pharmacologically active compounds, most research has focused on the primary psychoactive constituent, tetrahydrocannabinol (∆9-THC or THC). Recent research has expanded the collective understanding of the so-called “entourage effect,” wherein the effects of multiple cannabinoids appears to be more than the sum of its parts. In other words, while THC is indeed responsible for much of the psychoactive effect profile of cannabis, recent research has expanded to consider additional components of the plant such as cannabidiol (CBD) as well as the aromatic terpenes and flavonoids.
Tetrahydrocannabinol
The psychoactive effects of cannabis are most commonly attributed to THC, which acts as a partial agonist at the CB1R.[9] Concentration of THC in cannabis ranges from 1% to more than 25% and has been increasing steadily for the past several decades.[10] However, while most research has focused on this chemical as a model for all of the effects of cannabis, recent attention has been focused on the role of the other several-hundred chemicals present in the plant and how the overall composition of cannabis may alter its effects.[11] As such, researchers should carefully consider their treatment regimen and content when designing cannabis models for research.
Cannabidiol (CBD)
Recently, a great deal of interest has focused on one of the naturally occurring phytocannabinoids present in cannabis, known as cannabidiol (CBD). Ostensibly, CBD is a non-psychoactive cousin of the more commonly known cannabinoid, THC (which is largely responsible for the psychoactive effects of cannabis). However, the mechanism of action by which CBD exerts effects remains unknown.[12] Nonetheless, research into its use for the treatment of multiple disorders from anxiety[13] and psychosis[14] continues and indeed, despite a lack of understanding regarding its mechanism of action, it has recently been approved as a medication for the treatment of severe epilepsy.[15]
Terpenes, Flavonoids and other Phytocannabinoids
While an exhaustive list of the hundreds of compounds present in cannabis is beyond the scope of this article, it is important to note that the essential oils present in the cannabis plant have significant pharmacological effects.[16] Many of these pharmacological effects may indeed have medicinal potential and should therefore not be ignored by researchers interested in understanding cannabis and its full spectrum of effects.[17]
Cannabis during pregnancy: Models and outcomes
While the cognitive and behavioral effects of cannabis in adult rodents have already been described, here we will outline the behavioral findings regarding exposure to cannabis during pregnancy. Importantly, cannabis use during pregnancy is a pressing issue as the rate of consumption continues to increase amongst pregnant women worldwide in conjunction with a global increase in acceptance of the drug.[18] As recently as 2015, survey data indicate that 70% of women believe that there is no harm in using cannabis during pregnancy[19] and medical marijuana practitioners have even begun to suggest the drug as a reasonable medication for pregnant women. However, no small amount of behavioral impact has been observed in the offspring of cannabis-using mothers (for more comprehensive reviews, see [20-22]).
How is in utero cannabis exposure modeled in rodents?
Because cannabinoid receptors are present from the first days following fertilization, many models of in utero cannabis exposure begin very early during the gestational period in rodents.[23] On the other end, neuronal development during the first week of postnatal life in rodents is roughly equivalent to the third trimester of in utero development in humans,[24] leading many researchers to continue cannabinoid exposure during this period in rodent models. Thus, the most common models of in utero cannabis exposure in mice and rats begin at gestational day (GD) 5 and continue until postnatal day (PND) 9-10. While some variations on this model exist (such as beginning at GD1-2, stopping at birth, or continuing until weaning at PND21), these days represent the most common range of drug administration.[25]
While the active ingredient of cannabis, ∆9-THC, seems like the most obvious choice for modelling cannabis exposure during pregnancy, many researchers prefer to use synthetic cannabinoids due to issues with legality and availability of ∆9-THC. The most common amongst these is WIN 55,212-2 (WIN). In this case, WIN is most commonly administered at a dose of 0.5mg/kg either intraperitoneally or via subcutaneous injection. When ∆9-THC is used, dosages are most commonly either 2mg/kg or 5mg/kg.
What are the effects of in utero cannabis exposure?
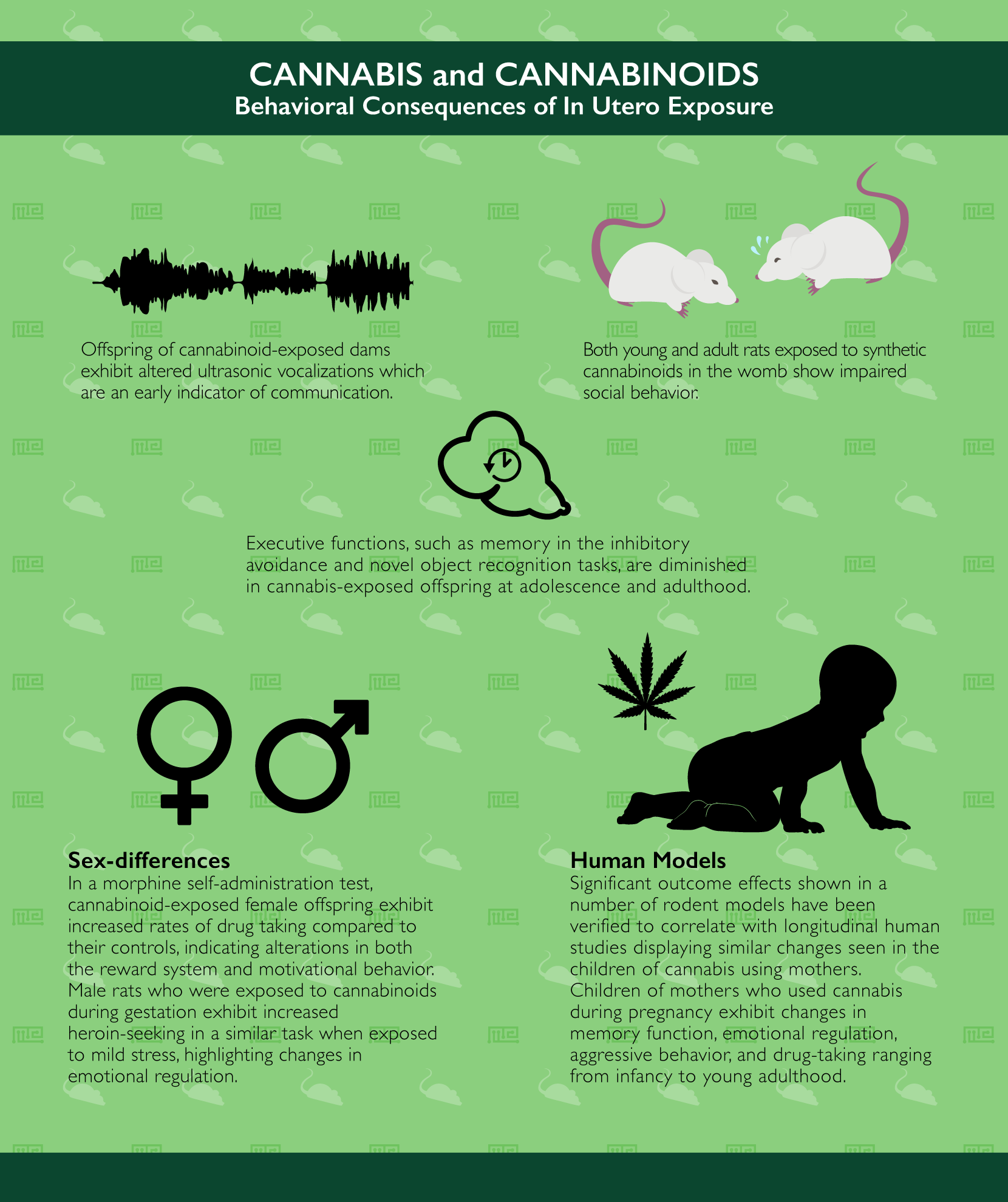
Early in life, the offspring of cannabinoid-exposed dams exhibit deficits in olfaction-based social discrimination[26] as well as ultrasonic vocalizations (an early indicator of communication).[27][28] Later in life, animals who were exposed to cannabinoids in the womb have also been shown to exhibit significant deficits in social interaction. For instance, in a social memory recognition task, male rats exposed to synthetic cannabinoids in the womb show a lack of discrimination between familiar and novel partner rats.[29] Other forms of executive functions are also diminished in cannabis-exposed offspring at adolescence and adulthood such as memory retention in the inhibitory avoidance task[30-32] and in the novel object recognition task.[33]
Interestingly, some of the effects of in utero cannabis exposure in rodent models appear to be dependent on the sex of the offspring. For example, in a morphine self-administration paradigm, exposed females exhibit increased rates of drug taking compared to their age- and sex-matched controls,[34] an effect which is not present in a progressive-ratio style self-administration task,[35] indicating alterations in both the reward system and motivational behavior. On the other hand, male rats who were exposed to cannabinoids during gestation exhibit increased heroin-seeking in a similar task when exposed to mild stress,[36] highlighting changes in emotional regulation.
Clearly, cannabis exposure during pregnancy has a number of significant outcome effects shown in a number of rodent models. These have been verified to correlate with similar changes seen in the children of cannabis using mothers in numerous longitudinal human studies such as the Ottawa Prenatal Prospective Study,[37] the Maternal Health Practices Child Development Study[38] and the ongoing Generation R study.[39] Similarly to the above findings in rodent models, children of mothers who used cannabis during pregnancy exhibit changes in memory function, emotional regulation, aggressive behavior, and drug-taking ranging from infancy to young adulthood.
Conclusions
Cannabis (and other cannabinoids) function through interactions with the endocannabinoid system. Their effects imbue both acute and long-lasting behavioral changes in a variety of domains such as memory, emotional and executive functions such as learning and reward behaviors. Importantly, exposure to cannabis and cannabinoids during pregnancy has significant effects on the behavior of offspring which presents an important field of resarch considering the increased use of the drug worldwide. While many of these effects have been well-described, significant questions remain including a precise definition of the most sensitive periods of exposure, delineation of effects which may be sex-dependent, and what additional behaviors may be impacted following in utero exposure to the drug. Therefore, significant opportunities for future studies are abundant and present a pressing objective for researchers.
References
- Azofeifa, A. et al. (2016) Monitoring Marijuana Use in the United States: Challenges in an Evolving Environment. JAMA 316, 1765–1766
- Russo, E. B. (2016). Beyond Cannabis: Plants and the Endocannabinoid System. Trends in Pharmacological Sciences, 37(7), 594–605.
- Donvito, Giulia; Nass, Sara R.; Wilkerson, Jenny L.; Curry, Zachary A.; Schurman, Lesley D.; Kinsey, Steven G.; Lichtman, Aron H. (31 August 2017). “The Endogenous Cannabinoid System: A Budding Source of Targets for Treating Inflammatory and Neuropathic Pain”. Neuropsychopharmacology. 43 (1): 52–79.
- Hojo, M. et al. (2008) mu-Opioid receptor forms a functional heterodimer with cannabinoid CB1 receptor: electrophysiological and FRET assay analysis. J. Pharmacol. Sci. 108, 308–319
- Przybyla, J.A. and Watts, V.J. (2010) Ligand-Induced Regulation and Localization of Cannabinoid CB1 and Dopamine D2L Receptor Heterodimers. J Pharmacol Exp Ther 332, 710–719
- Viñals, X. et al. (2015) Cognitive Impairment Induced by Delta9-tetrahydrocannabinol Occurs through Heteromers between Cannabinoid CB1 and Serotonin 5-HT2A Receptors. PLoS Biol 13
- Scheyer, A. F., Martin, H. G. S., & Manzoni, O. J. (2017). The Endocannabinoid System in Prefrontal Synaptopathies. In Endocannabinoids and Lipid Mediators in Brain Functions (pp. 171–210).
- Fowler, C. J., Doherty, P., & Alexander, S. P. H. (2017). Endocannabinoid Turnover. Advances in Pharmacology (San Diego, Calif.), 80, 31–66.
- Elphick MR, Egertová M (2001). “The neurobiology and evolution of cannabinoid signalling”. Philosophical Transactions of the Royal Society B: Biological Sciences. 356 (1407): 381–408.
- Mehmedic Z, Chandra S, Slade D, et al. Potency trends of Δ9-THC and other cannabinoids in confiscated cannabis preparations from 1993 to 2008. J Forensic Sci. 2010;55(5):1209-1217.
- Center for Behavioral Health Statistics and Quality (CBHSQ). Treatment Episode Data Set (TEDS): 2003-2013. National Admissions to Substance Abuse Treatment Services. Rockville, MD: Substance Abuse and Mental Health Services Administration; 2015. BHSIS Series S-75, HHS Publication No. (SMA) 15-4934.
- Mannucci, C., Navarra, M., Calapai, F., Spagnolo, E. V., Busardò, F. P., Cas, R. D., … Calapai, G. (2017). Neurological Aspects of Medical Use of Cannabidiol. CNS & Neurological Disorders Drug Targets, 16(5), 541–553.
- Blessing, E. M., Steenkamp, M. M., Manzanares, J., & Marmar, C. R. (2015). Cannabidiol as a Potential Treatment for Anxiety Disorders. Neurotherapeutics: The Journal of the American Society for Experimental NeuroTherapeutics, 12(4), 825–836.
- Rohleder, C., Müller, J. K., Lange, B., & Leweke, F. M. (2016). Cannabidiol as a Potential New Type of an Antipsychotic. A Critical Review of the Evidence. Frontiers in Pharmacology, 7, 422.
- Samanta, D. (2019). Cannabidiol: A Review of Clinical Efficacy and Safety in Epilepsy. Pediatric Neurology.
- Russo, E. B., & Marcu, J. (2017). Cannabis Pharmacology: The Usual Suspects and a Few Promising Leads. Advances in Pharmacology (San Diego, Calif.), 80, 67–134.
- Nuutinen, T. (2018). Medicinal properties of terpenes found in Cannabis sativa and Humulus lupulus. European Journal of Medicinal Chemistry, 157, 198–228.
- Young-Wolff, K.C. et al. (2017) Trends in Self-reported and Biochemically Tested Marijuana Use Among Pregnant Females in California From 2009-2016. JAMA 318, 2490–2491
- Ko, J.Y. et al. (2015) Prevalence and patterns of marijuana use among pregnant and nonpregnant women of reproductive age. Am. J. Obstet. Gynecol. 213, 201.e1-201.e10
- Higuera-Matas, A. et al. (2015) Long-term consequences of perinatal and adolescent cannabinoid exposure on neural and psychological processes. Neurosci Biobehav Rev 55, 119–146
- Trezza, V. et al. (2012) Altering endocannabinoid neurotransmission at critical developmental ages: impact on rodent emotionality and cognitive performance. Front Behav Neurosci 6, 2
- Navarro, M. et al. (1995) Behavioural consequences of maternal exposure to natural cannabinoids in rats. Psychopharmacology (Berl.) 122, 1–14
- Correa, F. et al. (2016) Endocannabinoid system and pregnancy. Reproduction 152, R191–R200
- Spear, L.P. and File, S.E. (1996) Methodological considerations in neurobehavioral teratology. Pharmacol. Biochem. Behav. 55, 455–457
- Schneider, M. (2009). Cannabis use in pregnancy and early life and its consequences: animal models. European Archives of Psychiatry and Clinical Neuroscience, 259(7), 383–393.
- Antonelli, T. et al. (2005) Prenatal exposure to the CB1 receptor agonist WIN 55,212-2 causes learning disruption associated with impaired cortical NMDA receptor function and emotional reactivity changes in rat offspring. Cereb. Cortex 15, 2013–2020
- Trezza, V. et al. (2012) Altering endocannabinoid neurotransmission at critical developmental ages: impact on rodent emotionality and cognitive performance. Front Behav Neurosci 6, 2
- Manduca, A. et al. Sex-specific behavioral deficits induced at early life by prenatal exposure to the CB1 cannabinoid receptor agonist WIN 55,212-2 depend on mGlu5 receptor signaling.
- Bara, A. et al. (2018) Sex-dependent effects of in utero cannabinoid exposure on cortical function. Elife 7.
- Mereu, G. et al. (2003) Prenatal exposure to a cannabinoid agonist produces memory deficits linked to dysfunction in hippocampal long-term potentiation and glutamate release. Proc. Natl. Acad. Sci. U.S.A. 100, 4915–4920
- Campolongo, P. et al. (2007) Perinatal exposure to delta-9-tetrahydrocannabinol causes enduring cognitive deficits associated with alteration of cortical gene expression and neurotransmission in rats. Addict Biol 12, 485–495
- Silva, L. et al. (2012) Prenatal tetrahydrocannabinol (THC) alters cognitive function and amphetamine response from weaning to adulthood in the rat. Neurotoxicol Teratol 34, 63–71
- O’Shea, M. et al. (2006) Repeated cannabinoid exposure during perinatal, adolescent or early adult ages produces similar longlasting deficits in object recognition and reduced social interaction in rats. J. Psychopharmacol. (Oxford) 20, 611–621
- Vela, G. et al. (1998) Maternal exposure to delta9-tetrahydrocannabinol facilitates morphine self-administration behavior and changes regional binding to central mu opioid receptors in adult offspring female rats. Brain Res. 807, 101–109
- González, B. et al. (2003) Effects of perinatal exposure to delta 9-tetrahydrocannabinol on operant morphine-reinforced behavior. Pharmacol. Biochem. Behav. 75, 577–584
- Spano, M.S. et al. (2007) Prenatal cannabis exposure increases heroin seeking with allostatic changes in limbic enkephalin systems in adulthood. Biol. Psychiatry 61, 554–563
- Fried, P.A. (1980) Marihuana use by pregnant women: neurobehavioral effects in neonates. Drug Alcohol Depend 6, 415–424
- Day, N. et al. (1992) The effects of prenatal tobacco and marijuana use on offspring growth from birth through 3 years of age. Neurotoxicol Teratol 14, 407–414
- El Marroun, H. et al. (2008) Demographic, emotional and social determinants of cannabis use in early pregnancy: the Generation R study. Drug Alcohol Depend 98, 218–226