Branched-chain amino acids are essential amino acids involved in protein synthesis and breakdown. They are also indirectly responsible in particular neurotransmitter synthesis. Abnormal levels of branched-chain amino acids have been linked to certain conditions such as insulin resistance, obesity, anxiety and depression. Supplementation of branched-chain amino acids has been used in the treatment of certain diseases, such as phenylketonuria, as well as in brain injury.
What are Branched-chain Amino Acids?
Branched-chain amino acids (BCAAs) are comprised of valine, leucine and isoleucine, which constitute for three of the nine essential amino acids in humans since they cannot be synthesized by the body.[1] These are the only three of the twenty-one amino acids to contain branched R groups, and they account for one-third of dietary essential amino acids and 20% of the total protein content. Since humans are not able to synthesize these amino acids, they must be obtained from food such as brown rice, lentils, chickpeas, lima and baked beans and soy proteins. They can also be acquired by eating animal protein, such as chicken, beef and fish, as these animals have a diet that includes these essential amino acids.[2]
BCAA Catabolism
Early studies using ex vivo assays on extracts from various tissues have indicated that BCAAs do not enter the liver first, unlike other amino acids.[1] This is thought to be due to the lack of BCAA transaminase (BCAT), which is the first enzyme in the catabolic pathway of BCAAs. They undergo transamination to form the corresponding branched-chain keto acids (BCKAs) as well as glutamate with the help of BCAT in the kidneys, muscle, adipose tissue and the heart.[3] In this reversible reaction, the amino group from the BCAA is transferred to α-ketoglutarate to form glutamate which will be used as a substrate for ammonia detoxification to glutamine or as the amino group source to produce alanine from pyruvate.[3] After the transamination reaction, the BCKAs are transported back into circulation and enter the liver, primarily, to undergo oxidation with the help of branched-chain α-keto acid dehydrogenase (BCKDH) and form acyl-coenzyme A (acyl-CoA). The resultant acyl-CoA is oxidized further by other enzymes to produce either acetyl-CoA and acetoacetate (if the BCAA was leucine), succinyl-CoA (if the BCAA was valine) or acetyl-CoA and succinyl-CoA (if the BCAA was isoleucine). These products are available to enter the tricarboxylic acid cycle for the synthesis of adenosine triphosphate (ATP).
Additional in vivo isotropic tracing has reported that numerous peripheral organs are involved in the oxidation and breakdown of BCAAs.
The Effect of BCAAs on Obesity
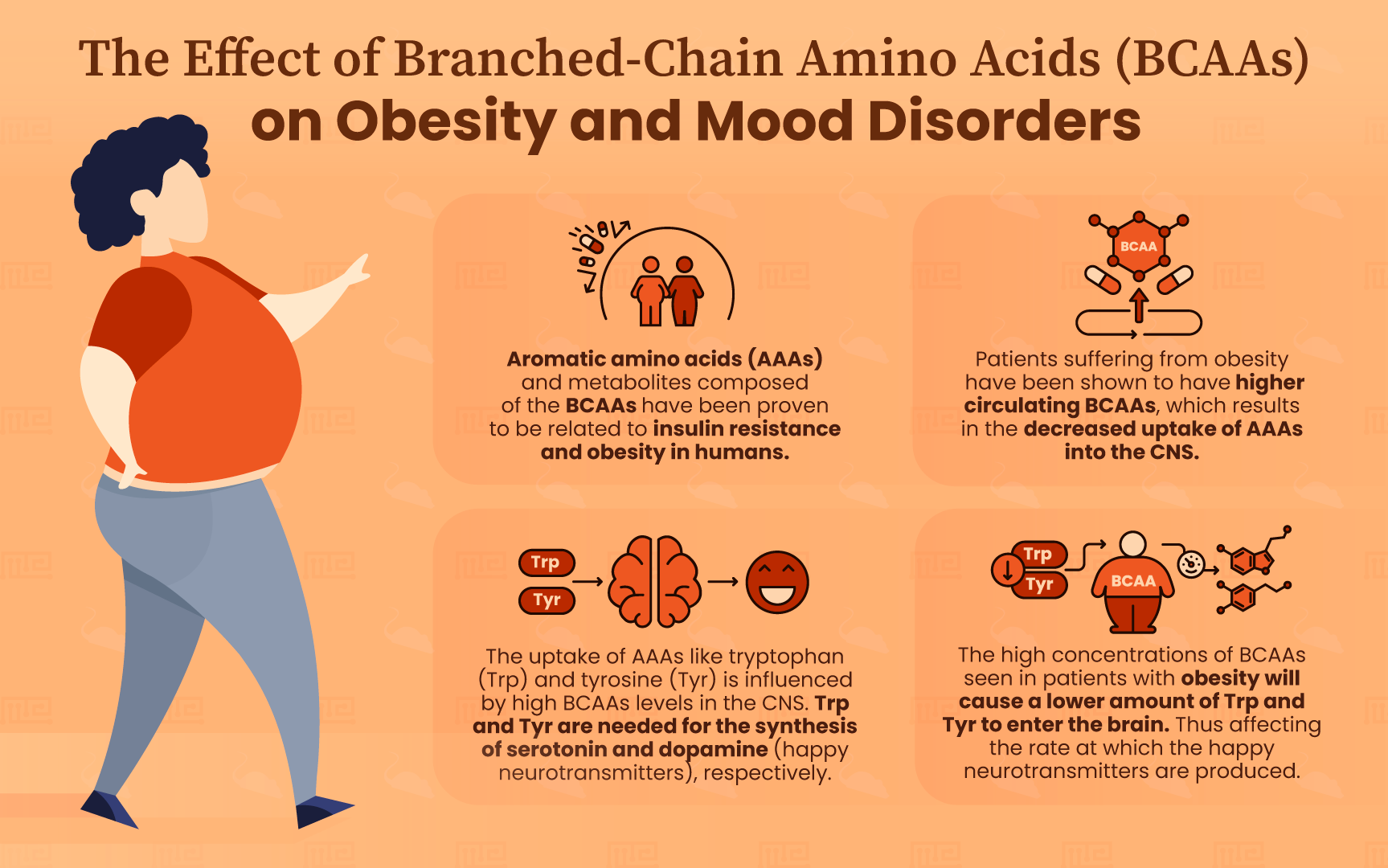
Aromatic amino acids (AAAs) and metabolites that are composed of the BCAAs have been proven to be related to both insulin resistance and obesity in humans.[4] Felig et al[5] were one of the first to report a relationship between BCAAs and obesity in 1969 and this relationship was confirmed by She et al[6]. Patients suffering from obesity have been shown to have higher levels of circulating BCAAs.[1] Not only are the levels of BCAAs and their derived intermediates, BCKAs, elevated in the blood plasma of patients with type 2 diabetes, obesity or insulin resistance; but they are also very strong predictors for risk of developing these conditions, regardless of ethnicity and age.
The Relationship between BCAAs, Obesity and Mood Disorders
Studies have shown that there is a high frequency of mood disorders observed in patients suffering from obesity. Patients who receive treatment for obesity and suffer from depression often experience an alleviation in their depressive symptoms, highlighting the relationship between the two conditions.[4] There is growing interest in this relationship since little is known about the underlying mechanisms linking them.
Dopamine (DA) and serotonin (5-HT) are neurotransmitters known as the “happy hormones” as they both play vital roles in mood and mood disorders. 5-HT is responsible for regulating mood and processing emotions, whereas DA is essential in motivation and reward.[7] DA is synthesized from tyrosine (Tyr) and 5-HT from tryptophan (Trp), and they rely on these AAAs’ transportation into the central nervous system (CNS) to sustain their synaptic activity and levels.[4]
BCAAs travel into the CNS from the blood by passing through the blood-brain barrier with the help of the large neutral amino acid transporter 1 (LAT1).[4] The LAT1 is responsible for the transportation of all large neutral amino acids (LNAAs), including BCAAs and AAAs, and this system is saturated at physiological amino acid concentrations.[4] Uptake of amino acids in either one of these groups will be competitive with the uptake of that from a different group. Therefore, an increased amount of BCAAs, as seen in individuals suffering from obesity, will result in a decreased uptake of AAAs into the CNS. This will ultimately affect the rate at which neurotransmitters are produced.
With the knowledge of this competitive behavior between AAAs and BCAAs uptake into the brain, it is evident that the higher concentrations of BCAAs seen in patients with obesity will cause a lower amount of Trp and Tyr to enter the brain. This will ultimately affect the rate at which 5-HT and DA are produced. Abnormal levels of 5-HT and DA have been linked to mood disorders such as depression; with decreased levels of DA causing symptoms associated with depression and reduced levels of 5-HT being a risk factor of developing depression.[8]
The Effect of BCAAs on Anxiety
As mentioned before, the uptake of BCAAs will affect 5-HT synthesis and availability. Low levels of 5-HT have been linked to anxiety disorders.[8] Coppola et al set out to determine the level of anxiety in rats as a relationship to BCAA concentration.[4] Rats were divided into four groups based on the diet they received.
These groups were:
- LF group which received a low-fat, high-sucrose diet
- LF/BCAA group which received the same diet as the LF group, but with supplemented BCAAs
- HF group which received a high-fat diet
- HF/BCAA group which received the same diet as the HF group, but with supplemented BCAAs
The researchers evaluated the level of anxiety in rats by testing them with an Elevated Plus Maze (EPM) after being on their respective diet for nine weeks. No significant differences were noticed in food intake between the LF and LF/BCAA or the HF and HF/BCAA groups, therefore indicating that BCAA supplementation does not affect the taste of food. The researchers observed that the rats in the LF/BCAA and HF/BCAA groups spent less time in the open arms of the EPM when compared to the LF and HF groups respectively.[8] The decreased exploration observed by the mice on BCAA supplementation suggests that they experienced a higher level of anxiety than the other two groups. This result indicates that a higher level of BCAAs may cause anxiety in rats.
The Effect of BCAAs on Cognition
BCAA Supplementation as Treatment for Maternal Phenylketonuria
Hyperphenylalaninemia is a condition in which the blood phenylalanine levels of an individual exceed the upper limit range of 2mg/dL or 120µmol/L but are below the standards for phenylketonuria (PKU). [9] PKU is a condition characterized by very high levels of circulating phenylalanine that is caused due to the patient’s body not being able to metabolize the amino acid. This condition may cause nerve and brain damage in the patient as a result of the elevated phenylalanine levels. Studies have indicated that 95% of women suffering from PKU and who have serum phenylalanine levels of more than 20mg/dL may give birth to at least one child with mental impairments. [10] This phenomenon is due to transplacental hyperphenylalaninemia.
Phenylalanine shares a common transport carrier with other LNAAs for transport into the CNS, and it is observed that placental carrier-mediated transport of amino acids is similar. Therefore, it has been suggested and demonstrated that increasing the level of one member of the LNAA group will inhibit the transport of another member into the brain.[10] Researchers have been interested in utilizing this relationship in the treatment of maternal PKU, especially in an attempt to prevent cognitive impairments in their offspring.
Vorhees et al conducted experiments to establish if a particular mixture of BCAAs will save CNS development in the progeny of pregnant rats with induced PKU.[10]
The authors divided the pregnant rats into four groups and began to issue their experimental diets on embryonic days 5-15:
- A PKU group that were fed a diet consisting of additional L-phenylalanine and p-chlorophenylalanine (PCPA), a phenylalanine hydroxylase inhibitor
- A PKU-VIL group that received the same diet as the PKU group, but supplemented with added valine, isoleucine and leucine (VIL)
- A control group, PFC, that were fed a base diet
- A control group, PFC-VIL, that were fed the base diet with additional VIL
Neither PCPA nor L-phenylalanine alone induce any noticeable long-term effects on behavior and water maze performance.
The progeny of the pregnant rats were subjected to various tests in order to determine the effects of VIL supplementation on cognition.
The researchers reported a significant increase in path length during the Morris Hidden Platform Maze, to test spatial learning and memory, for both the PKU and PKU-VIL groups in the female rats when compared to their control groups. Therefore they concluded that no improvement was observed after treatment with VIL.[10] The researchers were not able to acquire results for re-acquisition or evaluations without a platform, thus the effect of PKU and the VIL treatment on memory could not be tested with this maze. No significant difference was observed in males; therefore, it was not possible to test the effect of VIL on male rats in this maze.
After the Morris Hidden Platform Maze, the researchers measured food consumption by the rats, after which they restricted food to 60% for postnatal days 86-93 and during testing with the Eight Arm Radial Maze. The maze was enclosed and rested on the floor, rather than being open and elevated, and a rat had to travel to the end of an arm for it to be considered entered. The PKU group obtained fewer reinforcements when compared to the PFC and PFC-VIL groups. In contrast, the PKU-VIL group received significantly more reinforcements when compared to the PKU group and approximately the same as the PFC and PFC-VIL groups. These results support the suggestion that supplementation of VIL in pregnant rats suffering from PKU may improve the detrimental effects of hyperphenylalaninemia observed in their offspring.
The researchers also tested the progeny rats with the Cincinnati Water Maze and observed significantly more errors in the PKU group than the PKU-VIL group for path A; however, there was no substantial difference in the number of errors obtained for the two groups on path B. It was theorized that the difference in results is due to path A being less complicated and more forced-choice than path B; therefore suggesting that supplementation with VIL has an ameliorative effect on problem-solving of a less demanding task. No significant benefit of VIL supplementation seems to be present for a more challenging problem; however, Vorhees et al did not establish if the ratio and dose of VIL used were optimal. Therefore future studies can experiment with these parameters to perhaps achieve an improvement in path B.
It was also reported that supplementation of VIL improved cognitive performance in the PKU-VIL group without much of an increase in VIL levels. A promising result that suggests sustained periods of elevation or higher doses of VIL may provide even better outcomes in cognitive function.
BCAA Supplementation as Treatment for Cognitive Impairments in Traumatic Brain Injuries
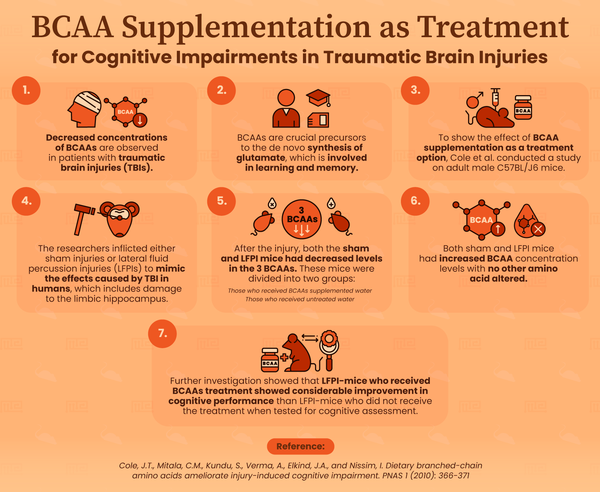
Patients with traumatic brain injuries (TBIs) have been observed to have decreased concentrations of BCAAs.[11] The biological process of removal of released glutamate from the synaptic cleft and successive amidation to glutamine that will be returned to the neuron is inefficient. Therefore, de novo synthesis of glutamate is vital to ensure synaptic neurotransmitter pools are maintained and provides about 40% of the glutamate that will be released from the synapse. BCAAs are crucial precursors to de novo synthesis of glutamate with approximately 50% of brain glucose-containing nitrogen being acquired from BCAAs.[11]
Cole et al conducted an experiment on adult male C57BL/J6 mice to determine if supplementation of BCAAs will improve cognitive impairment caused by TBI.[11] The researchers either inflicted sham injuries or lateral fluid percussion injuries (LFPIs) to mimic the effects caused by TBI in humans. An LFPI damages the limbic hippocampus, a brain structure that is often damaged in TBI and is involved in memory and learning. After inflicting the mice with the injuries (sham or LFPI), the BCAAs concentration decreased. The researchers noted that these were the only three of the eighteen amino acids quantified to have a substantial change in concentration after the injury. Two days later the mice (sham and LFPI) received either untreated water (the control) or water containing all three BCAAs (the treatment). After five days on this diet the levels of BCAA in the ipsilateral hippocampus of the test mice were compared. The researchers found that the mice who received LFPIs and were treated with BCAAs presented with increased BCAA concentration levels that were not much different to the levels in the sham mice. The treatment did not alter the other amino acids tested.
The researchers went on to prepare another group of mice and test them using a hippocampal-dependent cognitive assessment with retrograde and anterograde fear conditioning.[11] The LFPI-mice who received treatment with BCAAs showed considerable improvement in cognitive performance that was not observed in the LFPI-mice who did not receive the treatment. These mice also presented with similar net synaptic efficacy levels compared to the sham-mice in both the dentate gyrus and area CA1.[11]
Another group of mice were prepared; this time to prove that cognitive improvement observed in the previous experiments was specific to the BCAA treatment. These mice were given a diet supplemented with phenylalanine, another LNAA, and had no increase in cognitive performance nor any restoration of network excitability in the hippocampal subregions.[11]
Conclusion
It is of vital importance to ensure that the concentration of circulating BCAAs are kept at normal levels as abnormal levels have been associated with conditions such as depression, obesity, anxiety and insulin resistance. Varying levels have also been shown to influence the entry of other LNAAs into the brain, causing a possible effect on reliant neurotransmitters.
BCAAs are essential dietary amino acids which have been shown to improve symptoms of cognitive impairment observed in patients with TBI and the offspring of mothers with PKU. More research should be done to discover more positive effects of these amino acids, especially on memory and learning.
References
- Siddik, A.B. and Shin, A.C. Recent progress on branched-chain amino acids in obesity, diabetes and beyond. Endocrinology and Metabolism 3 (2019): 234-246
- “Branched-Chain Amino Acids.” WebMD, https://www.webmd.com/vitamins-and-supplements/branched-chain-amino-acids-uses-risks
- Holeček M. Branched-chain amino acids in health and disease: metabolism, alterations in blood plasma, and as supplements. Nutrition and Metabolism 33 (2018) doi: 10.1186/s12986-018-0271-1
- Coppola, A., Wenner, B.R., Ilkayeva, O., Stevens, R.D., Maggioni, M., Slotkin, T.A., et al. Branched-chain amino acids alter neurobehavioral function in rats. American Journal of Physiology, Endocrinology and Metabolism (2012) doi: 10.1152/ajpendo.00373.2012
- Felig, P., Marliss, E., and Cahill, G.F. Plasma amino acid levels and insulin secretion in obesity. The New England Journal of Medicine 281 (1969): 811-816
- She, P., Van Horn, C., Reid, T., Hutson, S.M., Cooney, R.N., and Lynch, C.J. Obesity-related elevations in plasma leucine are associated with alterations in enzymes involved in branched-chain amino acid metabolism. American Journal of Physiology, Endocrinology and Metabolism6 (2007): 1552-1563
- “What’s the Difference Between Dopamine and Serotonin?” Healthline, https://www.healthline.com/health/dopamine-vs-serotonin
- “Dopamine and serotonin: Brain chemicals explained.” Medical News Today, https://www.medicalnewstoday.com/articles/326090.php
- “Hyperphenylalaninemia.” Medscape, https://emedicine.medscape.com/article/945180-overview
- Vorhees, C.V., Acuff-Smith, K.D., Weisenburger, W.P., Minck, D.R., and Berry, H.K. Branched chain amino acids improve radial-arm maze acquisition and water maze forced-choice learning in rat offspring exposed in utero to hyperphenylalaniemia. Neurotoxicology and Teratology 1 (1992): 35-41
- Cole, J.T., Mitala, C.M., Kundu, S., Verma, A., Elkind, J.A., and Nissim, I. Dietary branched chain amino acids ameliorate injury-induced cognitive impairment. PNAS 1 (2010): 366-371