Introduction to Aging
Aging is the progressive degeneration of physiological structure and function. Although people are living longer on average (thanks to improved medical standards), the world’s aging population is rapidly increasing. Thus, the health community is observing a subsequent increase in age-related chronic diseases.
Notably, aging is the primary risk factor for neurodegenerative diseases such as Alzheimer’s disease (AD) and Parkinson’s disease (PD).[1][2] In fact, neurodegenerative diseases are often said to be diseases of “brain aging.”
If aging seems to be the chief culprit of debilitating chronic diseases and cognitive decline in later life, is it a better idea to study aging as a disease model and therapeutic target instead of focusing on a specific form of neurodegeneration? This article explores how the field has indeed been moving towards treating aging and shown promising results.
Hallmarks of Aging
Before we dive into what behavioral research has to say about aging and neurodegenerative diseases, let’s take a quick look at key pathophysiological characteristics of aging.
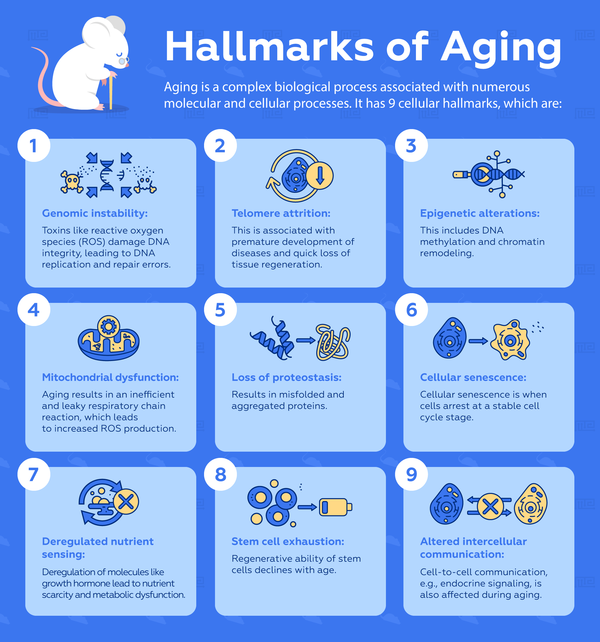
Aging is a complex biological process that is associated with a multitude of molecular and cellular processes. Unlike some genetic disorders, it is nearly impossible to have just a few biomarkers that researchers can rely on for diagnosis or treatment. Therefore, aging is an umbrella term for the following 9 cellular hallmarks:[3]
- Genomic instability: Genomic instability occurs when the integrity of DNA is continuously challenged by toxins such as reactive oxygen species (ROS), thereby inducing DNA replication and repair errors.
- Telomere attrition: Telomere attrition is problematic because it is associated with premature development of diseases and rapid loss of tissue regenerative abilities.
- Epigenetic alterations: Epigenetic alterations including DNA methylation and chromatin remodeling accompany aging and have been shown to reduce longevity in animal models.
- Mitochondrial dysfunction: As cells age, the respiratory chain reaction that is responsible for supplying cellular energy becomes less efficient and leaky. As a result, there is increased production of toxic ROS which ultimately exacerbates aging and cellular damage.
- Loss of proteostasis: Cells use quality control mechanisms to ensure proteins are properly folded and stable. Loss of proteostasis during aging can lead to misfolded and aggregated proteins that contribute to pathologies in neurodegenerative diseases.
- Cellular senescence: Cellular senescence is when cells arrest at a stable cell cycle stage, a phenomenon that becomes more common with aging. It is postulated that this process may be a protective mechanism when cells accumulate genetic damage.
- Deregulated nutrient sensing: Nutrient-sensing molecules, including the growth hormone, IGF1 and insulin often become deregulated as a consequence of other aging-related cellular damage. They lead to nutrient scarcity and metabolic dysfunction, which exacerbates aging.
- Stem cell exhaustion: Stem cell proliferation is essential for the maintenance of an organism. This regenerative capacity declines with aging, likely as a result of multiple types of aging-related cellular damage.
- Altered intercellular communication: In addition to changes that happen within cells, important cell-to-cell communication such as endocrine signalling is also affected during aging.
The establishment of this framework has meant that aging research is complicated, versatile and collaborative. Researchers can step in from multiple angles, much like how a 2000-piece puzzle is put together by a group of friends.
Studying the Role of Aging in Neurodegenerative Diseases
Among the 9 hallmarks of aging, AD and PD involve all 9, Huntington’s diseases (HD) involves 7, amyotrophic lateral sclerosis (ALS) involves 3, and ataxia telangiectasia (AT) involves 2.[2] It is obvious that aging plays a role in neurodegenerative diseases, but the question is: what are the sequences of these molecular events and how do they unfold? Do aging processes cause neurodegeneration, and if so, how?
Take AD as an example. It is the most common neurodegenerative disease, characterized by memory loss, learning deficits, and behavioral changes. Yet, the only way to diagnose it is through standardized questionnaires and assessing cognition while ruling out other possible causes. Ultimately, the final diagnosis of AD is established in post-mortem histology when Aβ plaques and Tau neurofibrillary tangles are detected at high levels.[4] Although these pathologies can be screened for during one’s lifetime through neuroimaging or cerebrospinal fluid analyses, they can only help to confirm the diagnosis when symptoms are already present.[5]
Despite the fact that these cardinal pathological features have been known for over a century, the root causes of AD, early diagnostic tools and effective treatment options remain elusive.
Is Aging a Key Piece of the AD Puzzle?
After decades of research focused on Aβ plaques and Tau, scientists have now realized that they are likely just middle steps in complicated cascades of events in the progression of brain aging. To develop effective treatments, we need to know which comorbidities contribute to brain aging in the first place. Ultimately, therapies will probably need to target multiple processes to inhibit primary disease pathogenesis, as well as secondary downstream responses.[4]
Aging as a Pathophysiological Process
In fact, a number of aging-related changes do manifest earlier and are likely to play causal roles in developing AD.[1][2] Today, there is enough cumulative data to support the notion that pathogenesis begins with aging processes that promote Aβ and Tau accumulation, which in turn exacerbate hallmarks of aging.
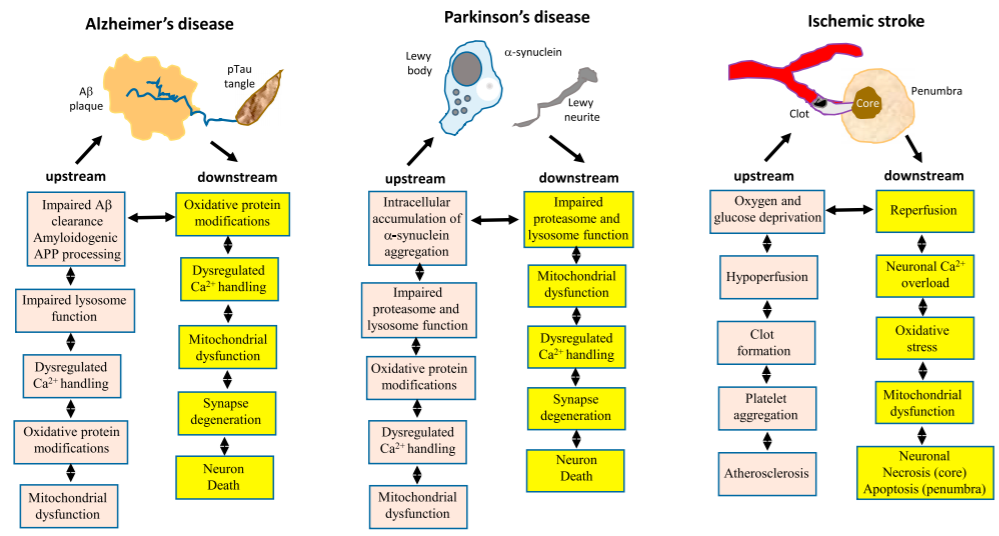
One specific example of the roles of aging in AD pathogenesis is the interrelated changes among lipid peroxidation, oxidative stress, Ca2+ homeostasis, and Aβ pathology. First, proteins modified by lipid peroxidation accumulate in the brain during aging and increase the production of an Aβ precursor. Aβ then accumulates on neuronal membranes generating ROS and more lipid peroxidation. In this process, membrane-related functions such as glucose transport and Ca2+ homeostasis are disrupted, rendering neurons vulnerable to damage and metabolic dysfunction. At the same time, the vulnerable neurons may become even weaker by other hallmarks of aging such as deficits in DNA repair and mitochondrial functions.
Apart from AD, the hallmarks of aging act upstream and downstream of the disease-defining features of PD and ischemic stroke in similar fashions.
Source: Review by Mattson and Arumugam, 2018[1]
In summary, emerging evidence now supports complicated interactions between aging-related processes and the progression of neurodegenerative diseases. It is clear that a specific pathology-based etiology such as the Aβ hypothesis needs to be revised. In light of these findings, the disappointing phase III clinical trials of Solanezumab, an Aβ-clearing medication, in patients with mild AD seems to make sense.[6] Would scientists have a better chance at combating AD by therapeutically targeting more upstream aging-related processes instead?
Targeting Aging to Treat Neurodegenerative Diseases
While targeting aging for therapeutic intervention sounds promising, we first need to address a problem: multiple hallmarks of aging are involved in the pathophysiology of neurodegenerative diseases and they never occur in isolation.
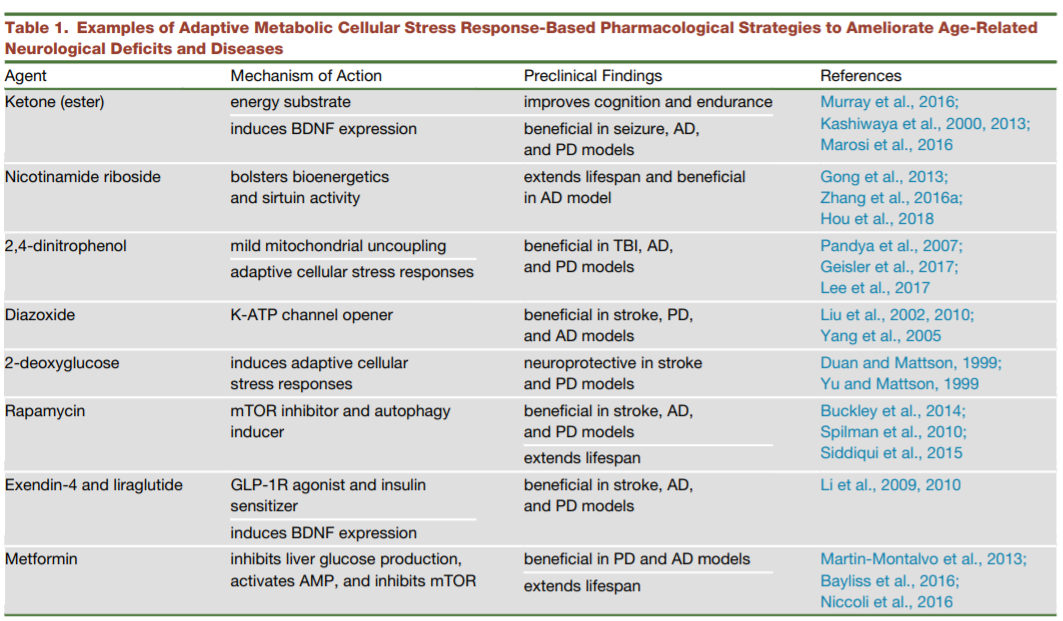
In fact, various aging-related cellular processes and molecules have been targeted for pharmacological interventions for neurodegeneration, including metabolic pathways, oxidative stress, autophagy, stem cell proliferation, telomere and mitochondrial functions.[1] The results are largely promising as many approaches or molecules have shown neuroprotective effects, including caloric restriction, antioxidants, and anti-inflammatory agents. Some therapeutics are now in active phase II/III clinical trials, such as Nicotinamide (vitamin B3) .
Metabolic factors affect all hallmarks of aging
Notably, accumulating evidence suggests an overarching role of metabolic factors that seem to accelerate all hallmarks of aging, which may help address the issue of co-occuring hallmarks of aging observed in neurodegenerative diseases.[1]
In our modern society, metabolic morbidities including obesity, dyslipidemia and insulin resistance, are prevalent. These metabolic morbidities are predominantly due to a poor lifestyle that’s characterized by a daily energy surplus and lack of physical activity. In turn, they increase the risk of all major age-related diseases.
For example, brain imaging studies have found reduced grey and white matter volumes in metabolically morbid individuals. Controlled experiments in animal studies have also shown that a chronic energy surplus is detrimental to brain structure and function.[1]
One of the consequences of metabolic defects is the excessive production of ROS. ROS react with proteins, and nucleic acids, ultimately causing damage to the body and mind. They promote accelerated senescence and contribute significantly to the metabolic dysfunctions as well as aging.[7]
Using Aging Metabolic Factors to Treat Neurodegenerative Diseases
Animal models that mimic the modern day metabolic dysfunctions have revealed many aging-related cellular signals, including: oxidative damage, impaired autophagy, dysregulated neuronal network activity and stem cell neurogenesis.[1] Downstream of these effects are an accelerated development of pathologies involved in neurodegenerative diseases, such as Aβ plaques and Tau accumulation in AD. Therefore, it is not surprising that antioxidants have proven to be powerful anti-aging agents that are now being actively investigated for their potential to treat and manage neurodegenerative diseases.
If brain aging, at least in part, results from metabolic dysfunction, metabolic modifications should be able to curb aging processes. In fact, emerging evidence supports using lifestyle changes to do this. For example, creating metabolic changes that lead to intermittent depletion of liver glycogen storage (such as caloric restriction), thereby promoting adaptive activities in the brain.[1] These include increased neuronal network activity, upregulation of the expression of trophic factors, neural plasticity, and resilience during aging. Thus, positive outcomes in the brain can be observed as a result of metabolic-related interventions.
Apart from lifestyle changes, therapeutics that modify metabolism-accelerated aging in a similar fashion to caloric restriction and exercise have shown promising results. To this end, a number of adaptive metabolic agents have been tested, some of which are effective in ameliorating brain aging in animal models.[1]
Source: review by Mattson and Arumugam, 2018[1]
Behavioral Tools Used in Animal Studies of Brain Aging
Thanks to the meticulous basic research in animal models, cellular and molecular level explanations for the relationships between metabolism, aging, and neurodegenerative diseases are rapidly expanding. These are significant advances in life science as the results are often relevant to all age-related diseases.
Future research using aging as a disease model and therapeutic target holds promise for improving whole-body health, and especially aging-induced cognitive decline. Since most neurodegenerative diseases are diagnosed primarily based on behavioral symptoms, and improvements in behaviors are the ultimate goal of therapeutic interventions, having appropriate behavioral assays is crucial.
This next section demonstrates how researchers can use behavioral assays in an aging mouse model to study neurodegeneration. There are a variety of animal models for brain aging as different hallmarks of aging can be induced by different approaches.[6]
For the sake of introducing the behavioral assays that can be applied to any aging model, we next use two models as examples in the table below, followed by brief descriptions of the behavioral assays used. No one model can fully recapitulate brain aging, and behavioral tools are useful in determining what disease aspects are manifested in different models. From there, behavioral tools continue to be useful for studying disease mechanisms and therapeutic efficacies.
| Behavioral assays | Senescence-accelerated-prone mouse 8 (SAMP8) model | d-galactose induced aging model
|
| Active and passive avoidance task | Impaired performance[10]
Improved performance when supplemented with long-term probiotics[11] |
|
| Morris water maze (MWM)
|
Impaired performance[10][12]
|
Impaired performance[13]
Impaired performance in d-galactose treatment, but rescued with melatonin[14]
Improved performance with ECGG, an antioxidant from green tea[15] |
| T-maze
|
Impaired performance[10]
|
|
| Y-maze
|
No change in groups supplemented with long-term probiotics[11] | Impaired performance in d-galactose treatment, but rescued with melatonin[14] |
| Elevated plus maze
|
Increased anxiety level[16]
Increased anxiety level in old SAMP8 mice, but rescued with fisetin diet[17]
|
Atorvastatin, a cholesterol lowering drug, reduced anxiety-like behaviors[18] |
| Barnes maze
|
Impaired performance in old SAMP8 mice, but rescued with fisetin diet[17]
Improved performance when supplemented with long-term probiotics[11] |
|
| Object recognition test
|
Impaired performance[12]
Impaired performance in old SAMP8 mice, but rescued with fisetin diet[17]
|
Impaired performance[13]
Improved performance with Atorvastatin[18] |
| Open field test | Impaired performance in old SAMP8 mice, but rescued with fisetin diet[17] |
Active and Passive Avoidance Task
The Active and passive avoidance task assess experience-based learning. Passive avoidance is when animals learn to avoid an aversive event by suppressing a behavior. On the other hand, active avoidance is when the animals have to avoid the aversive event by performing a learned behavior.
Aging is associated with compromised cognition and learning. Thus, researchers may use this task to specifically assess how learning that depends on experience is affected by aging.
Morris Water Maze (MWM)
The MWM is a test for experience-based learning, one that relies on spatial memory. The MWM is based on an animal’s desire/motivation to escape a pool of water by finding a hidden platform.
In aging rodents, spatial memory is not as intact as in younger mice.
T-Maze
The T-Maze is widely used as a test for spatial recognition. The maze contains two arms (a left and right arm) for rodents to explore and seek food rewards.
If the rodent has normal working memory, it will naturally explore the un-visited arm instead of the same arm that it visited before, after a successful retrieval of food. In aging mice, working memory is likely to be impaired.
Y-Maze
The Y-Maze is built on the concept of a T-maze and is also a test for spatial recognition, memory, and cognition. Similarly, it can assess whether working memory is affected by aging, but can be a preferred choice as the design makes turning more gradual and learning faster.
Elevated Plus Maze
The Elevated Plus Maze is a widely used test for assessing anxiety-like behavior. It is based on rodents’ natural aversion for open and elevated areas.
Some aging mouse models show increased anxiety behavior, and can be used to test behavioral changes after a treatment intervention.
Barnes Maze
The Barnes Maze is another apparatus for assessing learning and memory, specifically by using distant visual cues. Rodents are motivated to find the target location due to their dislike for open and bright spaces. To succeed in this task, rodents need to use their hippocampal-dependent spatial reference memory. Unlike MWM, the animals are not exposed to a stressful situation such as being placed in water and forced to swim (a strong aversive stimulus).
Since aging mouse models may be more susceptible to stress and anxiety, using a Barnes Maze can be a solution to this confounding factor.
Object Recognition Test
The Object Recognition Test is a visual learning and memory task based on rodents’ innate curiosity to explore novel objects, a motivation that is also seen in humans.
This is a useful test to determine whether a therapeutic has effects on behaviors that have translational value to humans.
Open Field Test
The Open Field Test is a popular assay for locomotion, exploratory behavior, and anxiety. The task exploits the innate fear of open, bright spaces to evaluate anxiety-like behaviors.
Animals with high levels of anxiety will show reduced locomotion and exploration in this task.
Future Avenues of Brain Aging Research
As neurodegeneration researchers shift gears towards targeting aging as a disease model and therapeutic intervention, several future avenues are promising for pre-clinical researchers to explore, such as identifying:
- metabolic markers for early diagnosis of brain aging
- metabolic markers for reliable measurement of the progression of brain aging and related neurodegenerative diseases
- therapeutic and lifestyle interventions that can slow, arrest, or reverse the progression of brain aging
- mechanistic relationships between cellular pathways of aging and specific symptoms of neurodegenerative diseases
- genetic predispositions and/or environmental factors which may increase the risk of brain aging onset and neurodegenerative diseases later on
Conclusion
In summary, neurodegenerative diseases are highly interactive with the aging processes. Their disease-defining features are often not the root causes and effective targets for treatment.
Finding the mechanisms and effective interventions for neurodegeneration requires us to treat aging as a disease model and therapeutic target. Since aging encompasses multiple hallmarks and myriad cellular processes, there are potentially many avenues worth exploring. Among them, metabolism is one that exacerbates all hallmarks of aging and it is therefore particularly relevant for future research. Animal models of aging and behavioral assays will continue to be fundamental tools in discovery and translational studies, contributing to our eventual success in treating and preventing neurodegenerative diseases.
References
- Mattson, M.P., and Arumugam, T.V. (2018). Hallmarks of Brain Aging: Adaptive and Pathological Modification by Metabolic States. Cell Metab 27, 1176-1199.10.1016/j.cmet.2018.05.011
- Hou, Y., Dan, X., Babbar, M., Wei, Y., Hasselbalch, S.G., Croteau, D.L., and Bohr, V.A. (2019). Ageing as a risk factor for neurodegenerative disease. Nat Rev Neurol 15, 565-581.10.1038/s41582-019-0244-7
- Lopez-Otin, C., Blasco, M.A., Partridge, L., Serrano, M., and Kroemer, G. (2013). The hallmarks of aging. Cell 153, 1194-1217.10.1016/j.cell.2013.05.039
- Deture, M.A., and Dickson, D.W. (2019). The neuropathological diagnosis of Alzheimer’s disease. Mol Neurodegener 14, 32.10.1186/s13024-019-0333-5
- Lee, J., Hong, S., & Kim, Y. (2019). Diagnosis of Alzheimer’s disease utilizing amyloid and tau as fluid biomarkers. Experimental & Molecular Medicine, 51(5), 1–10. https://doi.org/10.1038/s12276-019-0250-2
- Folch, J., Ettcheto, M., Petrov, D., Abad, S., Pedros, I., Marin, M., Olloquequi, J., and Camins, A. (2018). Review of the advances in treatment for Alzheimer disease: Strategies for combating beta-amyloid protein. Neurologia 33, 47-58.10.1016/j.nrl.2015.03.012
- Bonomini, F., Rodella, L.F., and Rezzani, R. (2015). Metabolic syndrome, aging and involvement of oxidative stress. Aging Dis 6, 109-120.10.14336/AD.2014.0305
- Skouta, R. (2016). Neuroprotective effect of antioxidant compounds. Neural Regeneration Research, 11(4), 566–567. https://doi.org/10.4103/1673-5374.180738
- Folgueras, A.R., Freitas-Rodriguez, S., Velasco, G., and Lopez-Otin, C. (2018). Mouse Models to Disentangle the Hallmarks of Human Aging. Circ Res 123, 905-924.10.1161/CIRCRESAHA.118.312204
- Miyamoto, M. (1997). Characteristics of age-related behavioral changes in senescence-accelerated mouse SAMP8 and SAMP10. Exp Gerontol 32, 139-148.10.1016/s0531-5565(96)00061-7
- Corpuz, H.M., Ichikawa, S., Arimura, M., Mihara, T., Kumagai, T., Mitani, T., Nakamura, S., and Katayama, S. (2018). Long-Term Diet Supplementation with Lactobacillus paracasei K71 Prevents Age-Related Cognitive Decline in Senescence-Accelerated Mouse Prone 8. Nutrients 10.10.3390/nu10060762
- Pang, K.C., Miller, J.P., Fortress, A., and Mcauley, J.D. (2006). Age-related disruptions of circadian rhythm and memory in the senescence-accelerated mouse (SAMP8). Age (Dordr) 28, 283-296.10.1007/s11357-006-9013-9
- Wei, H., Li, L., Song, Q., Ai, H., Chu, J., and Li, W. (2005). Behavioural study of the D-galactose induced aging model in C57BL/6J mice. Behav Brain Res 157, 245-251.10.1016/j.bbr.2004.07.003
- Ali, T., Badshah, H., Kim, T.H., and Kim, M.O. (2015). Melatonin attenuates D-galactose-induced memory impairment, neuroinflammation and neurodegeneration via RAGE/NF-K B/JNK signaling pathway in aging mouse model. J Pineal Res 58, 71-85.10.1111/jpi.12194
- He, M., Zhao, L., Wei, M.J., Yao, W.F., Zhao, H.S., and Chen, F.J. (2009). Neuroprotective effects of (-)-epigallocatechin-3-gallate on aging mice induced by D-galactose. Biol Pharm Bull 32, 55-60.10.1248/bpb.32.55
- Bodea, L.G., Evans, H.T., Van Der Jeugd, A., Ittner, L.M., Delerue, F., Kril, J., Halliday, G., Hodges, J., Kiernan, M.C., and Gotz, J. (2017). Accelerated aging exacerbates a pre-existing pathology in a tau transgenic mouse model. Aging Cell 16, 377-386.10.1111/acel.12565
- Currais, A., Farrokhi, C., Dargusch, R., Armando, A., Quehenberger, O., Schubert, D., and Maher, P. (2018). Fisetin Reduces the Impact of Aging on Behavior and Physiology in the Rapidly Aging SAMP8 Mouse. J Gerontol A Biol Sci Med Sci 73, 299-307.10.1093/gerona/glx104
- Kaviani, E., Rahmani, M., Kaeidi, A., Shamsizadeh, A., Allahtavakoli, M., Mozafari, N., and Fatemi, I. (2017). Protective effect of atorvastatin on d-galactose-induced aging model in mice. Behav Brain Res 334, 55-60.10.1016/j.bbr.2017.07.029