Definition
Self-grooming, also known as autogrooming, refers to any grooming behavior that a mouse performs on itself. When self-grooming, a mouse is cleaning its own body and fur by means of grooming-related behaviors such as licking and scratching.
Description
Self-grooming is one of the many forms of grooming behaviors that are exhibited by mice. In this form, the mouse is specifically preoccupied with grooming itself. It contrasts with allogrooming (allo in Greek means ‘other’ or ‘another’) wherein a mouse is grooming another mouse.
Self-grooming is a core behavior for mice. It is estimated that mice are grooming for about 40% of their waking time. When self-grooming, a mouse is cleaning its fur and body, thus it is a maintenance behavior.
When self-grooming is studied in detail, the sequence of cephalocaudal grooming behavior is usually measured. Cephalocaudal grooming is a specific type of self-grooming wherein the mouse grooms itself by following a specific pattern (which researchers believe is innate) that begins with grooming the face and head, then the body, and concludes at the tail.
Related Self-Grooming Behaviors
The following behaviors are implemented during self-grooming as they enable the mouse to groom itself:
- A mouse uses its tongue to lick its fur and keep it clean.
- Scratching eases itching that a mouse might feel and is done by the mouse scraping its skin with its paws.
- Nibbling is a succession of small, delicate bites that a mouse does to its fur and skin, and which allows it to efficiently clean itself.
Cephalocaudal Grooming: A type of self-grooming
Cephalocaudal grooming is a type of self-grooming behavior which refers to a very specific grooming style or sequence that the mouse applies on itself. Cephalocaudal grooming is defined by a specific set and sequence of grooming behaviors which a mouse initiates in order to maintain its hygiene and cleanliness.
During cephalocaudal grooming, the mouse performs a sequence of cleaning steps in a particular order starting at the head area, then moving down the torso to the anogenital area, and finishing with the tail.
The grooming begins with the mouse licking its paws. Then the mouse uses both its wet paws to clean the nose by making elliptical brushing movements. After it cleans the nose area, it uses single paws to clean whiskers and the area around the eyes, and then both paws to finish the head with backward and upward sweeps. After that, the mouse proceeds to clean the torso area by licking the belly and the back. The last step in the grooming sequence is the cleaning of the anogenital area and tail.
While self-grooming can be used to describe any of the related self-grooming behaviors (lick, scratch, nibble) when they are directed toward the self, cephalocaudal grooming is a term that can be used to describe the mouse’s self-grooming pattern only if the mouse follows a certain pattern or sequence. Please refer to our cephalocaudal grooming article for more details on this behavior.
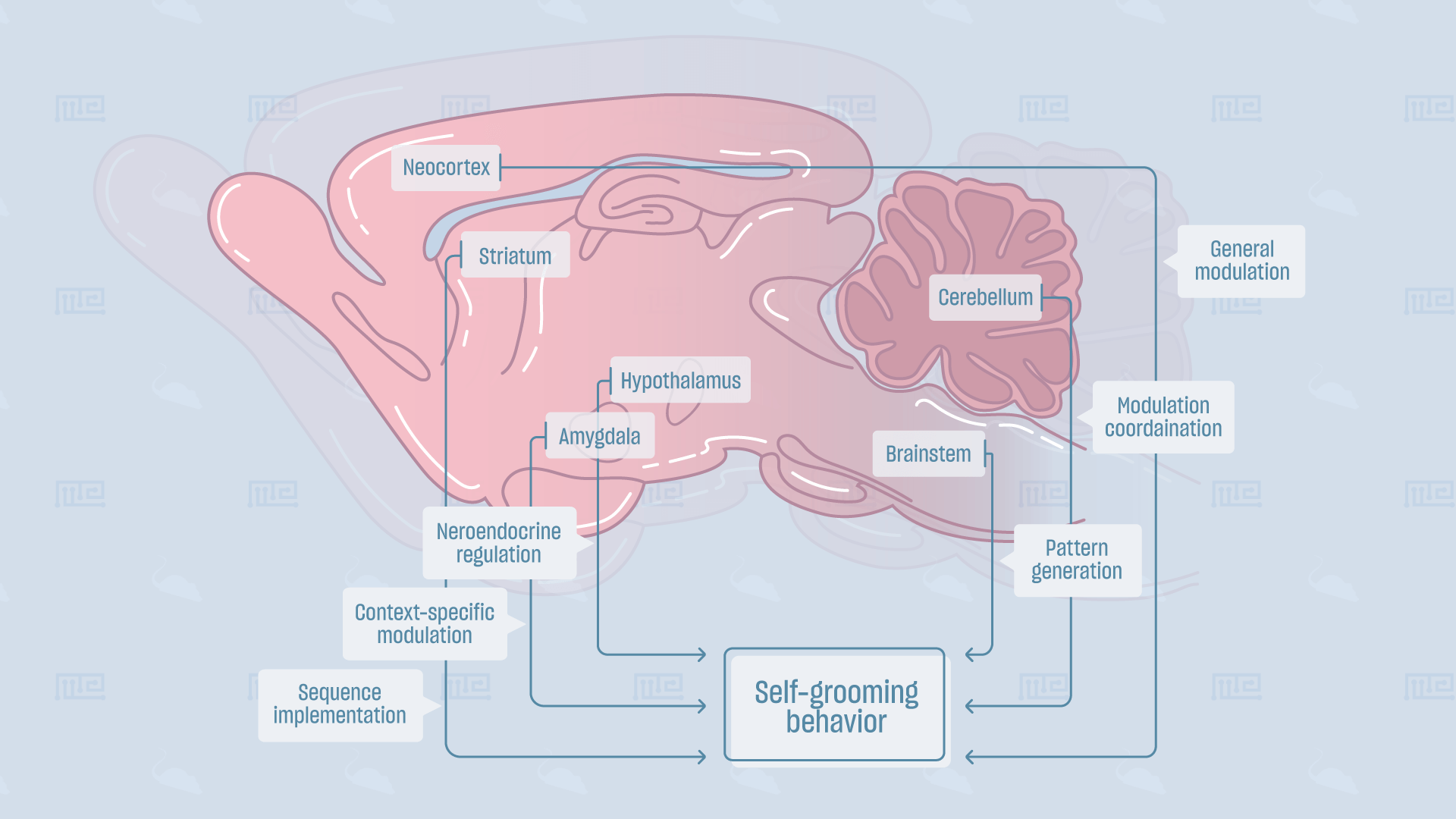
Function/Physiology of Behavior
Self-grooming is a core behavior of the mouse ethogram. A mouse’s self-grooming serves many functions, all of which ultimately aid to increase its chances of survival, including:
- For thermoregulation. Self-grooming is a means of regulating body temperature, so it is essential for survival because optimal body temperature is necessary for homeostasis. When self-grooming, the mouse will cover its fur with saliva which, when it evaporates, will cool off the mouse.
- To protect the body from foreign material. When a mouse has an injury, it may lick that wound (and the surrounding fur) in order to heal itself. Since saliva has some antimicrobial properties, such a grooming behavior may be protective by eliminating pathogens which otherwise may have caused infection and endangered the mouse’s health and survival.
- To clean its fur. Clean fur is associated with health. A mouse must clean its fur in order to maintain optimal levels of hygiene. Thus, self-grooming serves as a means of maintaining hygiene which lowers the chance of acquiring diseases.
- To self-soothe. It has been shown that self-grooming is associated with stress levels. Thus, one of its functions is to alleviate the perceived stress that a mouse has. By self-grooming, a mouse is able to self-soothe and keep its stress levels at a more stable level, thus ensuring its well-being by maintaining homeostasis. This is useful in situations where a mouse has to decrease its pain.
- To eliminate louse colonies. Lice are pests that can endanger a mouse’s health and homeostasis. Thus, self-grooming enables a mouse to rid itself from lice found on its body. However, a mouse cannot self-groom its own head, so that’s where allogrooming comes in. Thus, these two related behaviors share an overlapping function.
Application of the Behavior
Self-grooming is at the core of a mouse’s behavioral profile and takes up a good portion of a mouse’s life and daily activities. Since self-grooming has many functions, it also has many applications and points where it can be observed, including:
- Under low-stress levels. Under low-stress levels, a mouse may perceive itself to be in a safe environment and thus perform self-grooming as a part of its daily routine. Under low-stress levels, instances of uninterrupted self-grooming in a cephalocaudal manner are more likely to be observed.
- Under high-stress levels. If a mouse has high levels of stress and is in a safe environment (eg., not needing to escape immediately from a predator or threat), it may perform self-grooming as a means of self-soothing and bringing its physiological state back to baseline. In contrast to non-stressful or low-stress levels where a mouse is in a ‘comfort’ situation and is likely to elicit bouts of cephalocaudal grooming, under high-stress levels a mouse is more likely to display short bouts of grooming which occur rapidly and do not follow a particular pattern.
- When the mouse feels dirty. If a mouse feels like it is not clean, such as when its fur is dirty or when it must clean its wound, a mouse will initiate self-grooming in order to restore its cleanliness back to the desired levels.
- Before and after the diurnal sleep period. Mice are likely to be observed grooming just before sleeping and after waking up. Thus, self-grooming is an important behavior of a mouse’s everyday routine.
Research Techniques
A variety of research techniques can be used for studying self-grooming mice, including:
- Behavioral studies. Behavioral studies are valuable as they allow the researcher to profile, describe, and quantitatively evaluate how mice self-groom under various conditions. By using a variety of behavioral tests which assess cognition and physical functioning, self-grooming can be observed and better understood in the context of everyday functioning or maladaptive repetition.
- Pharmaceutical studies. Pharmaceutical studies involve the administration of drugs and supplements to mice and the subsequent observation of the influence they have on their self-grooming profile. By observing the effect of pharmaceutical substances (be they pharmacological or non-pharmacological), researchers can get identify relationships between specific molecular pathways and self-grooming. Such an approach is especially useful in the context where self-grooming is abnormally high or low due to abnormalities such as neuropsychological disorders, more on this later.
- Genetic studies. Genetic studies aim to identify, target, and manipulate the genes which might be involved in self-grooming. Just like pharmaceutical studies, genetic studies are valuable for research that focuses on abnormalities which influence behavior and thus lead to a high or low expression of self-grooming.
Behavioral tests for assessing the behavior
- Grooming Behavior Test. The grooming behavior test involves adult male mice, about 5 months old. For two weeks the mice are placed in individual housings and videotaped for 2 hours between 7 and 9 pm, under red lighting. The mice grooming includes face wiping, scratching and rubbing of the head area including ears, and cleaning of the entire body. With Noldus Observer software, the grooming behavior is manually coded and analyzed determining the total grooming duration within the 2 hours.
- Social Interaction Test. The social interaction test involves introducing a new mouse to an already acquainted group and observing the interaction behavior. The mice are placed in a cage for 15 minutes to get accustomed to it, and then a new mouse (with the same sex and from the same strain)is brought in for a duration of 10 minutes. The social interaction that involves sniffing, chasing, allogrooming and mounting is digitally recorded and determined by observers not previously apprised with the particulars of the testing conditions and genotype. When the mouse is not interacting with other mice, instances of self-grooming can also be quantified.
- Two-Trail Direct Interaction Test. This test is done in an opaque white open field in order to assess social memory. Instances of self-grooming are also observed and recorded during the test. In order to run it, one day before the testing, the mice are given 20 minutes to familiarize themselves with the testing grounds. They get another five minutes on the day of the test before a novelty mouse (trail 1) is introduced for social interaction. The novelty mouse is removed after the first testing. After 1 hour it is either reintroduced or another novelty mouse (trail 2) is put in the field. The social interactions between mice involve anogenital and nose-to-nose sniffing, allogrooming, and following at the 2 cm distance. Any aggressive encounters are blocked and excluded from analysis. Although mice will interact and groom with each other as a means of social interaction, there are also moments when a mouse will be busy self-grooming.
- The Spray Test. In the spray test, a mouse is sprayed once with a mist of water. Then, it is placed in a clean cage with ad-lib water and food available to it. After this, its behavior is recorded for 15 minutes. The video will be analyzed by viewers who are trained to assess specific forms of grooming behaviors. Sometimes, the protocol may vary and a mouse will be sprayed 3 times (instead of once) and will be placed in plastic cylinders (instead of cages) in order for grooming to be analyzed. The spray test is a simple way to elicit grooming behaviors in an experimental setting where water is the stimulus that triggers the behavior. A variation of this test is the “Splash Test” wherein the mouse is sprayed with water containing sucrose which creates a noticeable change to the mouse’s fur. Thus, a mouse that delays initiating self-grooming is scored as having decreased motivation which parallels to depression and apathy.
- The Visible Burrow System. This test uses a cage designed to mimic mice’s natural environment. It’s arrangement has an open area linked side panels which resemble runnels, thus creating an environment optimal for a wide range of behaviors to occur. Before an experiment begins, mice are individually labeled or marked with hair dye and introduced to the setting at the beginning of their dark cycle. A video recording system is used to capture the mice’s behaviors over a long period of time. Some experiments which use the visible burrow system can last for weeks.
Pharmaceutical Studies on the Behavior
Pharmaceutical studies have shown that a variety of different substances can affect self-grooming behavior.
XAV939 Injection Increases Self-Grooming Behaviors
XAV939 is a small compound that can be administered to developing embryonic mice via in utero injections. XAV939 affects mouse development, including the way that the neocortex forms, which, in turn, later affects observable behaviors such as self-grooming. Mice that receive XAV939 injections in utero have increased levels of the Axin protein in radial glia cells. Such high levels of Axin affect cellular processes and ultimately lead to heightened intermediate progenitors and thus excess levels of pyramidal neurons within the neocortex. Therefore, overproduction of pyramidal neurons in the neocortex as caused by XAV939 injections leads to a neocortical malfunction (due to malformation) which further affects observable behavior. Experimental mice injected with XAV939 display behavioral deficits resembling those found in autism, such as excessive repetitive behaviors. Mice receiving this injection spend significantly more time self-grooming (and also digging in the Marble Burying Test). The high instances of self-grooming due to the XAV939 injection demonstrates how malformation in high-order brain regions such as the neocortex can cause an imbalance in excitatory and inhibitory brain systems, ultimately leading to the manifestation of repetitive behaviors.
Valproic Acid Increases Repetitive Self-Grooming in Mice
Valproic acid is a form of valproate and may be used as a medication to treat medical conditions such as epilepsy, bipolar disorder, and migraines. In the United States, valproic acid is considered to be a safe medicine, is frequently used in health systems, and is even available as a generic medication. However, prenatal exposure to valproic acid has been demonstrated to be associated with an increased risk in autism. In animal research, mouse models of autism which were subjected to prenatal exposure to valproic acid are commonly used. In fact, mice that are exposed to valproic acid will demonstrate significantly higher levels of repetitive self-grooming than mice that are not exposed. The leading theory for which this behavioral change hypothesizes that valproic acid changes neuronal circuitry which leads to an imbalance between excitatory/inhibitory signaling, causing an increase in excitation and a decrease in inhibition. Thus, in terms of a mouse that has been exposed to valproic acid, high levels of excitation will be seen as higher frequencies and time spent self-grooming due to the inability to inhibit such actions.
Fluoxetine Reduces Elevated Self-Grooming in Ninj1 Knockout Mice
Fluoxetine is able to reduce elevated levels of self-grooming in mice with the Ninjurin 1 (Ninj1) cell adhesion molecule knocked out. (More specifics on this mouse strain can be found in the forthcoming section.) Ninj1 knockout mice lack a protein that is typically found on the cell membrane and involved in cell adhesion. Their behavioral phenotype includes high levels of self-grooming which results in progressive hair loss. Chronic treatment with fluoxetine, a drug that belongs to the selective serotonin reuptake inhibitor (SSRI) class and can be used for treating depression, has been shown to alleviate this mouse strain’s compulsive levels of self-grooming and is also able to decrease their high levels of anxiety-like behavior when given at a dose of 18 mg/kg via water for the span of 3 weeks.
MPEP Reduces Repetitive Self-Grooming
Administration of 2-methyl-6-(phenylethynyl)-pyridine (MPEP) leads to a reduced of repetitive self-grooming in BTBR mice, a particular mouse strain that is commonly used in animal research for the purposes of modeling autism spectrum disorders. MPEP is a mGluR5 metabotropic glutamate receptor antagonist. Since MPEP acts on mGluR5 receptors, it can affect the processes which mGluR5 receptors are involved in, including the regulation of N-methyl-D-aspartate (NMDA) receptors’ phosphorylation status and NMDA receptor-related neurotransmission. Thus, when BTBR mice are given MPEP there is a significant decrease in their high levels of self-grooming as a result. This change demonstrates that some alternation in NMDA receptors may be the reason that repetitive levels of self-grooming occur within the BTBR strain. Furthermore, MPEP administration is also able to significantly decrease repetitive self-grooming in other mice models of autism such as the commonly utilized valproic acid model.
Mouse Strains and Self-Grooming
Self-grooming is a behavior that is innate to all mice since it is a behavior that serves many functions which aid in survival, such as reducing stress or maintaining cleanliness. However, levels of self-grooming vary according to mouse strain.
NINJ1 Knockout Mice
Abnormalities to the Ninjurin 1 (Ninj1) cell adhesion molecule is associated with an abnormal phenotype which affects behavior. Cell adhesion molecules are located on the cell membrane and are implicated in binding with the cellular matrix or with other cells, thus making cell adhesion possible. Therefore, the integrity of cell adhesion molecules like Ninj1 is crucial for homeostasis. In Ninj1 knockout mice, behavior is altered when compared to mice that do not have this gene missing. Ninj1 knockout mice exhibit anxiety and high instances of repetitive behaviors which include increased levels of self-grooming behaviors. While Ninj1 mice have increased levels of ionotropic glutamate receptors, they also have reduced levels of glutamate, thus exhibiting glutamatergic abnormalities as well.
SAPAP3-mutant Mice
The SAP90/PSD95-associated protein 3 (SAPAP3, sometimes also referred to as DLGAP3 in scientific literature) is a scaffolding protein found in excitatory postsynapses and is highly expressed in the striatum, a part of the brain that is involved in automating movement. SAPAP3-mutant mice have this scaffolding protein genetically deleted and are frequently used in animal research for the purposes of modeling obsessive-compulsive disorder (OCD), an anxiety-spectrum disorder which is defined by unwanted persistent thoughts (referred to as ‘obsessions’) and repetitive actions (known as ‘compulsions’). SAPAP3-mutant mice have defects in their cortico-striatal synapses and a behavioral profile which reflects OCD-like behaviors, such as increased anxiety combined with elevated self-grooming behavior which persists in many cases until a mouse develops skin lesions and facial hair loss.
BTBR Mice
BTBR T+tf/J (BTBR) mice are typically used for modeling autism spectrum disorders in mice since their behaviors are consistent with the three diagnostic criteria for autism spectrum disorders. This inbred mouse strain was first developed at Columbia University by crossing mice which carried the wildtype T (brachyury) gene with mice that carried the tufted (tf) mutation. BTBR mice also have brain malformations, inclusive abnormalities of the corpus callosum and a significant reduction of volume of the hippocampal commissure. As mentioned previously, BTBR mice are characterized by abnormal behaviors, including impaired communication and social interaction, as well as increased levels of repetitive behaviors including higher bouts of self-grooming. In an experimental setting, when BTBR mice are placed in an environment supporting free social interaction they demonstrate lower levels of allogrooming and play behavior and high levels of self-grooming.
BALB/C Mice
BALB/C mice are commonly used in animal research. They are characterized by their sociability deficits and are used to model autism spectrum disorders, but can also be used to model other neurological diseases and tumors as well. Even though BALB/C mice are used to characterize autism, they demonstrate low self-grooming levels. This stands in contrast to other mouse models of autism which display high levels of repetitive self-grooming.
SHANK3 Mice
SHANK3 is a postsynaptic protein found at glutamatergic synapses and is involved in establishing normal development of connectivity in the brain. In general, proteins which belong to the Shank family are thought to work as scaffolding proteins which help orchestrate the postsynaptic signaling complex of glutamatergic synapses. Disruption of the Shank3 gene is believed to be the major cause of the 22q13 deletion syndrome (also known as the Pheland-McDermid syndrome) which is an autism spectrum disorder. Shank3 mutant mice are used to study the relationship between this gene, behavior, and autism spectrum disorder. Shank3 mutant mice exhibit repetitive self-grooming to the point it becomes a self-injurious behavior.
R6/2 Mice
R6/2 mice are transgenic mice used for modeling Huntington’s disease in animal research studies. R6/2 mice express exon 1 of the human Huntington’s disease gene with about 150 CAG trinucleotide repeats (i.e., the repetition of cytosine, adenine, and guanine). In R6/2 mice, the human huntingtin promoter drives the transgene expression which results to a 75% endogenous huntingtin expression. From all Huntington’s disease mouse models to date, R6/2 develop this disease the fastest and have the highest expressions of the huntingtin protein across different levels of the brain. These mice also show elevated levels of self-grooming, resembling stereotypy, combined with other changes in motor function, including altered gait patterns and involuntary movements.
HdhQ111/Q111 Mice
HdhQ111/Q111 mice are used to model Huntington’s disease since they carry an expanded polyglutamine stretch in the protein that is associated with Huntington’s disease. But, unlike other models, HdhQ111 mice show only a mild phenotype combined with a relatively slow disease progression. Furthermore, these mice demonstrate changes in locomotor function as well as an anxio-depressive-like phenotype. In females, decreased grooming duration is observed, indicating that this mouse strain exhibits a sex and genotype interaction. In this context, female mice which exhibit decreased grooming behaviors are interpreted as being in a depressive-live state which is not observed in male same-strain mice (which are more likely to demonstrate an anxiety-like phenotype).
Slitrk5 Knockout Mice
Slitrk5 is a neuron-specific transmembrane protein whose function is still unknown. However, mice that have the gene for Slitrk5 knocked out have alterations in their grooming pattern which is similar to the pathological grooming (combined with anxiety) that can be observed in SAPAP3 mice (which were discussed earlier). Slitrk5 mice demonstrate reduced neuronal transmission at corticostriatal synapses and reduced levels of the corticostriatal synapses NR1 and NR2B. Although Slitrk5 mice are used experimentally to model obsessive-compulsive disorder, genetic evidence which links the Slitrk5 gene with obsessive-compulsive disorder in humans is yet to be established.
Abnormalities in Self-Grooming
Altered self-grooming profiles can be observed in mice which certain abnormalities. By studying various abnormalities, such as neuropsychological conditions like autism, researchers can examine and get a better understanding of how self-grooming is affected, enabling them to develop insights on the underlying mechanisms and possible treatment for restoring normal grooming patterns.
Autism
Autism spectrum disorders (ASDs) are typically characterized by a similar neuropsychological deficits profile which includes limited social interactions, reduced communication, and repetitive behaviors. ASDs affect many children, people, and families and receive a lot of research attention including from scientists who make use of animal models.
Autism-like features are observed in mouse strains such as BTBR and SHANK3 mice. These two strains demonstrate increased frequency of self-grooming in combination with autism-specific deficiencies such as lack of social interaction.
Huntington’s Disease
Huntington’s disease is an autosomal dominant disorder. It occurs due to the expression of the huntingtin protein which expands and forms an abnormal protein, ultimately damaging brain cells. In mice modeling Huntington’s disease, altered levels of self-grooming can be observed when compared to wild-type controls. For example, by the time that R6/2 mice (the most commonly used mouse strain for modeling Huntington’s disease) reach 13 weeks of age, they will spend twice the time grooming when compared to wild-type controls.
Prion Disease
Prion disease can be acquired through exposure, spontaneously, or genetically. It is a disease characterized by the misfolding of the prion protein (a protein whose function is still not completely understood by scientists) which ultimately leads to dementia, severe ataxia, and even death. In animal research, the most common way to induce prion disease in mice is by infecting wild type mice with certain strains of prion. After being inoculated with prion disease for about 5.5 months, mice will begin to show decreased levels of self-grooming behavior when compared to uninoculated controls. However, even though mice with prion disease show decreased levels of self-grooming, they show hyperactivity in terms of locomotion.
Disease Models of Self-Grooming
Autism Disease Models and Self-Grooming
Since autism disease is increasing in prevalence and more children and adults are being diagnosed as being on the spectrum, autism is receiving a lot of attention from behavioral scientists. Animal models are used in order to better understand ASDs and how behavior varies in comparison to controls. Self-grooming is a variable of interest to behavioral scientists studying ASDs since it has an inverse relationship with other social behaviors. In other words, as instances of self-grooming increase, a decrease of other social behaviors is likely to be observed. Therefore, self-grooming is a behavior that is of interest to behavioral scientists interested in understanding and potentially treating ASDs. Autism disease models can be modeled in mice in many different ways, including:
Genetic Manipulation
By genetically manipulating mice, certain genes can be targeted. Therefore, scientists are able to study how observed behaviors are altered after manipulation on a cellular level. Mice such as BTBR, SHANK3, and SAPAP3-mutant mice, as mentioned previously, may be used for the purposes of studying self-grooming in ASDs.
Chemical Induction
ASDs can also be chemically induced in mice via injections of valproic acid, as mentioned previously. Since valproic acid exposure is associated with ASDs in humans, it becomes a relevant model for animal research. Mice that have been exposed to valproic acid also display behaviors that are analogous to those seen in humans with ASDs, including increased repetitive behaviors (seen as increased self-grooming in mice) and decreased social interactions.
Huntington’s Disease Models and Self-Grooming
As mentioned previously, Huntington’s disease models show altered levels of self-grooming. R6/2 transgenic mice and HdhQ111 mice are two commonly used mouse strains for studying Huntington’s disease in mice. While R6/2 mice show an increase of stereotypic self-grooming, HdhQ111 mice (especially females) demonstrate a decrease in self-grooming behaviors which is associated with a depressive-like phenotype.
Obsessive-Compulsive Disorder and Self-Grooming
Since self-grooming already takes up such a large proportion (about 40%) of a mouse’s waking time, any increase of its frequency is bound to be problematic. OCD models of self-grooming are associated with higher levels of self-grooming which, in some cases, even become self-injurious.
Using genetic mouse models is the most common method for studying OCD in mice. The SAPAP3 and slitrk5 mouse strains are used frequently in scientific research for the purposes of studying repetitive behaviors like self-grooming within an OCD model.
Although mouse models of repetitive, compulsive behavior which exhibit high levels of self-grooming are heuristically similar to OCD, they are sometimes also used to model other neuropsychiatric disorders with similar phenotypes including trichotillomania, Tourette syndrome, excoriation disorder, onychophagia, and autism.
Summary
- Self-grooming, also known as autogrooming, refers to any grooming behavior that a mouse performs on itself.
- Self-grooming is a core behavior for mice. It is estimated that mice are grooming for about 40% of their waking time.
- When self-grooming is studied in detail, the sequence of cephalocaudal grooming behavior is usually measured
- Licking, scratching, and nibbling are all behaviors which are related to self-grooming.
- Since self-grooming is a core mouse behavior, it has many functions, including thermoregulation, protection against foreign material, fur cleaning, self-soothing, and elimination of louse colonies.
- This behavior is commonly observed under low or high stress levels, when the mouse feels dirty, or just before or after the diurnal sleep period.
- Self-grooming can be studied using behavioral, pharmaceutical, and/or genetic studies.
- The following behavior tests are used to assess self-grooming: the grooming behavior test, the social interaction test, the two-trail direct interaction test, the spray test, and the visible burrow system.
- Pharmaceutical studies on the behavior have uncovered that self-grooming can be increased in mice through the administration of substances like XAV939 and valporic acid. Self-grooming can be reduced through the administration of substances like fluoxetine and MPEP.
- The following mouse strains have a specific self-grooming pattern: Ninj1 knockout mice, SAPAP3-mutant mice, BTBR mice, BALB/C mice, SHANK3 mice, R6/2 mice, HdhQ111/Q111 mice, and slitrk5
- Several abnormalities have been found to affect self-grooming profiles in mice, including autism, Huntington’s disease, and prion disease.
- Disease models can be used to study how self-grooming varies with respect to disease.
- Autism spectrum disease models, induced via chemical induction or genetic manipulation, can lead to significant changes in self-grooming.
- Huntington’s disease models also affect self-grooming. Depending on which genetic model is used, the outcome can be a significant increase or decrease in self-grooming.
- OCD mouse models are associated with high levels of self-grooming which, in most cases, may even result in personal injury.
References
- Grados, M., Prazak, M., Saif, A., & Halls, A. (2016). A review of animal models of obsessive-compulsive disorder: a focus on developmental, immune, endocrine and behavioral models. Expert opinion on drug discovery, 11(1), 27-43.
- Spruijt, B. V., Van Hooff, J. A., & Gispen, W. H. (1992). Ethology and neurobiology of grooming behavior. Physiological Reviews, 72(3), 825-852.
- Richardson, C. A. (2015). The power of automated behavioural homecage technologies in characterizing disease progression in laboratory mice: A review. Applied Animal Behaviour Science, 163, 19-27.
- Zhou, Y., Kaiser, T., Monteiro, P., Zhang, X., Van der Goes, M. S., Wang, D., … & Wells, M. (2016). Mice with Shank3 mutations associated with ASD and schizophrenia display both shared and distinct defects. Neuron, 89(1), 147-162.
- Tanaka, M., Sato, A., Kasai, S., Hagino, Y., Kotajima-Murakami, H., Kashii, H., … & Hall, F. S. (2018). Brain hyperserotonemia causes autism-relevant social deficits in mice. Molecular autism, 9(1), 60.
- Zhang, J. B., Chen, L., Lv, Z. M., Niu, X. Y., Shao, C. C., Zhang, C., … & Lang, B. (2016). Oxytocin is implicated in social memory deficits induced by early sensory deprivation in mice. Molecular brain, 9(1), 98.
- Fang, W. Q., Chen, W. W., Jiang, L., Liu, K., Yung, W. H., Fu, A. K., & Ip, N. Y. (2014). Overproduction of upper-layer neurons in the neocortex leads to autism-like features in mice. Cell reports, 9(5), 1635-1643.
- Burket, J. A., Herndon, A. L., Winebarger, E. E., Jacome, L. F., & Deutsch, S. I. (2011). Complex effects of mGluR5 antagonism on sociability and stereotypic behaviors in mice: possible implications for the pharmacotherapy of autism spectrum disorders. Brain research bulletin, 86(3-4), 152-158.
- Le, H., Ahn, B. J., Lee, H. S., Shin, A., Chae, S., Lee, S. Y., … & Seo, J. H. (2017). Disruption of Ninjurin1 Leads to Repetitive and Anxiety-Like Behaviors in Mice. Molecular neurobiology, 54(9), 7353-7368.
- Welch, J. M., Lu, J., Rodriguiz, R. M., Trotta, N. C., Peca, J., Ding, J. D., … & Dudek, S. M. (2007). Cortico-striatal synaptic defects and OCD-like behaviours in Sapap3-mutant mice. Nature, 448(7156), 894.
- Meyza, K. Z., Defensor, E. B., Jensen, A. L., Corley, M. J., Pearson, B. L., Pobbe, R. L., … & Blanchard, R. J. (2013). The BTBR T+ tf/J mouse model for autism spectrum disorders–in search of biomarkers. Behavioural brain research, 251, 25-34.
- Burket, J. A., Herndon, A. L., Winebarger, E. E., Jacome, L. F., & Deutsch, S. I. (2011). Complex effects of mGluR5 antagonism on sociability and stereotypic behaviors in mice: possible implications for the pharmacotherapy of autism spectrum disorders. Brain research bulletin, 86(3-4), 152-158.
- Peça, J., Feliciano, C., Ting, J. T., Wang, W., Wells, M. F., Venkatraman, T. N., … & Feng, G. (2011). Shank3 mutant mice display autistic-like behaviours and striatal dysfunction. Nature, 472(7344), 437.
- Li, J. Y., Popovic, N., & Brundin, P. (2005). The use of the R6 transgenic mouse models of Huntington’s disease in attempts to develop novel therapeutic strategies. NeuroRx, 2(3), 447-464.
- Orvoen, S., Pla, P., Gardier, A. M., Saudou, F., & David, D. J. (2012). Huntington’s disease knock-in male mice show specific anxiety-like behaviour and altered neuronal maturation. Neuroscience letters, 507(2), 127-132.
- Richardson, C. A. (2015). The power of automated behavioural homecage technologies in characterizing disease progression in laboratory mice: A review. Applied Animal Behaviour Science, 163, 19-27.
- Steele, A. D., Jackson, W. S., King, O. D., & Lindquist, S. (2007). The power of automated high-resolution behavior analysis revealed by its application to mouse models of Huntington’s and prion diseases. Proceedings of the National Academy of Sciences, 104(6), 1983-1988.