Racetams are a wide category of synthetic nootropics. The first racetam ever to be discovered was in 1972 by Corneliu E. Giurgea, a Romanian chemist and psychologist and that racetam was dubbed piracetam.[1][2] Needing a word to describe piracetam’s ability to enhance cognition, it was termed to be a “nootropic” (a word derived from Greek, νοῦς (nous) means “mind” and τρέπειν (trepein) means “to turn”) and that was the first time the word “nootropic” was used to refer to cognition-boosting supplements. All because of piracetam.
Since then, research on piracetam has expanded and many other related compounds have been developed, thus creating a wide class of supplements, collectively known as “racetams”.
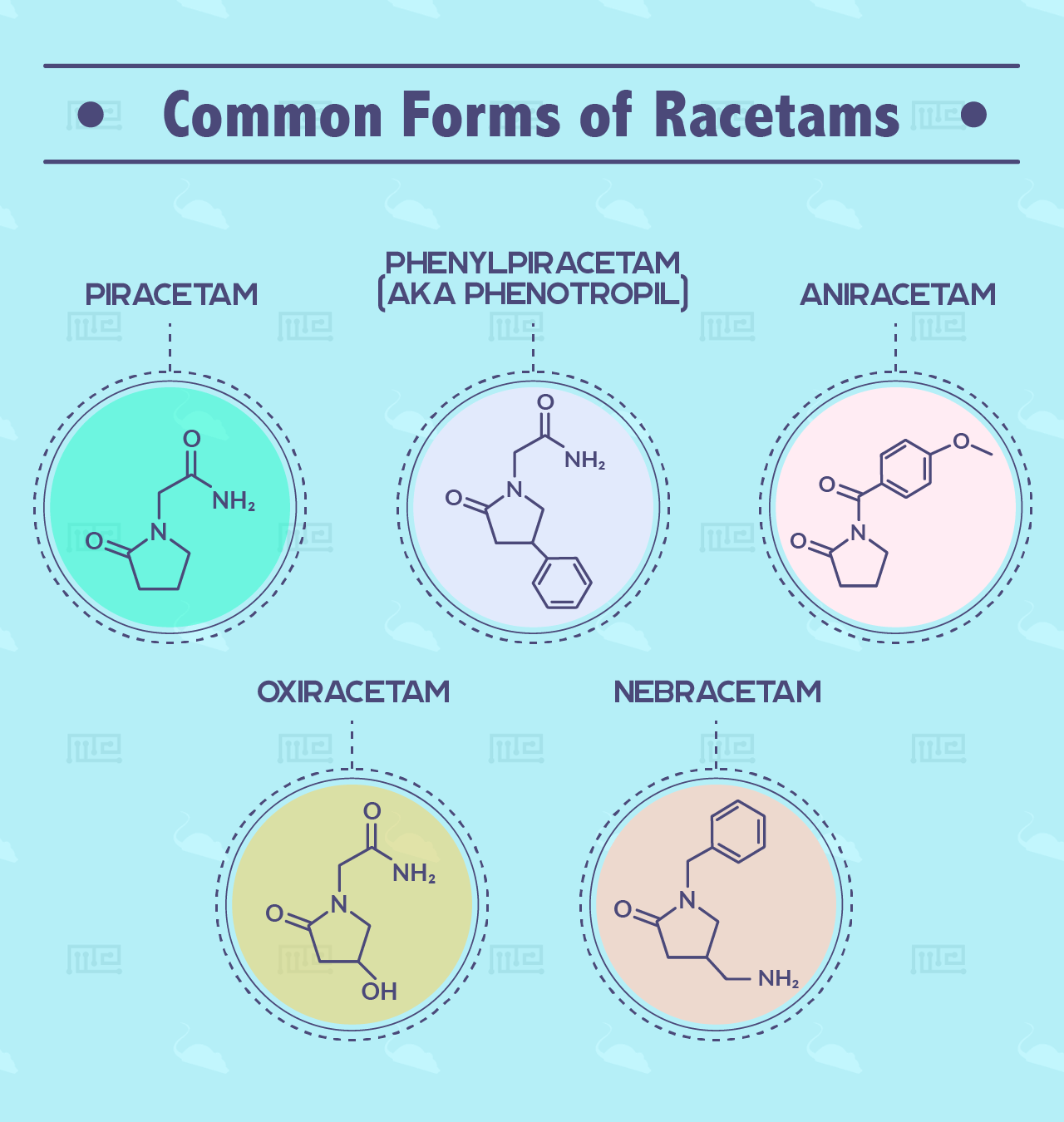
The following are the most common forms of racetams, of which some still need to be studied and some already serve as popular supplements for boosting cognition:
- Piracetam
- Phenylpiracetam (AKA phenotropil)
- Aniracetam
- Oxiracetam
- Nebracetam
In this article, we will take a closer look at each of these racetams and examine their effects on cognition and behavior as revealed by the use of animal models, testing, data collection and subsequent analysis.
The First Nootropic Ever: Piracetam
As mentioned earlier, piracetam was the first racetam ever to be discovered. It is was also the first nootropic ever, labeled as such due to its ability to heighten cognition. Piracetam has been found to enhance cognition in a variety of animal models.
Chemically, piracetam is a derivative of gamma-aminobutyric acid (GABA), the most abundant inhibitory neurotransmitter when it comes to the mammalian central nervous system. GABA’s major purpose in the brain is to reduce neuronal excitability and has been clinically used to reduce anxiety, treat attention deficit disorder, and improve mood. Therefore, since piracetam is a derivative of GABA and GABA is such an important contributor to the brain, piracetam may leave a certain mark on behavior and cognition.
Piracetam is also believed to be able to exert antioxidant properties, increase blood flow, normalize ATP metabolism, increase cellular glucose use, and stimulate ribosome function and phospholipid synthesis.[3]
Piracetam Improves Cognitive Deficits in Hypoperfusive Rats
Cerebral hypoperfusion refers to insufficient blood flow in the brain, thus creating neuronal damage. This pathophysiology is related to vascular dementia, a particular form of dementia that is produced by problems in vasculature (ischemia, hemorrhage, or hypoxia).
In an experiment using rats with chronic cerebral hypoperfusion, piracetam was demonstrated to be able to lessen cognitive impairments.[4] In this same experiment, special note was taken of the subsequent amino acid profile and neuronal behavior which resulted due to piracetam supplementation.
Cerebral hypoperfusion was modeled in this experiment by means of occluding the bilateral common carotid arteries which in turn caused neuronal damage and amnesic effects that impacted both behavior and brain physiology. This vascular dementia model was developed in 2006 by Guang and Du.[5]
The rats that had cerebral hypoperfusion showed decreased abilities in their spatial learning and had higher memory deficits than the control group, as established due to behavioral differences in the Morris Water Maze, a staple test in examining neurocognition in experimental animals. The ischemic rats had more difficulty in locating the water maze’s hidden platform and spent less time in the target quadrant (in which the hidden platform was located) and had longer escape latency times (indicating that it took longer to complete the task and locate the platform) than the sham-operated control group did.[4]
However, when piracetam was administered at 600 mg/kg in ischemic rats, their prolonged latency was reduced. The piracetam-treated group and the sham-operated group spent more time in the water maze’s target quadrant (and also had shorter latency time) than the hypoperfusion group did.
In terms of neurochemical assessments, piracetam supplementation was associated with reducing the morphological changes observed in the ischemic group’s cerebral cortex and hippocampal CA1 region, including reduced nuclei shrinkage and neuronal cell loss. The ischemic group also had the highest levels of the pro-apoptotic (cell-death signaling) proteins BAX and P53 in their cortical neuronal cells. The piracetam group, on the other hand, had no such abnormalities.[4]
These combined results demonstrating significant differences in behavioral and morphological characteristics between the ischemic and the piracetam-treated groups indicate that piracetam is able to impact the nervous system in notable and observable ways.
Piracetam and Choline Improves Memory in Aged Rats
In a different study, focusing on the effect of piracetam on cognitive function in aged rats, a group of researchers set out to see if piracetam can act as a cofactor.[6]
Piracetam was administered to aged rats to see if it would have a positive effect on their memory.
The experimental groups were divided as follows: control (on tap water), piracetam (200 mg/kg/day), choline (200 mg/kg/day), or combined piracetam and choline (100 mg/kg/day of each). This supplementation was carried out for a week and was considered to be chronic. Before testing, the groups were further subdivided to receive acute treatment (where additional supplementation would be given 30 minutes before training and behavioral testing) in order to determine whether the combined effect of chronic and acute supplementation would be greater or equivalent to chronic or acute supplementation alone.
All of the aged rats performed equally when their pre-training performance was measured with respect to their retention latency in the Passive/Avoidance Test. Then, when it came to the test performance one week later, significant differences in behavior between the supplementation groups emerged. In the retention phase, the groups that were solely on choline or piracetam performed better than the saline control group, indicating stronger memory abilities. However, the combined choline and piracetam group (which received both acute and chronic treatment) outperformed the other conditions by threefold.[6]
When further investigating the relationship between behavior and supplementation, the researchers found that the highest impact on cognition was on the chronic administration of piracetam and choline (100 mg/kg/day for one day) combined with acute administration of both supplements 30 minutes before testing. Significant improvements in the Light/Dark Test were also observed in the combined piracetam and choline chronic group (without acute treatment), but not as much as in the group that received both supplements in both acute and chronic time points. There was also a significant finding of improvement in the solely acute treatment of piracetam and choline (100 mg/kg of each 30 minutes per supplement) which received tap water in the week prior to testing. However, in the solely acute condition, the improvement was not as high as the other two conditions even though it was statistically significant. The various conditions and assessments demonstrate that the joint administration of piracetam and choline have the greatest effect when administered for a prolonged period of time and then once more shortly before cognitive-demanding tasks.[6]
Phenylpiracetam’s Psychoimmunomodulatory Effect
Phenylpiracetam, sometimes referred to as phenotropil, is another nootropic belonging to the racetam family and has also been shown to impact cognition in animals that have immune stress.[7]
To demonstrate phenylpiracetam’s effects, a group of scientists used rats with LPS-induced immune stress and subjected them to experimental tests. Immune stress is induced via intraperitoneal administration of Pseudomonas aeruginosa at 100 μg/kg for the span of three days. The LPS-induced immune stress model’s functional characteristics are:
- phagocytic activity of neutrophils in the peripheral blood as determined by the latex test
- an increase in the cellular delayed-type hypersensitivity index
- a positive antibody titer in the passive hemagglutination test
Experimental groups were created when the rats were divided into the following three conditions: the control group (receiving injections of saline), the experimental group with induced immune stress, and the treatment group (receiving phenylpiracetam on top of their experimentally induced immune stress). The treatment group’s phenylpiracetam dose was intraperitoneally injected at 25 mg/kg per day for the span of 5 days.
Phenotropil exerted an immunomodulatory effect. The animals’ anxiety and exploration levels were assessed using the Elevated-Plus Maze Test and the Open Field Test. The LPS-induced rodents had reduced exploratory and locomotor activities in these behavioral tests. However, the LPS-induced rodents with phenotropil treatment did not exhibit signs of sluggishness and/or anxiety as the untreated LPS-induced controls did. Instead, these rats demonstrated an increase in horizontal locomotion, crossing the open field’s central area and transitioning between the arms of the elevated-plus maze, and increased vertical rearing postures. Also, the rats which had received treatment had higher exploratory behaviors than their non-treated counterparts. The rats on phenotropil supplementation spent more time in the open arms and looking out from the closed arms towards the outside.[7]
This study established phenotropil’s effect as a psychostimulant in an immune stress model by demonstrating its enhancing effect on exploration and anxiolytic properties.
Aniracetam’s Anxiolytic Effects on Anxiety Mouse Models
Aniracetam is another nootropic supplement which belongs to the racetam family.
One study, interested in anxiety, compared the behavioral effects of aniracetam across three different behavioral tests which are typically used for examining anxiety levels in rodents (the social interaction test, the elevated-plus maze, and Fear Conditioning Test). Aniracetam was administered at various dosage levels 30 minutes before behavioral testing in two different strains of mice. Male ddY mice were used for the social interaction and fear conditioning test and male ICR mice were used for the elevated-plus maze test.[8]
When administering different dosages of aniracetam, in order to determine the relationship between dosage levels and behavior, the researchers found that giving aniracetam at 100 mg/kg to ddY mice specifically increased the amount of time they spent trunk sniffing another mouse introduced during the social interaction test. However, when given only 10 mg/kg of aniracetam, the ddY mice had significantly higher instances of following the other mouse. Additionally, it was established that aniracetam was able to increase overall social interaction time, as shown by the increased time and frequency measured by the total social interaction score.[8]
Next, to further assess anxiety levels, male ICR mice were systematically administered aniracetam. When subjecting these mice to an Elevated-Plus Maze test, the researchers found that aniracetam at 30 mg/kg was able to increase the time spent in the open arms of the maze as well as the percentage of entries into the open arms.
Furthermore, in the Fear Conditioning Test, only the mice that were on the 30 mg/kg dosage showed significantly different behavioral alterations from the vehicle controls. The ddY mice that received 30 mg/kg had a shorter freezing time. In the Fear Conditioning Test, increase in freezing time is generally associated with the development of generalized anxiety and panic disorder and is a test that is frequently used in order to induce such disorders for translational research. Aniracetam’s ability to reduce freezing time at a dosage level of 30 mg/kg means that this nootropic has anxiolytic properties in yet another anxiety-related domain.[8]
Oddly, in a different study, it was shown that aniracetam did not alter cognition or affective behavior.[9] This study used adult C57BL/6J mice which were divided into three groups: naive, placebo, and experimental (receiving aniracetam). In the Open Field Test, there was no significant difference between the control and the aniracetam-treated groups. The groups performed equally in the total distance traveled and in the total distance moving in the maze’s center. Also, there was no difference in the total sum of fecal boli produced during the test. However, in the Elevated-Plus Maze, the aniracetam-treated group produced significantly less fecal boli than the control. Besides that, there were no behavioral differences between the groups in the Elevated-Plus Maze and both groups spent equal time in the open and closed arms and traveled equal distance.[9]
Due to the varying and contradictory results, further investigation is required, in order to establish the exact ways and mechanisms involved in aniracetam’s effects on cognition and behavior.
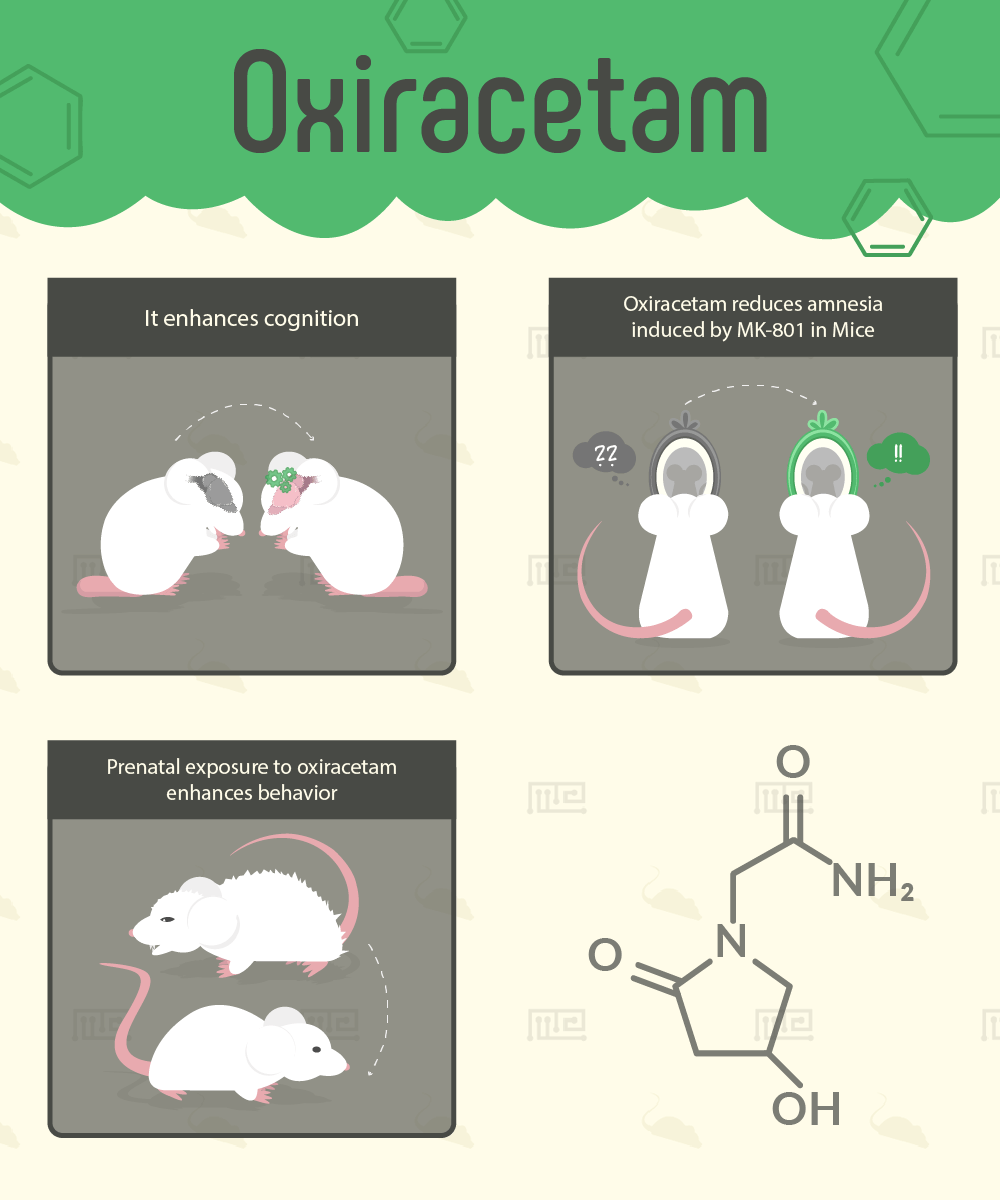
Oxiracetam Reduces Amnesia Induced by MK-801 in Mice
Oxiracetam, another nootropic belonging to the racetam family, is also able to affect behavior and cognition, according to research.
A group of scientists tested oxiracetam supplementation in mice that had amnesia induced by MK-801 which is a specific non-competitive NMDA receptor antagonist that has been shown previously as capable of inducing amnesia in numerous learning and memory tests.[10][11][12]
To test learning and memory, the researchers used a modified protocol of the Elevated-Plus Maze test. Although the Elevated-Plus Maze is typically used to measure anxiety-related behaviors, recent developments have made it possible to utilize this maze as a spatial learning and memory assessment tool.[13][14] This works by measuring the time it takes for the rodent to move from the open arm to the enclosed arm of the maze. When time is shortened between trials, spatial learning has taken place. When latency doesn’t decrease during the retention session, it suggests that the rodent cannot remember the maze’s configuration and/or that a supplement or drug has had an amnesic effect. This procedure, using the elevated plus-maze to study spatial learning, is considered to be a simple model for studying spatial memory and one that already has plenty of research supporting its efficacy and validity.[15][16]
The results showed that oxiracetam administration was able to reduce the learning and memory deficits which were associated with sole-administration of MK-801.
There were two experiments conducted. First, the mice were slightly trained on the elevated-plus maze with only 24 hours between the acquisition session and the retention session. Here, the mice were injected with oxiracetam (at either 3 mg/kg or 30 mg/kg depending on condition) immediately after the acquisition session. Then, before the retention session, MK-801 was injected.[17]
The results showed that oxiracetam was able to ward off the behavior difficulties associated with MK-801 during spatial learning. While the MK-801 mice had transfer latency times during the retention session that were similar to their times during the acquisition stage, the oxiracetam-treated mice at either dosage had significantly lower transfer latency times which were comparable to those from the control and sole oxiracetam-treated groups. The researchers interpreted such findings as oxiracetam having a significant effect on memory-related cognition.
In the second version of this experiment, the mice were more well-trained. The mice were subjected to 3 training sessions and there were two retention session (each session performed at a 24-hour interval after the previous). Here, oxiracetam was administered immediately after the third training section at 30 mg/kg. Then, 30 minutes before the first retention session, MK-801 was administered at either 0.15 mg/kg, 0.25 mg/kg, or 0.4 mg/kg, thus creating multiple experimental groups.[17]
The results showed throughout the training period schedule that there was a steady and stable decrease in transfer latency in all groups. However, statistical analysis revealed that there were significant behavioral differences in transfer latency between the first and second retention sessions. When administering solely MK-801, there was a significant increase in transfer latency. In the first retention session, only the mice treated at 0.4 mg/kg had transfer latency times that were comparable to the control group’s. However, during the second retention session, the latency score slightly decreased for the 0.25 and 0.4 mg/kg groups and significantly lower than the corresponding MK-801 groups’.
The researchers hypothesized that these results are due to the nootropic’s possible interaction with the central glutamatergic receptor function which is vital for memory and learning.
Prenatal Exposure to Oxiracetam Enhances Behavior
Another oxiracetam-focused study was conducted to examine the effects of prenatal exposure of oxiracetam on behavior.[18]
Adult CD1 nulliparous female mice were housed individually with adequate food and water for six days. On the seventh day, one male was introduced per cage. On the eighth day, the females were considered pregnant and were controlled for positive mating. On the same day until birth, half of the females were injected daily with saline solution and the other half were injected daily with 50 mg/kg of oxiracetam. When the pups were born, only the pups born to dams in which their delivery happened at 20-22 days after the first day of treatment and had litters of 10-13 pups were used for further behavioral testing.
In the Open Field Test, the pups with prenatal oxiracetam exposure performed differently than the control pups. They exhibited significantly less freezing behavior and more self-grooming. During the Novelty Object Recognition Test, the oxiracetam-treated pups had significantly higher ambulation and displayed investigation-related behavior (such as rearing and sniffing against the novel object). The control group, on the other hand, had higher rates of freezing when introduced to the novel object. Interestingly, the oxiracetam group had a higher amount of defecation boluses in the Open Field than the control group did.[18]
In an experiment using the Radial Arm Maze Task, the prenatally oxiracetam-treated mice had higher choice accuracy and had more correct trials overall. Furthermore, the treated mice had a more distinguished pattern of runs, moving in either clockwise or in a counterclockwise fashion, when compared to the controlled mice.
Nebracetam Reinstates Disrupted Spatial Cognition
Nebracetam is another nootropic within the racetam family. Nebracetam was shown to be able to restore disrupted spatial cognition and produce a cholinergic enhancing effect by a team of scientists.[19]
The researchers created a scopolamine-induced disruption of cognition animal model. This treatment reduced the noradrenaline contents in the hippocampus and frontal cortex, but this was reversed in rats given nebracetam.
In the 8-Arm Radial Maze, the nebracetam rats had significantly more correct responses and fewer performance errors than the group given only scopolamine.[19]
Such findings, highlighting both behavioral and brain changes, demonstrate that nebracetam is able to significantly impact cognition in rats.
Conclusion
In summary, racetams have a significant impact on cognition. But, research is still limited and needs to be expanded, in order to establish the exact effects produced on behavior and cognition by this nootropic class.
Although racetams are not approved by the Food and Drug Administration as prescribable pharmaceuticals, they still warrant attention by researchers. The growing body of evidence and experiments that demonstrate the cognitive enhancing effects of these supplements and the high rate of usage of these nootropics by the general public should signal the need for more experimental studies in this area. For example, in a study with healthy human volunteers, pramiracetam was shown to reduce the cognitive impairments created by scopolamine-induced amnesia.[20] Therefore, the effects cannot be ignored or neglected and the racetam family should receive further scientific attention.
Although some efforts have been made, more research is necessary to show the translational impact of nootropics in the human population.
References
- Giurgea, Corneliu. “Pharmacology of integrative activity of the brain. Attempt at nootropic concept in psychopharmacology.” Actualites pharmacologiques 25 (1972): 115.
- Winblad, Bengt. “Piracetam: a review of pharmacological properties and clinical uses.” CNS drug reviews 11.2 (2005): 169-182.
- Guang, Hong-Mei, and Guan-Hua Du. “Protections of pinocembrin on brain mitochondria contribute to cognitive improvement in chronic cerebral hypoperfused rats.” European journal of pharmacology 542.1-3 (2006): 77-83.
- He, Zhi, et al. “Piracetam improves cognitive deficits caused by chronic cerebral hypoperfusion in rats.” Cellular and molecular neurobiology 28.4 (2008): 613-627.
- Guang, Hong-Mei, and Guan-Hua Du. “Protections of pinocembrin on brain mitochondria contribute to cognitive improvement in chronic cerebral hypoperfused rats.” European journal of pharmacology 542.1-3 (2006): 77-83.
- Bartus, Raymond T., et al. “Profound effects of combining choline and piracetam on memory enhancement and cholinergic function in aged rats.” Neurobiology of aging 2.2 (1981): 105-111.
- Samotrueva, M. A., et al. “Psychoimmunomodulatory effect of phenotropil in animals with immune stress.” Bulletin of experimental biology and medicine 151.1 (2011): 51-54.
- Nakamura, Kazuo, and Mitsue Kurasawa. “Anxiolytic effects of aniracetam in three different mouse models of anxiety and the underlying mechanism.” European journal of pharmacology 420.1 (2001): 33-43.
- Elston, Thomas W., et al. “Aniracetam does not alter cognitive and affective behavior in adult C57BL/6J mice.” PloS one 9.8 (2014): e104443.
- Benvenga, Mark J., and Theodore C. Spaulding. “Amnesic effect of the novel anticonvulsant MK-801.” Pharmacology Biochemistry and Behavior 30.1 (1988): 205-207.
- Bliss, Tim VP, and Graham L. Collingridge. “A synaptic model of memory: long-term potentiation in the hippocampus.” Nature 361.6407 (1993): 31.
- Butelman, Eduardo R. “A novel NMDA antagonist, MK-801, impairs performance in a hippocampal-dependent spatial learning task.” Pharmacology Biochemistry and Behavior 34.1 (1989): 13-16.
- Itoh, Jiro, Toshitaka Nabeshima, and Tsutomu Kameyama. “Utility of an elevated plus-maze for the evaluation of memory in mice: effects of nootropics, scopolamine and electroconvulsive shock.” Psychopharmacology 101.1 (1990): 27-33.
- Itoh, Jiro, Toshitaka Nabeshima, and Tsutomu Kameyama. “Utility of an elevated plus-maze for dissociation of amnesic and behavioral effects of drugs in mice.” European journal of pharmacology 194.1 (1991): 71-76.
- Miyazaki, S., M. Imaizumi, and K. Onodera. “Ameliorating effects of histidine on learning deficits in an elevated plus-maze test in mice and the contribution of cholinergic neuronal systems.” Methods and findings in experimental and clinical pharmacology 17 (1995): 57-63.
- Miyazaki, Shuichi, Masahiro Imaizumi, and Kenji Onodera. “Ameliorating effects of histidine on scopolamine-induced learning deficits using an elevated plus-maze test in mice.” Life sciences 56.19 (1995): 1563-1570.
- Hliňák, Zdeněk, and Ivan Krejčı́. “Oxiracetam prevents the MK-801 induced amnesia for the elevated plus-maze in mice.” Behavioural brain research 117.1-2 (2000): 147-151.
- Ammassari-Teule, Martine, et al. “Enhancement of radial maze performances in CD1 mice after prenatal exposure to oxiracetam: Possible role of sustained investigative responses developed during ontogeny.” Physiology & behavior 42.3 (1988): 281-285.
- Iwasaki, Katsunori, Yoshiaki Matsumoto, and Michihiro Fujiwara. “Effect of nebracetam on the disruption of spatial cognition in rats.” The Japanese Journal of Pharmacology 58.2 (1992): 117-126.
- Mauri, Marco, et al. “Pramiracetam effects on scopolamine-induced amnesia in healthy volunteers.” Archives of gerontology and geriatrics 18.2 (1994): 133-139.